April 2024—Roche announced FDA approval of the Cobas Malaria test, a qualitative in vitro nucleic acid screening test for the direct detection of Plasmodium RNA and DNA in whole blood samples from individual human donors. The test can be performed with other routine blood donor screening tests and is designed for use on the Cobas 6800/8800 systems in the United States.
MARKETPLACE
FDA clears Diasorin Liaison Plex system
April 2024—Diasorin announced FDA 510(k) clearance for its Liaison Plex platform and Flex respiratory assay. The fully automated, sample-to-answer system uses room-temperature–stable consumables and has a hands-on time of two minutes per sample. Results are available in less than two hours. Diasorin says Flex testing allows users to generate and pay for a subset of specific results based on a patient’s clinical picture.
Read More »Siemens offers anti-müllerian hormone assay
April 2024—Siemens Healthineers now offers the Anti-Müllerian Hormone assay to evaluate ovarian reserve. The assay runs on the Atellica IM analyzer and Advia Centaur immunoassay systems from serum and plasma samples. Results are available in about 28 minutes.
FDA clears Sebia free light chain assays
April 2024—Sebia announced it has received FDA 510(k) clearance for its free light chain kappa and lambda assays, intended to aid in diagnosing and monitoring patients who have multiple myeloma and immunoglobulin light chain amyloidosis. The assays quantify kappa or lambda free light chains in human serum using an enzyme-linked immunosorbent assay procedure. The kits are for in vitro diagnostic use only.
Read More »StatLab acquires Poly Scientific R&D
April 2024—StatLab Medical Products announced the acquisition of Poly Scientific R&D, a manufacturer and supplier of pathology stains, reagents, and paraffin for anatomic pathology laboratories. The acquisition includes Histology Control Systems, a division of Poly Scientific that manufactures and supplies tissue control slides.
Bio-Rad launches QC online learning center
April 2024—Bio-Rad Laboratories has launched its online Quality Controls Learning Center, a free resource designed to provide laboratories with the educational resources needed to maintain quality control patient test results. The site provides a library of articles and best practices on quality control process and design, laboratory management, quality control data management, quality control troubleshooting and solutions, and the benefits of independent quality control.
Read More »Sekisui gets EUA for Osom flu, SARS-CoV-2 test
April 2024—Sekisui Diagnostics has received emergency use authorization for its Osom Flu SARS-CoV-2 Combo test for use in professional and home testing settings. The lateral flow immunochromatographic assay is intended for the qualitative detection and differentiation of influenza A and B nucleoprotein antigens and SARS-CoV-2 nucleocapsid antigen. The test is for in vitro diagnostic use.
SpotFire R/ST panel gets 510(k) clearance, CLIA waiver
April 2024—BioMérieux has received FDA 510(k) clearance and CLIA waiver approval for the BioFire SpotFire Respiratory/Sore Throat (R/ST) panel. The multiplex PCR test is for the detection and identification of nucleic acids from up to 15 of the most common bacteria, viruses, and viral subtypes responsible for respiratory or sore throat infections. Samples can be taken from a nasopharyngeal swab when a respiratory tract infection is suspected or from a throat swab in case of a pharyngitis syndrome. Results are available in about 15 minutes on the BioFire SpotFire system.
Verichem releases microprotein reference materials
April 2024—Verichem Laboratories offers a multilevel set of clinical reference materials intended for the calibration verification of clinical systems testing for total protein and albumin in urine and cerebrospinal fluid samples. The liquid-stable and ready-to-use materials are suitable for use with turbidimetric and colorimetric test methods and incorporate human protein components.
Bayer, Thermo Fisher to develop NGS CDx assays
April 2024—Bayer and Thermo Fisher Scientific announced that they will develop next-generation sequencing–based companion diagnostic assays to help identify patients who may benefit from Bayer’s precision cancer therapies. The assays will be developed using Thermo Fisher’s Oncomine Dx Express test on the Ion Torrent Genexus Dx system. The Genexus Dx instrument and Oncomine Dx Express test are CE-IVD marked and only available in countries that accept the CE mark.
Read More »De novo classification granted for ADAMTS13 activity test
April 2024—Technoclone and its distributor in the United States, DiaPharma Group, announced that the FDA has granted de novo classification for the Technozym ADAMTS13 Activity ELISA. The assay is intended for the qualitative determination of ADAMTS13 activity in platelet poor human citrated plasma and indicated for use in conjunction with other clinical and laboratory findings as an aid in diagnosing thrombotic thrombocytopenic purpura in adult and pediatric patients being evaluated for thrombotic microangiopathy.
Bio-Rad gets My Green Lab certification
April 2024—Bio-Rad Laboratories announced it received My Green Lab certification for its R&D facilities in Irvine, Calif. It has been certified at My Green Lab’s highest rating, green, which reflects the percentage of possible green lab best practices that have been adopted and the extent to which they have been adopted by the lab.
Read More »Recordati Rare Diseases introduces educational website
April 2024—Recordati Rare Diseases has introduced its new educational website, thinkimcd.us. The platform is tailored for pathologists, offering comprehensive support for the diagnostic process of idiopathic multicentric Castleman disease (iMCD). The site provides an overview of iMCD diagnostic criteria, practical steps for assessing lymph node biopsies, and links to additional resources for understanding CD/iMCD. Designed to enhance professional awareness and comprehension, thinkimcd.us aims to be a valuable tool for the medical community.
Read More »Verichem reference materials for creatinine assays
March 2024—The Matrix Plus chemistry reference kit and standalone, ultra-high Matrix Plus level F for the calibration verification testing of creatinine assays by enzymatic and colorimetric methods are available from Verichem Laboratories. The ready-to-use reference materials offer constant protein content and pH across all concentration levels. The five-level Matrix Plus kit and level F feature a creatinine range from 0.2 mg/dL to 30.0 mg/dL and are intended to be treated as patient specimens. Shelf life and open-vial stability are 21 months from the manufacturing date.
Read More »Bio-Rad publishes third workbook in QC series
March 2024—Bio-Rad Laboratories has published the third workbook in a P.A.C.E.-approved series designed to provide lab professionals with the training and education needed to maintain reliable test results. “Quality Control Process Optimization with Cost of Quality Application” focuses on a practical approach of cost of quality to the laboratory workflow process of quality control. After using the workbook, laboratorians will be able to construct a basis worksheet to calculate the failure costs of a selected failed laboratory process.
Agilent, Incyte collaborate to develop CDx
March 2024—Agilent Technologies announced an agreement with Incyte to develop companion diagnostics in hematology and oncology. The agreement enables Agilent to continue to expand its companion diagnostics portfolio with novel biomarkers and Incyte to leverage Agilent’s expertise in IVD assay development, global regulatory approvals, and commercialization to support clinical trials as well as the potential registration and commercialization of companion diagnostics in the United States and Europe.
Innovative Lab Solutions welcomes VP of solutions
March 2024—Innovative Lab Solutions announced the appointment of Bridget Ketron as its vice president of solutions. With a focus on enhancing efficiency, boosting cash collections, and ensuring compliance, Ketron will play a key role in driving the company’s growth and strategic partnerships.
FDA clears Avails Medical AST system
March 2024—Avails Medical announced it has received FDA 510(k) clearance for its Equant system, an automated lab inoculation preparation system that consists of a single module instrument that holds a cuvette with an electrical sensor. It is intended for use with positive blood cultures samples for direct antimicrobial susceptibility testing without the traditional overnight subculture. The system works by preparing samples for direct susceptibility testing by manual agar disk diffusion.
Randox releases bladder cancer array
March 2024—Randox has released its female bladder cancer array, designed to help clinicians stratify patients presenting with hematuria. The array detects interleukin-12p70, interleukin-13, midkine, and clusterin from a single urine sample to determine an individual’s biomarker risk score. A person’s risk for developing bladder cancer is determined by combining the biomarker risk score with clinical risk scores generated using the company’s algorithms.
Read More »Bruker to acquire EliTechGroup
March 2024—Bruker Corp. has entered into a definitive share purchase agreement to acquire EliTechGroup for €870 million (about $940 million) in cash, excluding EliTech’s clinical chemistry business. Bruker expects to close the transaction in the second quarter of this year.
Read More »Roche to acquire LumiraDx’s POC technology
March 2024—Roche has entered into a definitive agreement to acquire select parts of the LumiraDx group related to LumiraDx’s point-of-care technology. The transaction is expected to close by the middle of this year, after which the acquired entities will be fully integrated into Roche Diagnostics.
Read More »FDA clears EDTA cartridges for SeptiCyte Rapid
March 2024—Immunexpress announced FDA clearance of EDTA blood compatible cartridges for use with the SeptiCyte Rapid test. SeptiCyte Rapid is a sample-to-answer, cartridge-based, host response molecular test for sepsis using reverse transcription polymerase chain reaction to quantify the relative expression levels of host response genes isolated from whole blood. It is intended for in vitro diagnostic use and runs on the Biocartis Idylla platform.
Read More »Binx, Cardinal Health enter into distribution agreement
March 2024—Binx Health announced it has entered into a national distribution agreement with Cardinal Health to expand access to care with its CLIA-waived, FDA-cleared Binx IO. The molecular point-of-care platform is used to detect chlamydia and gonorrhea in male and female patient samples and provides results in about 30 minutes.
Read More »Cepheid gets CLIA waiver for Xpert Xpress MVP
March 2024—Cepheid announced it has received FDA clearance with a CLIA waiver for the Xpert Xpress MVP, a multiplex vaginal panel that can now be performed in near-patient settings. The test is intended to aid in the diagnosis of bacterial vaginosis, vulvovaginal candidiasis, and trichomoniasis from a single specimen. It runs on Cepheid’s GeneXpert Xpress instruments and provides results within an hour.
Read More »OGT expands NGS operations in the U.K.
March 2024—OGT announced the opening of state-of-the-art facilities in Oxford Technology Park in the U.K. The facilities will drive development of OGT’s SureSeq next-generation sequencing product portfolio, as well as deliver expert-led training and opportunities for collaboration. Customers can receive face-to-face support with OGT’s field application specialists at the new facility and in-depth demonstrations of the company’s NGS and FISH products.
Read More »Copan gets third clearance for Colibrí system
March 2024—Copan Diagnostics announced the third FDA 510(k) clearance for its Colibrí automated ID/AST preparation instrument. The in vitro diagnostic specimen preparation system has been cleared to prepare MALDI-TOF targets for qualitative identification and microbial suspension for the Beckman Coulter MicroScan WalkAway antimicrobial susceptibility testing system for qualitative testing of isolated colonies of Gram-negative and Gram-positive bacterial species grown on solid culture media.
Read More »Qiagen Digital Insights launches NGS analysis for somatic cancer
March 2024—Qiagen Digital Insights announced its enhanced CLC Genomics Workbench Premium with LightSpeed technology now supports next-generation sequencing for somatic cancer secondary analysis. The software accelerator converts raw sequencing data in FASTQ files to interpretable lists of genetic variants in variant call format files. Qiagen says LightSpeed can analyze a 275-gene comprehensive cancer panel at 3377× coverage in six minutes for less than $0.72 per test within standard cloud environments, or laboratories can run the Qiagen CLC LightSpeed technology on existing local hardware with similar performance.
Read More »BD Biosciences releases RealBlue reagents
March 2024—BD Biosciences has released its BD Horizon RealBlue 744 and 705 laser-specific reagents, designed to reduce spillover and well suited for low- to medium-expression surface and intracellular markers. RB 744 can be used on five-laser BD FacSymphony analyzers and spectral flow cytometers. RealBlue 705 is an alternative to BD PerCP-Cy5.5 and BD Brilliant Blue 700 reagents and is compatible with conventional and spectral flow cytometry.
Read More »StatLab launches PiSmart slide printer
February 2024—StatLab announced the availability of its PiSmart thermal transfer slide printer for the automated printing of histology slides with patient information in three to five seconds. The slide printer connects with all on-market LIS systems or can be used as a standalone printer. The printer incorporates two hoppers for preloading different slide types. The single-slide manual load option allows users to bypass the hoppers when needed. PathSmart tracking software is included.
New chapter in digital pathology
February 2024—Advancements in whole slide imaging, integration of AI, and global collaboration networks are three areas the new president of the Digital Pathology Association highlighted in his first communication with DPA members this year.
Verichem bilirubin, urine chemistry reference materials
February 2024—Verichem Laboratories now offers its Tru-Zero Bilirubin Standard as part of the company’s line of liquid-stable and protein-based total and direct bilirubin clinical reference materials. The standard is intended to be treated as a patient specimen and features universal instrument compatibility. The concentration level for the total and direct bilirubin assays is 0.0 mg/dL. The product has an open-vial stability claim of five days and a shelf life of 14 months when stored at 2° to 8°C.
Read More »Machaon offers updated aHUS genetic panel
February 2024—Machaon Diagnostics has updated its genetic panel for detecting atypical hemolytic uremic syndrome. The aHUS genetic panel 3.0 now includes two more genes, for a total of 22, associated with thrombotic microangiopathies, including aHUS, thrombotic thrombocytopenic purpura, C3 glomerulopathy, congenital B12 deficiency, and others. Turnaround time is one week.
FormaPath, UB CAT announce clinical R&D collaboration
February 2024—FormaPath and the University at Buffalo Center for Advanced Technology in Big Data and Health Sciences (UB CAT) announced their collaboration to further test and develop FormaPath’s AdiPress, an automated lymph node dissection device.
CLSI introduces country-based pricing model
February 2024—The Clinical and Laboratory Standards Institute has implemented new country-based pricing for countries that meet economic criteria set forth by the World Bank. Under this model, low- and lower-middle income countries receive a 90 percent discount off list prices and upper-middle income countries receive a 50 percent discount. The CLSI says that the pricing structure enables laboratories and clinicians around the world, regardless of resources, to more feasibly access its library of standards documents, training and support materials, and membership.
Read More »Horiba launches next-gen HELO hematology solution
February 2024—Horiba Medical has launched the CE-IVDR–approved HELO 2.0 high-throughput automated hematology platform. The platform is composed of the Yumizen H1500 and H2500 high-throughput hematology analyzers, Yumizen T6000 automated conveyor, Yumizen P8000 middleware and expert validation station, and Yumizen SPS automatic slide maker. In addition, CellaVision digital cell morphology systems can be integrated with the P8000 middleware.
Thermo Fisher, Aesku sign distribution agreement
February 2024—Thermo Fisher Scientific announced an exclusive distribution agreement with Aesku Group to market, sell, and support Aesku’s portfolio of FDA-cleared immunofluorescence products, automated instruments, and software in the United States. The agreement expands Thermo Fisher’s immunology product portfolio of Elia autoimmune diagnostics, ImmunoCap allergy diagnostics, and Phadia systems to include complementary reagents and systems from Aesku.
BioMérieux acquires software company Lumed
Feburary 2024—BioMérieux has acquired the entire share capital of software company Lumed, increasing its stake from 16 to 100 percent. The acquisition of 84 percent of the capital represents an investment of about $9.75 million.
Read More »Sekisui, Aptitude partner to provide POC COVID test
February 2024—Sekisui Diagnostics formalized an exclusive distribution agreement with Aptitude Medical Systems to sell the Aptitude Metrix COVID-19 test in the United States. The EUA-approved, single-use molecular in vitro diagnostic test is for the qualitative detection of nucleic acid from SARS-CoV-2 using anterior nasal swab and saliva samples.
TechLab test detects C. difficile infection
February 2024—TechLab’s C. Diff Quik Chek Complete test is a rapid membrane enzyme immunoassay for the simultaneous detection of Clostridium difficile glutamate dehydrogenase antigen and toxins A and B in a single reaction well. Results are available in less than 30 minutes.
Read More »Fujirebio introduces pTau 217 plasma assay
February 2024—Fujirebio announced the availability of its Lumipulse G pTau 217 plasma assay for the fully automated Lumipulse G immunoassay systems. The chemiluminescent enzyme immunoassay provides a quantitative measurement of pTau 217 in human K2EDTA plasma within 35 minutes. The assay is for research use only.
Read More »Roche launches next-generation qPCR system
February 2024—Roche launched the LightCycler Pro system, designed and labeled for research and in vitro diagnostic workflows. Enhancements of this LightCycler system include a new vapor chamber for temperature uniformity across the block, new and improved software algorithms, and updated software and user interface. Users can develop their own tests or choose from a portfolio of more than 200 LightMix modular research assays and more than 60 LightMix CE-IVD assays from Roche subsidiary TIB Molbiol.
Read More »Qiagen STI assay cleared for use on NeuMoDx systems
February 2024—Qiagen announced FDA clearance of the NeuMoDx CT/NG assay 2.0 for its NeuMoDx 96 and 288 integrated PCR-based clinical molecular testing systems in the United States. The assay is designed for direct detection of asymptomatic and symptomatic bacterial infections for Chlamydia trachomatis and Neisseria gonorrhoeae. Results are delivered in about an hour.
Read More »Flagship, Offspring partner to provide assay development services
February 2024—Flagship Biosciences and Offspring Biosciences announced a strategic partnership to provide a comprehensive suite of preclinical and clinical services. The partnership allows the companies to leverage their combined expertise in preclinical and clinical trial assays to provide their drug development partners an end-to-end solution within North America and Europe.
Read More »FormaPath launches AdiPress for lymph node dissection
January 2024—FormaPath announced the commercial launch of AdiPress, an automated tool to standardize lymph node dissection.
FDA clears MeMed BV test for use on whole blood
January 2024—MeMed announced that the FDA has granted 510(k) clearance for the company’s MeMed BV test for use on whole blood samples on the MeMed Key analyzer.
Genomadix Cube cleared for CYP2C19 testing
January 2024—The Genomadix Cube CYP2C19 system is a PCR-based test that provides genotyping results of the *2, *3, and *17 alleles of the CYP2C19 gene from a buccal swab sample in one hour.
OGT, Intelliseq launch NGS reporting for myeloid malignancy panels
January 2024—OGT and Intelliseq released GeneSpect Somatic Reporter for use with OGT’s SureSeq portfolio of next-generation sequencing panels, including SureSeq myeloid panels.
Smart In Media launches grossing camera and software
January 2024—Smart In Media announced the release of its PathoZoom LiveView Macro camera and PathoZoom software.
FDA approves treatment for rare blood clotting disorder
January 2024—The Food and Drug Administration approved Adzynma (Takeda Pharmaceuticals), the first recombinant protein product indicated for prophylactic or on-demand enzyme replacement therapy in adult and pediatric patients with congenital thrombotic thrombocytopenic purpura (cTTP), a rare and life-threatening blood clotting disorder.
CLSI releases jointly developed 2023 breakpoint implementation toolkit
January 2024—The Clinical and Laboratory Standards Institute, Association of Public Health Laboratories, American Society for Microbiology, the CAP, and Centers for Disease Control and Prevention have jointly developed a breakpoint implementation toolkit (BIT) to assist clinical laboratories in updating minimal inhibitory concentration breakpoints. The toolkit includes links to other resources that explain the rationale behind breakpoint updates, regulatory requirements for updating breakpoints, and detailed instructions for performing an AST breakpoint validation or verification. Manufacturers of AST systems can provide guidance on breakpoints used and clearance status with their systems.
Read More »FDA clears BD MiniDraw capillary blood collection system
January 2024—BD announced it has received FDA 510(k) clearances for its BD MiniDraw, a novel blood collection device that obtains blood samples from a fingerstick. The clearances include low-volume blood collection for a lipid panel, selected chemistry tests, and hemoglobin and hematocrit testing.
Read More »Biocare Medical unveils IntelliPath+ staining instrument
January 2024—Biocare Medical has introduced the IntelliPath+ advanced staining instrument, intended for in vitro diagnostic use and redesigned with input from users and the latest technological advancements. It is an open system that accommodates continuous random access with immediate stat capabilities and can process 50 slides per run.
Read More »Tosoh G8 now connects to Sysmex XN-9000, XN-9100 systems
January 2024—Tosoh Bioscience has entered into an agreement with Sysmex America in which its HLC-723G8 automated glycohemoglobin analyzers can be connected to Sysmex XN-9000 and XN-9100 automated hematology systems in the United States, Latin America, and Canada. With this agreement, Tosoh says it aims to expand its HbA1c offering to the high-volume testing market and further automate hemoglobin A1c testing.
Read More »Roche launches Elecsys HBeAg quant in CE markets
January 2024—Roche launched the Elecsys HBeAg quant, an immunoassay for the in vitro qualitative and quantitative determination of hepatitis B e antigen in human serum and plasma. In conjunction with other laboratory results and clinical information, HBeAg quantification may be used as an aid in the diagnosis and monitoring of patients with hepatitis B viral infection. It is for use on Cobas e analyzers in countries that accept the CE mark.
Read More »FDA clears QuidelOrtho Savanna PCR platform and HSV 1+2/VZV assay
January 2024—QuidelOrtho has received FDA 510(k) clearance for its Savanna PCR platform and Savanna HSV 1+2/VZV in vitro diagnostic test for the detection and differentiation of herpes simplex virus types 1 and 2 and varicella zoster virus nucleic acids isolated and purified from swabs obtained from cutaneous or mucocutaneous lesion specimens from symptomatic patients.
Read More »Ad Astra QScout hematology analyzer gets 510(k) clearance
January 2024—Ad Astra Diagnostics has received 510(k) clearance from the FDA for its QScout rapid-result hematology system. The analyzer provides point-of-care white blood cell counts and a neutrophil-to-lymphocyte ratio and differentiates the number and percent of five types of mature WBCs as well as immature granulocytes. To run the test, whole blood is added to a QScout rapid leukocyte differential test, which contains a dried reagent that stains cells. When the test is inserted in the QScout analyzer, an optical system takes images and an algorithm identifies the cells in real time. Results are displayed in about two minutes.
Read More »LGC launches unmethylated, methylated ctDNA mutation mixes
December 2023—LGC Clinical Diagnostics has launched Seraseq unmethylated and methylated ctDNA mutation mixes for assay development, validation, and routine performance monitoring. Key features of the products include global methylation of CpG sites to support all CpG methylation assays and enzymatically fragmented ctDNA for low background noise and physiologically relevant fragment sizes. Methylation status is quantified using digital PCR assays and validated by targeted next-generation sequencing panels. The products are manufactured in GMP-compliant, ISO 13485-certified facilities.
Read More »Mayo, Progentec to market biomarker tests for autoimmune diseases
December 2023—Mayo Clinic Laboratories announced a strategic collaboration to bring Progentec’s proprietary biomarker blood tests for the management of autoimmune diseases to market. The collaboration aims to increase accessibility for providers and patients across the United States and select global markets.
Read More »OGT, Intelliseq partner to advance clinical insight from NGS data
December 2023—OGT announced a partnership with Intelliseq, a genome informatics company and provider of next-generation sequencing analysis solutions. The collaboration combines OGT’s SureSeq NGS portfolio with Intelliseq’s iFlow engine, providing a sample-to-report workflow. Users will be able to easily interpret variant calls made by OGT’s NGS software via the Intelliseq iFlow engine.
Read More »Cardiac Advance now compatible with Beckman instruments
December 2023—Bio-Rad Laboratories has announced the expanded compatibility of its cardiac control, Cardiac Advance, to include Beckman Coulter instruments. The next-generation control is optimized with troponin I and troponin T targets near the limit of instrument detection and contains 10 of the most tested cardiac analytes, including troponin, CK-MB, BNP/proBNP, and myoglobin. Cardiac Advance is available in multiple formats, including the Liquicheck and InteliQ human serum-based controls.
Read More »TriVerity gets FDA breakthrough device designation
December 2023—Inflammatix announced that the FDA has granted breakthrough device designation to its TriVerity acute infection and sepsis test system. The system, which is currently under development, includes the TriVerity test and Myrna instrument.
Read More »BD launches next-gen blood draw technology
December 2023—BD launched its FDA 510(k)–cleared Pivo Pro needle-free blood collection device, which is compatible with integrated and long peripheral IV catheters, including the Nexiva closed IV catheter system with NearPort IV Access. This expands current Pivo compatibility with short peripheral IV catheters.
Read More »FDA authorizes Invitae panel for common hereditary cancers
December 2023—The FDA has granted de novo marketing authorization for the Invitae Common Hereditary Cancers panel, an in vitro diagnostic test that can help detect hundreds of genetic variants associated with an elevated risk of developing certain cancers. The test can also help identify potentially cancer-associated hereditary variants in people who have an already-diagnosed cancer.
Read More »Quest, Scipher partner to expand testing for patients with RA
December 2023—Quest Diagnostics and Scipher Medicine announced a collaboration designed to expand patient access to diagnostic services for rheumatoid arthritis. Under a multiyear collaboration, Quest will provide RNA extraction and next-generation sequencing services for Scipher’s PrismRA test, a blood-based molecular signature response classifier aimed at predicting a patient’s response to TNF inhibitor therapy. Quest will enable specimen collection at about 7,300 patient access points and more than 2,100 patient service center locations, as well as courier services that transport patient specimens between Quest and Scipher laboratories and provider sites.
Read More »Revvity study shows value of NGS in newborn screening
December 2023—A recent study by Revvity Omics has shown the clinical value of proactive, sequencing-based screening in apparently healthy newborns (Balciuniene J, et al. JAMA Netw Open. 2023;6[7]:e2326445). The objective of the study was to assess the clinical utility of genome sequencing versus a gene panel for a curated set of medically actionable pediatric-onset conditions in a large cohort of apparently healthy newborns and children tested at a clinical laboratory.
Read More »Veracyte announces IVD agreement with Illumina
December 2023—Veracyte has entered into a multiyear agreement with Illumina to develop and offer some of its high-performing molecular tests as decentralized in vitro diagnostic tests on Illumina’s NextSeq 550D× next-generation sequencing instrument. The agreement is part of Veracyte’s expanded, multiplatform IVD approach, which will also include qPCR.
Read More »FDA approves therapy for myelodysplastic syndromes
December 2023—The FDA has approved ivosidenib (Tibsovo, Servier Pharmaceuticals) for the treatment of adult patients with relapsed or refractory myelodysplastic syndromes with an isocitrate dehydrogenase-1 mutation as detected by an FDA-approved test. The agency also approved the Abbott RealTime IDH1 assay as a companion diagnostic for the selection of R/R MDS patients with an IDH1 mutation.
Read More »FoundationOne CDx approved for capivasertib plus fulvestrant
December 2023—Foundation Medicine has received FDA approval for its FoundationOne CDx to be used as a companion diagnostic for capivasertib (Truqap, AstraZeneca) in combination with fulvestrant, which has been contemporaneously approved for the treatment of adult patients with HR-positive, HER2-negative locally advanced or metastatic breast cancer with one or more PIK3CA/AKT1/PTEN-alterations following progression on at least one endocrine-based regimen in the metastatic setting or recurrence on or within 12 months of completing adjuvant therapy.
Read More »Verichem reference materials for serum and urine testing
November 2023—Verichem Laboratories announced the availability of its Urine Uric Acid Standard kit, Matrix Plus Cholesterol Reference kit, and a standalone, ultra-high Matrix Plus Cholesterol Reference level F for the calibration verification testing of cholesterol and urine and serum uric acid assays. The Urine Uric Acid Standard kit is a five-level kit with uric acid concentrations ranging from 1 to 101 mg/dL. The materials feature universal testing compatibility and are composed of a biosynthetic matrix with urine-like activity. Storage temperature is −15° to −25°C and Verichem says the product can tolerate up to 10 freeze-thaw cycles with no effect on accuracy or performance. Shelf life is 19 months.
Read More »FDA clears Thermo Fisher chromogranin A assay
November 2023—Thermo Fisher Scientific announced FDA clearance of its Thermo Scientific Brahms CgA II Kryptor, an automated immunofluorescent assay for the quantitative determination of the concentration of chromogranin A in human serum. The biomarker is to be used in conjunction with other clinical methods as an aid in monitoring disease progression during the course of disease and treatment in patients with gastroenteropancreatic neuroendocrine tumors (GEP-NET), grades 1 and 2.
Read More »LGC acquires Kova International
November 2023—LGC Clinical Diagnostics announced the acquisition of Kova International, a developer and manufacturer of in vitro urinalysis and toxicology quality control products for clinical laboratories.
Read More »BioMérieux announces CE mark for Vidas TBI test
November 2023—BioMérieux announced it has received the CE mark for its Vidas TBI (GFAP, UCH-L1), a test to support the assessment of patients who have mild traumatic brain injury. The blood test measures the concentration of glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase-L1 (UCH-L1), two brain biomarkers that are released into the bloodstream during the first hour following a brain injury. It aims to fill a gap in patient screening methods by ruling out acute intracranial lesions and helping to determine if a CT scan is necessary.
Read More »Bio SB launches TintoStainer Plus automatic IHC stainer
November 2023—Bio SB has launched a fully automated immunohistochemistry platform for deparaffinization and antigen retrieval and staining. Applications include immunohistochemistry, Mohs IHC, immunocytochemistry, and immunofluorescence of formalin-fixed, paraffin-embedded tissue, frozen tissue, and cell specimens.
Read More »FDA clears Nova Prime Plus for microcapillary sample mode
November 2023—Nova Biomedical announced that the FDA has granted 510(k) clearance for a microcapillary sample mode on the company’s Stat Profile Prime Plus critical care blood gas analyzer.
Read More »FDA approves Qiagen CDx for GIST
November 2023—Qiagen announced FDA approval of its Therascreen PDGFRA RGQ PCR kit, a companion diagnostic intended for use to aid clinicians in identifying patients with gastrointestinal stromal tumors (GIST) who may be eligible for treatment with avapritinib (Ayvakit, Blueprint Medicines). Ayvakit is approved in the United States for the treatment of adults with unresectable or metastatic GIST harboring a platelet-derived growth factor receptor alpha (PDGFRA) exon 18 mutation, including PDGFRA D842V mutations. Qiagen says its kit is the first PDGFRA assay to receive FDA approval as a companion diagnostic.
Read More »FloBio bleeding risk Dx gets FDA breakthrough device designation
November 2023—FloBio announced that the FDA has granted breakthrough device designation for its rapid bleeding risk diagnostic test. The point-of-care test is designed for in vitro diagnostic use to determine blood clotting status and whether a patient is on a direct oral anticoagulant.
Read More »FDA clears Aptiva Connective Tissue Disease Essential reagent
November 2023—Werfen announced FDA 510(k) clearance of its Aptiva Connective Tissue Disease Essential reagent, to aid in diagnosing connective tissue disease. The Aptiva CTD Essential complements Werfen’s previously cleared Aptiva Celiac Disease reagent.
Read More »HemoSonics Quantra awarded Innovative Technology contract
November 2023—HemoSonics announced that its Quantra hemostasis system received an Innovative Technology contract award from Vizient. The Quantra system consists of the Quantra hemostasis analyzer with QPlus and QStat cartridges and provides comprehensive whole-blood coagulation analysis at the point of care in less than 15 minutes.
Read More »Feasibililty study results evaluating Mainz advanced adenoma biomarkers
October 2023—Mainz Biomed announced results of an independent feasibility study conducted in collaboration with members of the National Cancer Institute’s Early Detection Research Network to evaluate the company’s portfolio of five mRNA biomarkers, which it acquired from Université de Sherbrooke, Quebec.
Read More »MMQCI releases BCR-ABL IS linearity panel
October 2023—Now available from Maine Molecular Quality Controls is the Xpert BCR-ABL IS p210 Linearity Panel C207. It’s intended for use as an assayed external quality control to monitor the performance of the in vitro quantitative detection of BCR-ABL1 translocation mRNA e14a2/b3a2 transcripts and the ABL1 endogenous control mRNA transcript. It is designed to be used with the Xpert BCR-ABL Ultra assay on Cepheid GeneXpert instruments. Each kit comprises 12 bottles with two bottles of each international scale percent (%IS) value in 4 mL of synthetic BCR-ABL1 RNA transcript and synthetic ABL1 control gene RNA transcript suspended in a stabilizing matrix with a noninfectious solution of buffers and preservatives.
Read More »FDA clears PixCell HemoScreen for direct capillary sampling
October 2023—PixCell Medical announced that the FDA has granted 510(k) clearance for direct capillary sampling with the HemoScreen 5-part differential CBC analyzer. The clearance enables collection of a sample directly from a patient’s finger without an intermediate tube. HemoScreen is also FDA cleared for point-of-care use with venous and capillary blood.
Read More »FDA approves Bosulif for pediatric patients with CML
October 2023—The FDA approved bosutinib (Bosulif, Pfizer) for pediatric patients who are one year old and older with chronic phase Ph+ chronic myelogenous leukemia that is newly diagnosed or resistant or intolerant to prior therapy. Efficacy was evaluated in the BCHILD trial (NCT04258943), a multicenter, nonrandomized, open-label trial conducted to identify a recommended bosutinib dose in pediatric patients with newly diagnosed chronic phase Ph+ CML and resistant or intolerant Ph+ CML; estimate the safety, tolerability, and efficacy; and evaluate bosutinib pharmacokinetics in this patient population.
Read More »Pillar Biosciences launches OncoReveal Core LBx NGS kit
October 2023—Pillar Biosciences announced the global launch of OncoReveal Core LBx, a research use only, liquid biopsy–based next-generation sequencing kit for pan-cancer tumor profiling. The panel interrogates 104 clinically relevant genes in one multiplex reaction, analyzes cfDNA present in plasma for genetic alterations in cancer, including assessment of microsatellite instability, and can batch more than 20 clinical samples on a single Illumina NextSeq run. Mutation detection performance is as low as 0.1 percent.
Read More »Parasitology, infectious Dx company Eiger launches
October 2023—Eiger Diagnostics has been formed to provide high-quality parasitology and infectious disease diagnostics worldwide. Eiger’s first product releases are parasitology serology IgG and IgM ELISA assays. The company plans to add to its product portfolio this year.
Read More »Wren Labs molecular Dx predicts patient response to PRRT
October 2023—Wren Laboratories launched its PRRT (peptide receptor radionuclide therapy) Predictor Quotient, or PPQ, a companion diagnostic to its NETest, a liquid-biopsy neuroendocrine tumor diagnostic. The test classifies patients as either a responder, a patient who will experience disease stabilization and have a longer time to disease progression (usually greater than 18 months after the end of PRRT treatment), or as a nonresponder, who will have a shorter time until the disease progresses (usually less than 12 months after the start of PRRT). According to a research paper published in April (Bodei L, et al. J Nucl Med. 2023;64[9]:1329–1330), the PPQ delivers 96 percent accuracy in determining patient response to PRRT.
Read More »Primera unveils Signature EVO slide and cassette printers
October 2023—Primera Technology has launched its next-generation Signature EVO slide printer and Signature EVO cassette printer.
Read More »FDA clears Alinity h-series hematology system
October 2023—Abbott has received FDA clearance for its Alinity h-series hematology system, which includes the Alinity hq, an automated hematology analyzer, and Alinity hs, an integrated slide maker and stainer. The Alinity hq leverages multiangle polarized scatter separation (MAPPS) technology, which uses light scattering to distinguish cellular features and identify various blood cells, and processes up to 119 CBC results per hour. The system can be integrated into an existing core lab operation.
Read More »Verichem enzyme calibration verifiers for liver function testing
October 2023—Verichem Laboratories offers multilevel enzyme calibration verification materials for liver function testing as part of its liquid-stable Enzyme ER Verifier kit. The kit contains the enzymes alanine aminotransferase (ALT/SGPT), alkaline phosphatase (AP/ALP), aspartate aminotransferase (AST/SGOT), gamma-glutamyl transferase (GGT/GGTP), lactate dehydrogenase (LD/LDH), amylase, cholinesterase, creatine phosphokinase, and lipase. Its proprietary formulation is designed to include at least one concentration for each enzyme within the normal range. The verifiers are intended to be treated as patient specimens and are compatible with wet chemistry systems from Abbott, Beckman Coulter, Roche, and Siemens Healthineers. Shelf life is 18 months.
Read More »Sophia Genetics expands relationship with Gustave Roussy
October 2023—Sophia Genetics announced the expansion of its relationship with Gustave Roussy in which the cancer center will use the Sophia DDM digital analytics platform for all relevant samples, including those related to solid tumors and hematologic and hereditary cancers. Gustave Roussy, which has two campuses in France, began working with the cloud-native software company in 2017. Sophia Genetics is located in France and Boston.
Read More »Visiopharm, Boston Cell Standards to develop IHC technology
October 2023—Visiopharm and Boston Cell Standards announced they are partnering to integrate immunohistochemistry calibration standards with image analysis software for quality assurance. The joint technology solution will enable laboratories to meet key provisions of regulatory changes proposed in an editorial published in Archives of Pathology & Laboratory Medicine.
Read More »Illumina, Pillar Biosciences announce partnership
September 2023—Illumina and Pillar Biosciences announced a strategic partnership to make Pillar’s suite of oncology assays commercially available worldwide as part of Illumina’s portfolio of oncology products. The companies say the agreement will result in an unprecedented offering of complementary next-generation sequencing solutions that will enhance the efficiency, accuracy, and cost-effectiveness of oncology testing through advanced sequencing techniques.
Read More »New book in Foundations in Diagnostic Pathology series
September 2023—Differential Diagnoses in Surgical Pathology: Tumors and their Mimickers, a new volume in the Foundations in Diagnostic Pathology series, has been published by Elsevier. The book, by Fadi W. Abdul Karim, MD, MEd, and Charles Sturgis, MD, aims to help readers quickly differentiate entities that have similar, overlapping histopathologic features and guide them through the decision-making process, providing a road map to the main differential diagnostic considerations that must be addressed when formulating a diagnosis. The book includes high-quality illustrations of similar-looking but distinct entities for side-by-side comparison in a user-friendly format with at-a-glance boxes and tables throughout the text, as well as selected references for further study. An enhanced ebook version is included with purchase.
Read More »Comanche drug for preeclampsia gets fast track designation
September 2023—Comanche Biopharma announced it has received fast track designation from the FDA for its CBP-4888 as a novel small interfering ribonucleic acid (siRNA) therapy for preeclampsia. CBP-4888 is a fixed-dose combination of two chemically synthesized, lipid-conjugated siRNA duplex oligonucleotides, siRNA-2283 and siRNA-2519, targeting two soluble fms-like tyrosine kinase–1 (sFLT1) mRNA isoforms. Fast track is designed to facilitate the development and expedite the review process of drugs to treat serious conditions and fill an unmet medical need.
Read More »Stago Max Generation systems get DOD authorization to operate
September 2023—Diagnostica Stago received an authorization to operate from the Department of Defense Health Agency (DHA) for the company’s Max Generation analyzers and middleware solution. The cybersecurity clearance allows Department of Defense military treatment facilities worldwide to connect Stago Max Generation systems to the DHA network.
Read More »American Registry of Pathology Press digital library
September 2023—The American Registry of Pathology Press digital library offers a fully searchable books platform with full-text versions of the books in ARP Press’ current series of fascicles, including the fourth and fifth series of the AFIP Atlases of Tumor and Non-tumor Pathology series. The site, arppress.org, offers pathologists robust searches across the series for images and text covering clinical features, differential diagnoses, immunohistochemistry, molecular biology, treatment, and prognosis.
Read More »FDA clears Siemens Atellica CI analyzer
September 2023—Siemens Healthineers announced that its Atellica CI analyzer for immunoassay and clinical chemistry testing has received FDA clearance and is available in many countries.
Read More »Owen Mumford introduces Unistik venous blood collection devices
September 2023—Owen Mumford has introduced its Unistik venous blood collection devices. Unistik VacuFlip features a safety guard that flips over to cover the needle after it is withdrawn from the patient’s arm. An audible click signals the safety guard is in place and the needle is locked. VacuFlip is available in three needle gauges and needle lengths.
Read More »KSL Diagnostics opens transplant immunology laboratory
September 2023—KSL Diagnostics, Buffalo, NY, has opened a transplant immunology laboratory to serve the western New York region. KSL is the provider for Erie County Medical Center and Kaleida Health. ConnectLife, a community blood bank and federally designated not-for-profit organ procurement organization, uses KSL’s new lab and services Allegany, Cattaraugus, Chautauqua, Erie, Genesee, Niagara, Orleans, and Wyoming counties.
Read More »GADx reintroduces IT-Leish test for visceral leishmaniasis
September 2023—Global Access Diagnostics announced the launch of IT-Leish, a UKCA-marked rapid test used for diagnosing visceral leishmaniasis. The company acquired the manufacturing rights for IT-Leish from Bio-Rad in September 2022 to prevent the test’s withdrawal from the market and support the World Health Organization’s strategic plan to eliminate life-threatening neglected tropical diseases.
Read More »Evident Slideview DX VS-M1R digital scanner
September 2023—Evident has introduced its Slideview DX VS-M1R whole slide imaging system, for research use only.
Read More »Verichem reference materials for electrolyte testing
September 2023—Verichem Laboratories offers a line of liquid-stable, multianalyte kits for the calibration verification of electrolyte assays for use with systems employing flame photometer, direct or indirect ion-selective electrode, or standard enzymatic/colorimetric testing methods. The Electrolyte Standard kit contains the analytes chloride, ionized calcium, lithium, potassium, and sodium and has a 21-month stability claim. The ISE Standard kit contains chloride, lithium, potassium, and sodium and has an 18-month stability claim. The Carbon Dioxide Standard kit and standalone CO2 Standard level F both have 15-month stability claims. All products are free of azides, glycols, surfactants, and other potentially interfering substances.
Read More »FDA clears respiratory viral panel for BD Max
September 2023—BD announced FDA 510(k) clearance for its BD Respiratory Viral Panel for the BD Max system. It is a single molecular diagnostic combination test that uses a single nasal or nasopharyngeal swab sample to determine if a patient has SARS-CoV-2, influenza A, influenza B, or respiratory syncytial virus. Results are available in about two hours.
Read More »FDA clears Thermo Fisher assays for preeclampsia
September 2023—Thermo Fisher Scientific announced FDA clearance of its Brahms PlGF plus Kryptor and Brahms sFlt-1 Kryptor novel biomarkers for the risk assessment and clinical management of preeclampsia. The assays received FDA breakthrough designation in May.
Read More »Cole-Parmer ultra-low temp freezers
August 2023—Cole-Parmer introduced its North Sciences/Traceable TSi and TEC2 series ultra-low temperature freezers. The self-monitoring freezers are integrated with a Traceable data logger that is compatible with the TraceableLive cloud-based monitoring service, which provides audio and visual alerts via an Apple Watch, smartphone, tablet, or PC. The ULT freezers can reach temperatures down to −86°C and are ideal for the safe storage of samples and vaccines and compliant with Vaccines for Children storage requirements.
Read More »Siemens ELF test gets FDA breakthrough device designation
August 2023—The FDA has granted a breakthrough device designation for Siemens Healthineers Enhanced Liver Fibrosis test, the only blood test granted FDA marketing authorization for prognostication of disease progression in patients with advanced fibrosis due to nonalcoholic fatty liver disease.
Read More »OncoHost, BGN Technologies sign option agreement
August 2023—OncoHost has signed an option agreement with BGN Technologies, the technology transfer company of Ben-Gurion University of the Negev (Be’er Sheva, Israel), to receive a license for a novel biosensing technology called IcAR, or Immuno-checkpoint Artificial Reporter, developed by researchers from BGU. IcAR biosensing technology measures the binding functionality of PD-1 ligands, PD-L1 and PD-L2, to their receptor, PD-1. The researchers found that assessing the functionality of PD-1 ligands was an effective predictor of identifying who will positively respond to anti-PD-1 treatment.
Read More »FBI approves Qiagen ForenSeq Mainstay workflow for NDIS
August 2023—Qiagen announced that the U.S. Federal Bureau of Investigation has approved its ForenSeq Mainstay workflow, allowing accredited forensic DNA laboratories to process DNA casework samples and search resulting profiles against the U.S. National DNA Index System CODIS database. The ForenSeq Mainstay workflow is composed of the high-throughput Verogen ForenSeq Mainstay kit, MiSeq FGx next-generation sequencing system, and ForenSeq Mainstay analysis module in the Universal Analysis software.
Read More »Illumina launches DRAGEN 4.2
August 2023—Illumina launched the latest version of its DRAGEN software, version 4.2, for analyzing next-generation sequencing data. New features of the software include increased single nucleotide variant and structural variation calling accuracy powered by an enhanced machine learning model and multigenome graph, improvements in small copy number variation calling accuracy with joint SV/CNV calling, and the addition of four targeted callers for carrier screening—HBA 1/2, CYP21A2, RHD/RHCE, and LPA.
Read More »Werfen completes acquisition of Immucor
August 2023—Werfen announced it has completed the acquisition of Immucor after obtaining all necessary regulatory and antitrust approvals. The purchase price was about $2 billion.
Read More »Verichem calibration verification reference materials
August 2023—A multilevel set of liquid-stable clinical reference materials for use with ammonia and iron testing are available from Verichem Laboratories. The five-level standard kit, along with an optional, standalone ultra-high sixth level, is designed for the calibration verification of ammonia and iron assays on a range of automated clinical systems, including from Abbott Diagnostics, Beckman Coulter, Roche Diagnostics, and Siemens Healthineers. The materials are treated as patient specimens and free of glycols, surfactants, azides, and other interfering substances. Iron concentration levels of 10–1,000 μg/dL are verified using standard reference materials from the National Institute of Standards and Technology, and ammonia concentrations of 10–2000 μg/dL are verified using reagent-grade standards from the American Chemical Society. Shelf life is 24 months.
Read More »FDA approves ARUP Labs AAV5 DetectCDx
August 2023—ARUP Laboratories announced that the FDA approved AAV5 DetectCDx as a companion diagnostic to aid in the selection of adult patients eligible for treatment with Roctavian (valoctocogene roxaparvovec-rvox). Roctavian, a gene therapy developed by BioMarin Pharmaceutical, received contemporaneous FDA approval for the treatment of adults with severe hemophilia A (congenital factor VIII deficiency with FVIII activity <1 IU/dL) without antibodies to adeno-associated virus serotype 5 (AAV5) detected by an FDA-approved test.
Read More »LGC launches Seraseq 22q11 female-matched reference material
August 2023—LGC Clinical Diagnostics announced that its Trisomy 22q11 Female-Matched Reference Material is now included in its Seraseq noninvasive prenatal testing portfolio. It is a circulating cell-free DNA–like mixture of human genomic DNA (matched maternal and fetal) in a commutable matrix intended to be used as a reference material to monitor library preparation, sequencing, and detection performance. The material is processed to maintain the natural cell-free DNA size profiles of fetus and maternal DNA of approximately 170 base pairs.
Read More »Bio-Rad launches second installment in QC workbook series
August 2023—Bio-Rad Laboratories has published the second workbook in a series that offers Professional Acknowledgement for Continuing Education credits. The second installment, titled “Laboratory Quality Control Materials,” focuses on identifying proper QC materials and using and handling them appropriately to help monitor QC testing procedures that produce high-quality patient results. Lab professionals can receive a certificate after successfully completing a short exam at the end of the workbook and earn 2.0 contact hours.
Read More »Randox acquires Cellix
July 2023—Randox (Crumlin, U.K.) has acquired Cellix, a Dublin-based company that develops microfluidic tools and impedance flow cytometers for cell analysis.
Read More »General Data LaserTrack Flex cassette printer
July 2023—General Data Healthcare’s Flex is the latest addition to its LaserTrack portfolio of cassette printers.
Read More »LGC launches Accurun MS2 internal control
July 2023—LGC Clinical Diagnostics has launched the SeraCare Accurun MS2, a full-process internal control that can be used as a tool to monitor the process integrity of sample extraction and amplification of nucleic acid amplification–based assays. The product contains positive materials of intact MS2 bacteriophage, and the ready-to-use controls can be spiked into specimens that are going through DNA/RNA extraction.
Read More »Bio-Rad launches bulk urine, CSF negative controls
July 2023—Bio-Rad Laboratories launched two Exact Diagnostics products, Bulk Urine Negative and Bulk CSF Negative controls. The bulk urine negative control is intended to be validated as an independent external quality run control to monitor the absence of adenovirus, BK virus, Candida auris, Chlamydia trachomatis, cytomegalovirus, John Cunningham virus, Mycoplasma genitalium, Neisseria gonorrhoeae, Trichomonas vaginalis, and Zika virus in various molecular assays. The bulk CSF negative control is an independent external quality run control intended to monitor the absence of Anaplasma phagocytophilum, Babesia microti, Bartonella quintana, Borrelia burgdorferi, Ehrlichia chaffeensis, enterovirus, herpes simplex virus 1 and 2, and varicella-zoster virus in various molecular assays.
Read More »Siemens to distribute Scopio digital cell morphology platforms
July 2023—Siemens Healthineers has entered into an agreement with Scopio Labs to distribute the Scopio X100 and Scopio X100HT. These imaging platforms will complement Siemens hematology systems, including the Atellica Hema 570 and Hema 580 analyzers, to offer laboratories high-resolution, full-field viewing for peripheral blood specimens and artificial intelligence–based morphological analysis with remote viewing capabilities.
Read More »OGT partners with Applied Spectral Imaging
July 2023—OGT, a Sysmex Group company, announced a commercial partnership with Applied Spectral Imaging. The agreement will grant OGT rights to market ASI’s proprietary cytogenetic imaging and analysis solutions in Great Britain, establishing a strategic partnership that the companies say combines the strengths of OGT’s CytoCell FISH portfolio with ASI’s imaging and analysis capabilities.
Read More »Verichem offers free data reduction reports
July 2023—Verichem Laboratories now offers its customers free access to its Web-based and online calibration verification data reduction and test-reporting programs. Designed for use with the company’s clinical reference materials, these programs and services provide the necessary statistical data and test reports to satisfy CLIA requirements for the calibration verification of clinical assays. Users can register online with a password, log in, and then enter and submit data; there is no software to download. Reports are generated instantly in a PDF format and are ready to review, sign off, and file. Final reports can include clinical system test accuracy, linearity, precision, precision with peer comparison, and other supplemental study data. Technical support is available via phone to answer questions or troubleshoot.
Read More »BD launches FacsDuet Premium system
July 2023—BD announced the worldwide commercial launch of its BD FacsDuet Premium system. The automated instrument prepares samples for in vitro diagnostic and user-defined tests, including cocktailing, washing, and centrifuging, and then automatically transfers samples to the integrated BD FacsLyric clinical flow cytometry system, enabling a walkaway workflow solution.
Read More »Safety-Spec launches gross room trays
July 2023—Safety-Spec launched its Safety-Spec Gross Room Tray. The reusable trays are designed to ensure tissue specimens are easily accessible, properly labeled, and neatly organized. Each tray is constructed of high-quality, dense, closed-cell foam, providing a lightweight, flexible, nonbreakable, and washable tray that can accommodate up to four specimen bottles, eight cassettes (two per specimen), and specimen-related paperwork. Trays are available in packs of 30, 60, 90, and 120, with red, yellow, green, blue, purple, and white trays in each pack.
Read More »RMT introduces RmtHealthCheck
July 2023—Remote Medical Technologies introduced RmtHealthCheck, a new feature of the company’s RmtConnectPlus real-time telepathology solution. It notifies users via email of network disruptions that could impede system access. Once the issue is corrected, the system will email users that the network connection has been restored. Third-party software is not required for use.
Read More »Bruker launches TimsTOF Ultra mass spectrometer
July 2023—Bruker Corp. launched its TimsTOF Ultra mass spectrometer at the American Society for Mass Spectrometry annual conference, June 4–8, in Houston, Tex. TimsTOF Ultra can identify, with 125 pg of material, more than 5,500 protein groups in 22 minutes via a collision cross section–enabled TIMScore and TIMS DIA-NN 4D-Proteomics or Spectronaut 18 software. Its new CaptiveSpray Ultra ion source with vortex flow enhances ion formation across nanoflow liquid chromatography gradients. The platform has a parallel accumulation–serial fragmentation (PASEF) scan speed of up to 300 Hz for tandem mass spectrometry.
Read More »Danish National Genome Center selects Qiagen’s QCI Interpret
July 2023—Qiagen announced that its variant interpretation and reporting software, Qiagen Clinical Insight (QCI) Interpret, was chosen by the Danish National Genome Center to provide interpretation of oncology results generated from whole genome sequencing data. The initiative is part of a larger personalized medicine strategy that aims to provide WGS as the standard of care for relevant patient groups throughout Denmark.
Read More »FDA clears Immuno Concepts IgG Anti-nDNA test system
July 2023—Immuno Concepts announced FDA 510(k) clearance of its IgG Anti-nDNA fluorescent test system for use with the company’s Image Navigator system. The test is for in vitro diagnostic use for qualitative detection and semiquantitation of anti-nDNA antibodies of the IgG class in human serum to aid in diagnosing systemic lupus erythematosus in conjunction with other clinical and laboratory findings.
Read More »Leica launches Bond ChromoPlex II Dual Detection kit
July 2023—Leica Biosystems launched its Bond ChromoPlex II Dual Detection kit for use in immunohistochemistry and chromogenic in situ hybridization on Bond-Max and Bond-III instruments.
Read More »BioMérieux launches BioFire Fireworks software
June 2023—BioMérieux has launched BioFire Fireworks, an integrated software solution for BioFire systems that is part of the company’s Vision Suite data-management product line. Fireworks provides access to a centralized data-management Web portal, giving users insights and analytics into BioFire system performance, utilization, pathogen surveillance, and workflow management. The software was designed, developed, and implemented following industry standards and regulations and gives customers complete control over their data.
Read More »Worthington Biochemical product catalog
June 2023—Worthington Biochemical announced that the 23rd edition of its enzyme and biochemical catalog is now available in print and digital formats. The catalog covers a wide array of products for life science applications including immunology, cell biology, molecular biology, research, biochemistry, and bioprocessing. It features Worthington Animal Free enzymes for preclinical, bioprocessing, and biopharma applications produced under ISO 9001–certified and GMP guidelines and contains a number of references, including technical enzyme selection tools, product specifications and application tables, related products at a glance, and product listings by application.
Read More »ZeptoMetrix introduces Protrol QC material
June 2023—ZeptoMetrix introduced Protrol, a line of products designed to evaluate and monitor the performance of antigen-based assays, including lateral flow immunoassays for infectious diseases. Each lot is supplied with the protein concentration and TCID50 value.
Read More »FDA clears immunoassays to assess risk of preeclampsia
June 2023—Thermo Fisher Scientific announced the FDA has cleared the Thermo Scientific Brahms PlGF Plus Kryptor and Brahms sFlt-1 Kryptor novel biomarkers, both of which received breakthrough designation from the FDA.
Read More »SeraCare releases solid tumor CNV reference material
June 2023—LGC SeraCare has developed a comprehensive solid tumor copy number variation reference material in a formalin-fixed, paraffin-embedded format. The Seraseq FFPE Solid Tumor CNV RM includes 12 clinically relevant CNVs associated with solid tumors. Amplifications of these target genes are quantitated by digital PCR, blended in a well-characterized genomic background, and validated with next-generation sequencing. One 10-μm FFPE curl is provided per vial, and the product is designed to give a minimum yield of 100 ng per curl.
Read More »Siemens launches Atellica Hema analyzers
June 2023—Siemens Healthineers launched two solutions for high-volume hematology testing, the Atellica Hema 570 analyzer and Atellica Hema 580 analyzer. Both have a throughput rate of up to 120 tests per hour. The Atellica Hema 570 measures 43 cell parameters and an additional 12 parameters are available on the Hema 580, including immature red blood cell indicators relevant to certain patient populations and an optical-based platelet count that reduces analytical interferences found in other detection technologies. Their user-friendly designs aim to reduce daily maintenance while supporting rapid reagent changes.
Read More »Momentum Consulting offers plan for lab optimization
June 2023—Momentum Consulting has kicked off its laboratory optimization program for CLIA labs that are emerging from the COVID pandemic with broad or specific needs. The plan covers maximizing existing resources in a laboratory for output in areas such as specimen processing, workflow, new test menus, quality management, billing and collection, sales revenue, and more. Momentum has a team of subject matter experts in these fields to help laboratories improve their productivity.
Read More »Early Lyme disease detection
June 2023—Zeus Scientific has introduced the Zeus Borrelia Modified Two-Tiered Testing (MTTT) algorithm, an FDA-cleared all-ELISA algorithm designed to improve the detection of early Lyme disease by up to 30 percent compared with the standard two-tiered testing algorithm. It replaces a second-tier immunoblot with a second Zeus ELISA methodology to provide additional supportive evidence of infection. The company offers two algorithms: Zeus Borrelia MTTT-1 uses the Zeus ELISA Borrelia VlsE1/pepC10 IgG/IgM test system as a first-tier test followed by the Zeus B. burgdorferi IgG/IgM test system; Zeus Borrelia MTTT-2 uses the Zeus ELISA Borrelia VlsE1/pepC10 IgG/IgM test system as a first-tier test followed by the Zeus B. burgdorferi IgG test system and Zeus B. burgdorferi IgM test system.
Read More »Visiopharm launches Ki-67 algorithm
June 2023—Visiopharm announced the launch of a next-generation Ki-67 algorithm designed to automate the scoring of Ki-67 slides. Tumor nuclei are counted based on their Ki-67 expression and the resulting proliferation index for the whole tumor area is calculated. The algorithm has been cleared under the European Union in vitro diagnostic regulation and is for research use only in the United States.
Read More »Beckman Coulter unveils DxI 9000 Access
June 2023—Beckman Coulter unveiled its DxI 9000 Access immunoassay analyzer. The analyzer can run up to 215 tests per hour per square meter and does not require daily maintenance; its weekly maintenance time is less than 15 minutes. It is available in most countries.
Read More »BD gets clearance for MRSA diagnostics AI software
June 2023—BD received FDA 510(k) clearance for the BD Kiestra methicillin-resistant Staphylococcus aureus imaging application, which uses artificial intelligence to automate the task of inspecting Petri dishes to determine if there is bacterial growth. The application can evaluate single specimens or group together a large volume of plates with nonsignificant growth for batch review and release negative results. It uses AI algorithms to look for specific culture characteristics on the BBL Chromagar MRSA II plate. Based on that information and analysis by BD Synapsys informatics, plate images are automatically organized and sorted into meaningful worklists, the company says.
Read More »Verichem calibration verifiers for cholinesterase testing
June 2023—Verichem Laboratories offers multilevel calibration verification materials for cholinesterase activity as part of its liquid-stable and ready-to-use Enzyme ER Verifier kit. The verifiers are designed to include at least one concentration level of enzyme activity within the normal range and intended to be treated as patient specimens. The material’s protein balance, pH, and ion content are constant across concentration levels. The kit material also contains multilevel assigned activities for alanine aminotransferase, alkaline phosphatase, alpha-amylase, aspartate aminotransferase, creatine phosphokinase, gamma-glutamyl transferase, lactate dehydrogenase, and lipase. Each kit has 5 mL of material for each of the six levels and a shelf-life and open-vial stability of 18 months.
Read More »Trane Technologies acquires Helmer Scientific
June 2023—Trane Technologies has acquired Helmer Scientific. Trane says the acquisition will broaden its capabilities in precision temperature control and its existing portfolio of life science solutions under the Farrar brand. Trane will leverage its operating system and expertise in sustainability, services, worldwide distribution, and commercialization to accelerate Helmer’s continued growth.
Read More »FDA clears Panther Fusion SARS-CoV-2, flu, RSV assay
June 2023—Hologic announced the FDA has granted 510(k) clearance for its Panther Fusion SARS-CoV-2/Flu A/B/RSV assay. This molecular diagnostic test, previously under EUA, detects and differentiates SARS-CoV-2, influenza A and B, and respiratory syncytial virus. It runs on the Panther Fusion system, which provides initial results in about three hours and can process more than 1,000 tests in 24 hours.
Read More »Labcorp gets exclusive license for Proteomedix prostate cancer test
June 2023—Proteomedix (Schlieren, Switzerland) announced an exclusive license agreement in which Labcorp will develop and commercialize its Proclarix prostate specific antigen test in the United States. Proclarix is performed using the same blood sample as a PSA test and is designed to help determine the risk of clinically significant prostate cancer for men who have an elevated total PSA and a digital rectal examination finding that indicates elevated prostate volume but who are not suspected of having cancer.
Read More »Quansys Biosciences bovine-specific ELISAs
June 2023—Quansys Biosciences has developed Q-Plex Bovine Cytokine Panel 1 (5-plex), an ELISA kit that can measure five levels of bovine and African water buffalo cytokines in plasma, serum, and cell culture supernatants. Each kit contains a 96-well plate featuring the relevant biomarker panel in each well and all reagents required to perform testing.
Read More »Randox introduces PSA and BNP control
June 2023—Randox Labs has released two new products, a prostate-specific antigen and brain natriuretic peptide control. The liquid-frozen PSA is manufactured using human serum and has an open-vial stability of 30 days at 2°–8°C and a shelf life of one year from the date of manufacture. The control is optimized for use with Roche systems but is also suitable for use across a variety of other platforms, the company says.
Read More »Laboratory billing services provider
April 2023—Laboratory Billings is a revenue cycle management firm that aims to help laboratories achieve a higher first-pass claims rate and reduce accounts receivable. The company offers medical billing services to clinical and reference laboratories and shares real-time analytics about the revenue cycle. “We help laboratories become financially resilient and reduce the complexities in the administrative workflows,” according to a company statement. “Our billing and coding experts possess in-depth understanding of the revenue cycle management.” More information about the company is available at https://laboratorybillings.com.
Read More »FDA clears POC instrument for fentanyl testing
April 2023—Shenzhen Superbio Technology has received FDA clearance for the Ryan immunofluorescence analyzer, a point-of-care instrument for in vitro diagnostic use only. Carolina Liquid Chemistries Corp. will distribute the analyzer and fentanyl urine detection kit in partnership with Bioeasy USA. The fentanyl detection kit is intended for the qualitative detection of fentanyl in human urine at a cutoff concentration of 1 ng/mL. It is intended for use with the Ryan analyzer, which provides results in less than six minutes.
Read More »Siemens, Sysmex sign OEM agreement
April 2023—Siemens Healthineers signed a global OEM agreement on hemostasis products with Sysmex Corp. Under the multiyear agreement, the companies will individually distribute their combined portfolio of hemostasis instruments and reagents worldwide under their respective brands.
Read More »FDA clears Simplexa COVID-19, flu A/B assay
April 2023—DiaSorin has received FDA 510(k) clearance for its Simplexa COVID-19 and Flu A/B Direct assay. The test is a real-time RT-PCR assay that detects and differentiates influenza A, influenza B, and SARS-CoV-2 viruses and is performed using nasopharyngeal swab samples. Results are available in a little more than an hour on the Liaison MDX instrument.
Read More »BioGx Candida auris PCR assay adoption accelerates
April 2023—BioGx announced accelerated adoption of its high-performance Sample-Ready PCR assay for the detection of Candida auris. A complete PCR mix is provided in a single lyophilized tube format to be validated by laboratories on a variety of real-time PCR instruments, including the BD Max, Thermo Fisher QuantStudio, Bio-Rad CFX Touch, and BioGx Pixl platform. The company says its assay has been successfully validated and implemented by public health laboratories and hospitals in several states.
Read More »BD introduces Rhapsody HT Xpress system
April 2023—BD introduced the BD Rhapsody HT Xpress system for single-cell multiomics analysis. The system enables users to isolate, barcode, and analyze single cells at a high sample throughput—up to eight times more cells, BD says, than prior versions of its single-cell analyzers. Users can analyze multiple samples and different cell sizes and types, such as stem cells or cancer cells, at the same time.
Read More »Quest unveils transplant diagnostic services
April 2023—Quest Diagnostics unveiled Quest Advanced Specialized Transplant Services, designed to expand access to laboratory tests for transplanting solid organ, human cells, and tissue. Quest says its new offering features a menu of more than 170 specialized transplant and infectious disease tests, longitudinal trends analysis of test results in the electronic medical record, and support services that include medical consultation. Patients can access Quest’s network of 2,100 patient service centers and 5,000 mobile and at-home phlebotomists to support patient-compliant pre- and post-transplant testing. The services are available to recipients and living donors across the United States, excluding Alaska and Hawaii.
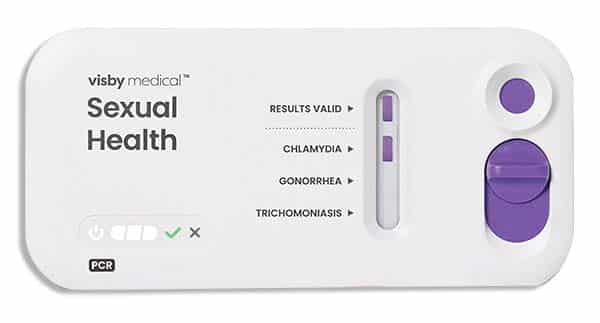
Read More »Visby gets FDA clearance, CLIA waiver for sexual health test
April 2023—Visby Medical has received 510(k) clearance and was granted a CLIA waiver from the FDA for its second-generation Sexual Health test. The point-of-care test uses PCR technology to detect sexually transmitted infections caused by Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis. Visby says the new device features improvements in workflow, manufacturability, and reliability. Test accuracy is about 97 percent and results are available in less than 30 minutes.
Read More »FDA clears Leica Biosystems MMR antibody panel
April 2023—Leica Biosystems announced FDA 510(k) clearance of the Bond mismatch repair antibody panel for use in screening patients who have colorectal cancer for the identification of Lynch syndrome. The panel is for in vitro diagnostic use on Leica’s Bond-III and Bond-Max immunohistochemistry automated instruments. Turnaround time is 2.5 hours on the Bond-III and about 3.5 hours on the Bond-Max.
Read More »Qiagen, Servier to develop CDx for acute myeloid leukemia drug
April 2023—Qiagen has entered into a strategic partnership with Servier to develop a companion diagnostic test for Tibsovo (ivosidenib), an isocitrate dehydrogenase-1 inhibitor indicated for the treatment of acute myeloid leukemia. Under the agreement, Qiagen will develop and validate a real-time in vitro PCR test that can be used to detect IDH1 gene mutations in patients with AML in whole blood and bone marrow aspirates. The companion diagnostic will run on Qiagen’s Rotor-Gene Q MDx instrument.
Read More »Enzo Biochem to sell clinical lab to Labcorp
April 2024—Enzo Biochem announced it has entered into an agreement in which Labcorp will acquire the assets of Enzo’s clinical laboratory division, Enzo Clinical Labs. Labcorp said the acquistion will bolster its commitment to the New York tristate health care communities. Completion of the sale is contingent on approval by Enzo’s shareholders and other customary closing conditions.
Read More »Verichem Matrix Plus chemistry reference materials
April 2024—Verichem Laboratories liquid-stable Matrix Plus chemistry reference materials for the calibration verification of wet chemistry assays are now available. The multilevel kit, along with an optional and standalone level F, supports overall system quality control and CLIA compliance. The six-level set of materials contains seven individual chemistry analyte components covering 48 separate and individual concentration levels.
Read More »Verichem reference materials for triglyceride assays
April 2023—Verichem Laboratories released ready-to-use reference materials for use with calibration verification testing of triglyceride assays. The product is available in a five-level standard kit and an optional, standalone, ultra-high sixth level, with levels ranging from 10 to 1,250 mg/dL. Tricglyceride values are established by gravimetric weight of glycerol expressed as triolein.
Read More »Bio-Rad, Element Biosciences to deliver RNA sequencing workflow
March 2023—Bio-Rad Laboratories and Element Biosciences announced a partnership to demonstrate the capabilities of the Bio-Rad Sequoia RNA sequencing library preparation portfolio on the Element Aviti benchtop sequencer.
Read More »Qiagen, Sophia partner to offer NGS solutions
March 2023—Qiagen has launched its QIAseq Platform Partnership program and a partnership with Sophia Genetics that will bring together QIAseq reagent technology for next-generation sequencing with the Sophia DDM digital analytics platform. The Platform Partnership program aims to enhance the use and compatibility of QIAseq NGS kits to support secondary analysis and tertiary interpretation workflows offered by other digital data-sharing and analytics companies. The kits are designed for use with any sequencer.
Read More »Verichem Enzyme ER Calibration Verifier kit
March 2023—Verichem Laboratories’ ready-to-use Enzyme ER Calibration Verifier kit is now available. The materials are designed for the calibration verification of enzyme assays on wet chemistry testing systems and contain nine clinical enzyme components covering 54 individual activity levels. The enzyme components within the materials include amylase, alanine aminotransferase, alkaline phosphatase, aspartate aminotransferase, cholinesterase, creatine phosphokinase, gamma glutamyltransferase, lactate dehydrogenase, and lipase. The materials are intended to be treated as patient specimens and have a shelf life of 18 months.
Read More »AscencioDx COVID-19 test, molecular detector get EUA
March 2023—Anavasi Diagnostics announced FDA emergency use authorization for its AscencioDx COVID-19 test and AscencioDx molecular detector for use in POC settings operating under a CLIA certificate of waiver, certificate of compliance, or certificate of accreditation. The system uses reverse transcription loop-mediated isothermal amplification for the qualitative detection of RNA from the COVID-19 virus in samples that are heated and illuminated via fluorescence signals in a reaction tube placed within the detector. Results are available in as few as 20 minutes. The detector is reusable for at least 3,000 test cycles, the company says, and is powered by a 12-volt adapter.
Read More »Globe Scientific introduces Globe/Euromex microscopes
March 2023—Globe Scientific has introduced a line of Globe/Euromex microscopes and microscope accessories. The line is composed of 24 CE-certified stereoscopic, compound, and inverted binocular and trinocular microscopes and 12 microscope accessories. Customers can access several support tools, including a microscope selection guide and Euromex Academy, which includes online video tutorials, user manuals, educational blogs, and more.
Read More »LGC launches BRCA1/2 reference materials
March 2023—LGC Clinical Diagnostics has launched the Seraseq gDNA BRCA1/2 LGR Somatic Mutation Mix and Seraseq gDNA BRCA1/2 LGR Inherited Mutation Mix. The products are intended for use as quality reference materials in developing, validating, and evaluating the routine performance of laboratory tests and are for research use only.
Read More »NRichDx products for developing liquid biopsy assays
March 2023—NRichDx has launched two products for developing liquid biopsy assays. The IVD-labeled NRichDx Revolution Circulating Free Total Nucleic Acid kit is used for extracting circulating cell-free total nucleic acid and comes in two kit sizes for sample volumes ranging from 1 to 20 mL.
Read More »Verichem reference materials for toxicology testing
March 2023—Verichem Laboratories announced the availability of ready-to-use, multilevel clinical reference materials intended for use with toxicology assays on a range of wet chemistry analyzers.
Read More »PerkinElmer debuts NGS workstation
March 2023—PerkinElmer debuted its Zephyr G3 NGS iQ workstation at the Society for Laboratory Automation and Screening conference in San Diego, Feb. 25–March 1. The system enables the automated construction of up to 96 NGS libraries and is composed of a high-performing liquid handler, integrated thermocycler, robotic arm, and deck accessories and peripherals. The workstation is available with an optional cloud-based software to further simplify creating and editing NGS protocols, the company says, by using a single pane of glass interface and developing methods from scientific intent rather than code.
Read More »FDA expands approval of BD Onclarity HPV assay
March 2023—BD announced FDA market approval for the Onclarity HPV assay to be used with the Hologic ThinPrep Pap test. The Onclarity HPV assay detects and identifies 14 high-risk human papillomavirus types in a single analysis. The assay reports genotypes 16, 18, 31, 45, 51, 52, 33/58, 35/39/68, and 56/59/66, and has FDA approval for use in vaccinated women.
Read More »Flagship, Genomenon partnership
March 2023—Flagship Biosciences announced a partnership with Genomenon in which Genomenon’s Mastermind knowledge base will provide disease-specific genomic data sets to enrich Flagship’s genomic profiling services. The partnership aims to accelerate biomarker discovery and companion diagnostics development solutions and improve patient stratification for clinical trials and treatment selection.
Read More »SeraCare releases HRD reference materials
March 2023—LGC SeraCare has developed three contrived, tumor normal cell-line–derived homologous recombinant deficiency reference materials: Seraseq FFPE HRD High-Positive RM, Seraseq FFPE HRD Low-Positive RM, and Seraseq FFPE HRD Negative RM.
Read More »Agilent releases on-deck thermal cycler for Bravo NGS platform
March 2023—Agilent Technologies released an on-deck thermal cycler that integrates with the company’s Bravo NGS automated liquid handling platform. The optional accessory enables the Bravo platform to provide thermal cycling as part of an automated protocol and extends walkaway time. The setup is beneficial, the company says, for applications such as library preparation and target enrichment steps in next-generation sequencing workflows, end-point PCR, and cell-based applications.
Read More »Bio-Rad launches real-time PCR system
March 2023—Bio-Rad launched its CFX Opus Deepwell Dx real-time PCR system, an open platform for developing in vitro diagnostic assays. The system multiplexes up to five targets and allows reaction volumes up to 125 µL in a 96-well format for quantitative PCR diagnostic assays. CFX Maestro Dx SE software provides data management and analysis tools.
Read More »BioMérieux Vidas Kube gets CE mark
January 2023—BioMérieux announced the CE marking of its Vidas Kube, a next-generation automated immunoassay system for the company’s Vidas range. The system features a stackable and modular benchtop design and is compatible with the existing routine test menu covering emergency and critical care, immunochemistry, and infectious diseases. The commercial launch is planned in selected countries at the beginning of the year, extending gradually to the rest of the world during the second quarter.
Read More »Roche launches antibody to identify PRAME protein expression
January 2023—Roche has launched its Anti-PRAME (EPR20330) Rabbit Monoclonal Primary Antibody to identify PRAME protein expression in tissue samples from patients who are suspected of having melanoma. Studies suggest that the detection of PRAME expression by immunohistochemistry complements findings from routinely used tests and enables more informed clinical decisions and improved patient outcomes, according to a company press release.
Read More »General Data launches LaserTrack Flex
January 2023—General Data Healthcare launched its LaserTrack Flex cassette printer. The Flex has a footprint of 13” L × 19” W and a capacity that starts at 480 cassettes, with each magazine holding 80 cassettes; it can be upgraded to hold up to 800 cassettes. The standard configuration prints on the face of the cassette and can be upgraded to also print on one or both sides. Cassettes can be loaded on the right or left side of the printer.
Read More »EKF handheld hemoglobin analyzer with POC connectivity
January 2023—EKF Diagnostics’ DiaSpect handheld hemoglobin analyzer now connects securely to EKF Link, the company’s point-of-care middleware. DiaSpect is also now equipped with Bluetooth and internal storage for 4,000 tests and QC results, as well as the capability to provide a calculated hematocrit value alongside the hemoglobin result. Test results are stored and accessed securely on a centralized platform.
Read More »Santé, Vanguard Healthcare Staffing announce partnership
January 2023—Santé Consulting and Vanguard Healthcare Staffing announced a partnership in which the companies will provide recruiting support for anatomic and clinical laboratories.
Read More » CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management