July 2022—Scopio Labs received FDA 510(k) clearance for its X100HT hematology digital cell morphology platform with peripheral blood smear review.
“We’re excited to expand our suite of fully digital AI-powered diagnostic platforms to accelerate PBS analysis, improve consistency of results, and reduce review time,” Erez Naaman, CTO and cofounder of Scopio Labs, said in a press statement. “At Scopio, we are determined to usher in the digital revolution to laboratory medicine. Our devices offer complete remote capabilities for real-time diagnosis and treatment decisions, allowing experts to review, collaborate, and consult from anywhere, and at any time, using our AI-powered applications.”
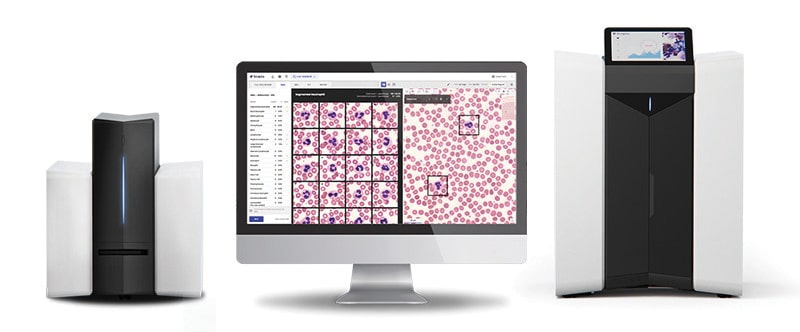
The X100HT holds 30 slides, processes up to 40 samples per hour, and supports remote review capabilities.
Scopio Labs, 631-637-4113