January 2018—It was baseball’s Yogi Berra who said, with the unique slant that was his hallmark, “In theory there is no difference between theory and practice. In practice, there is.” More vividly, boxer Mike Tyson once summed up the same reality when asked to comment on an opponent’s strategy in an upcoming match: “Everybody has a plan—until they get hit.”
Read More »January 2018
Genotype-guided dosing of warfarin: GIFT wrap-up
January 2018—In an ideal world, clinical research data would be applied with immediate and beneficial effect to clinical practice, especially when the data come from a well-controlled, well-run trial that meets the gold standard of being large, randomized, and blinded.
Read More »Next-gen sequencing finds further clinical utility in oncology
January 2018—One of the plenary sessions at the 2017 meeting of the Association for Molecular Pathology—“High Impact Molecular Diagnostics for Cancer and Inherited Diseases”—was a virtual mini-course in the latest and most useful applications of next-generation sequencing to detect germline and somatic mutations in cancer. Both speakers zeroed in on the clinical utility of their innovative diagnostic techniques.
Read More »New Color Atlas of Hematology to be used ‘in the wild’
January 2018—New this month from CAP Press is the second edition of the Color Atlas of Hematology: An Illustrated Field Guide Based on Proficiency Testing. “More and better are the watchwords,” senior editor Eric F. Glassy, MD, told CAP TODAY when we asked what the reader can expect. David Blomberg, MD, and Katherine Galagan, MD, are associate editors.
Read More »From the President’s Desk: A shared vision for cancer staging
January 2018—For me, friendships formed and lessons learned have more than compensated for the effort invested over the years on CAP committees, but make no mistake: When we meet, what we’re doing is work. The professional engagement is enjoyable, but a person can get tired toward the end of a two-day meeting, not to mention homework in the evenings.
Read More »Devices, decisions: POC glucose in the critically ill
January 2018—Using point-of-care glucose meters in critically ill patients can feel like tiptoeing through a regulatory minefield. Perhaps your preferred meter hasn’t been cleared by the FDA for use in this population. Or maybe you’re not sure which assay performance requirements should be regulating the performance of your meters. Or perhaps you’re still trying to define “critically ill.”
Read More »Lab needs driving coagulation analyzer market
January 2018—Customer wish lists help to define every generation of coagulation analyzers, test menus, and related technologies. That’s evident in the recent and upcoming launches and the ongoing work of the companies whose analyzers are profiled in this issue in the 2018 coagulation analyzer product guide.
Read More »AABB seeks comments on form to streamline transfusion adverse reaction reporting
January 2018—The AABB is seeking comments by March 30 on its common transfusion reaction reporting form, the seven pages of which are presented online at www.bit.ly/AABB-reportform. The fillable PDF form is intended to be used by hospitals and blood centers to communicate information about transfusion reactions to the blood supplier, particularly when there are multiple suppliers to the hospital transfusion service.
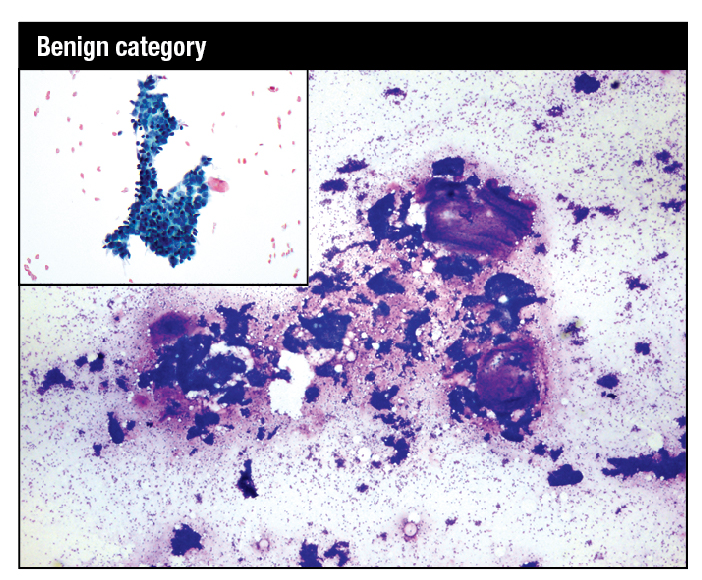
Read More »Cytopathology in Focus: Standardized reporting for breast FNAB cytology
January 2018—In countries with developed medical infrastructure, the use of breast fine-needle aspiration biopsy (FNAB) cytology has had its share of challenges over the past 20 years, among them the use of core needle biopsies. In developing countries where the use of FNAB cytology has been increasing rapidly, breast lesions are one of the most common sites sampled by FNAB. In 2016, the International Academy of Cytology Executive Council put together a “Breast Group,” which consists of cytopathologists, surgical pathologists, radiologists, surgeons, and oncologists working in breast care, with the aim of producing a comprehensive and standardized approach to breast FNAB cytology reporting.
Read More »Cytopathology in Focus: A right and a wrong way to use CAP educational kits
January 2018—The CAP Cytopathology Committee constructs educational and interlaboratory comparison kits that are distributed regularly to cytotechnologists, cytopathologists, and pathologists who want continuing education in cytopathology. The purpose of the kits is to make it possible for those who screen and diagnose cytology slides to maintain and update their skills. However, the Cytopathology Committee has been made aware that the kits have been employed for purposes other than education. We address here the potentially detrimental uses to which some laboratories are putting these educational kits and advise laboratories to use them only as they were intended.
Read More »Cytopathology in Focus: Pap proficiency testing: what’s permitted, what’s not
January 2018—Annual proficiency testing is mandated for all cytotechnologists and pathologists who sign out Pap tests. CAP staff frequently receive questions about the PT rules or hear of situations in which there was confusion about the rules, a few of which we will highlight here.
Read More »Cytopathology in Focus: HPV vaccines: the decade in review
January 2018—Diane Harper, MD, MPH, and Leslie DeMars, MD, provide an extensive review of the efficacy of available FDA-approved HPV vaccines in different age groups and describe immunogenicity findings in particular (Gynecol Oncol. 2017;146:196–204). The World Health Organization and the Centers for Disease Control and Prevention recommend a two-dose vaccine for younger children due to high rates of seroconversion and antibody titers in this age group. Girls age 15 and older should continue to get three doses.
Read More »Anatomic Pathology Abstracts, 1/18
January 2018—Analysis of desmoplastic pattern at the tumor front in colorectal cancer subtypes: Although recent findings of cancer biology research indicate that prognostic power arises from genes expressed by stromal cells rather than epithelial cells, desmoplastic reaction has not been completely examined as a prognostic marker for colorectal cancer. The authors conducted a pathologic review of 821 stage II and III patients who underwent R0 resection for colorectal cancer at four independent institutions.
Read More »Clinical Pathology Abstracts, 1/18
January 2018—Drone transport of chemistry and hematology samples over long distances: Interest in using unmanned aerial vehicles, or drones, to transport laboratory specimens is based on the need to move specimens from satellite facilities to a central hub for testing. Earlier studies of biological specimens transported by drones were performed in ambient or cold temperatures for a maximum flight length of 40 minutes.
Read More »Molecular Pathology Abstracts, 1/18
January 2018—Importance of interstitial genes that exist between gene fusion partners in prostate cancer: Prostate cancer is one of the most common cancers affecting men, yet understanding of the disease’s development and progression is limited. One of the most effective ways to stratify treatment and outcomes is based on pathology review of prostate biopsies, though the application of molecular testing to these samples is increasing.
Read More »Newsbytes, 1/18
January 2018—Why pathologists shouldn’t ‘pass the baton’ with IT: It may be tempting to stay in your comfort zone and leave the technology decisions to the information technology experts. But pathologists who abdicate oversight of IT projects within their departments are setting up those projects for failure, says John H. Sinard, MD, PhD, professor of pathology and medical director of pathology informatics at Yale University School of Medicine.
Read More »Q&A column, 1/18
January 2018—We are in the process of validating the Stago STA Compact Max and Stago STA R Max with cap piercing. The company is stating that the open and closed modes follow the same testing pathway and therefore validation between modes is not necessary. Is this correct? Is PHI (phosphohexose isomerase), also known as GPI (glucose phosphate isomerase), mainly responsible for metastasis and circulating tumor cells?
Read More »Put It on the Board, 1/18
January 2018—Some safety issues more gray than black and white: Most laboratory safety rules are free from ambiguity. Anyone who handles specimens must wear personal protective equipment, for example. Some issues are less clearly defined, though, and require a deeper dive into the guidelines. That is where Dan Scungio, MT (ASCP), SLS, CQA(ASQ), comes in.
Read More » CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management