April 2020—CAP Publications released this month a new book titled Transfusion Medicine: A Compendium of Educational Cases, from the CAP Transfusion, Apheresis, and Cellular Therapy Committee. In it are 20 cases, each with a history, discussion, and questions and answers. The cases are divided into six sections: regulatory issues, peripartum/neonatal/pediatric transfusion medicine, platelets and plasma, red blood cells, hemolysis, and transplantation.
CAP TODAY spoke with Julie Katz Karp, MD, one of the book’s editors (the others are Jay Hudgins, DO, MS; Matthew Karafin, MD, MS; and Joseph Schwartz, MD, MPH). She is associate professor, Department of Pathology, Anatomy, and Cell Biology, and director of transfusion medicine, Thomas Jefferson University Hospital. Here is what she told us. (For an excerpt, see below.)
 What led you and your colleagues to write this book, and what is the source of the content?
What led you and your colleagues to write this book, and what is the source of the content?
Each year, the members of the CAP Transfusion, Apheresis, and Cellular Therapy (TACT) Committee (formerly the Transfusion Medicine Resource Committee) work tirelessly to create educational enhancements for various proficiency testing Surveys, namely the J-Series and the Expanded Transfusion Medicine Exercises (ETME). However, once those Surveys are completed by each participating laboratory, the Survey and the educational enhancements are often relegated to a laboratory filing cabinet. The members of the TACT Committee wanted to find a way to compile those educational enhancements in a way that liberated them from the filing cabinets and made them more accessible to transfusion medicine practitioners and trainees. And so this book was born. The content of the book is largely unchanged from its original publication in the Surveys between 2008 to 2017, but content was updated, when appropriate, to reflect current practice.
Did the cases come from the patient files of the book’s editors and case authors? How were they selected?
The cases in the book were originally published as educational enhancements for various proficiency testing Surveys. TACT Committee members will volunteer to write these educational enhancements during their tenure on the committee. The topics covered by these cases will often reflect clinical cases or experiences, but also changes and hot topics in transfusion medicine. The cases in this book were selected for their educational value and to provide a broad overview of many areas of the field.
Who is the intended audience, and what is the most important thing for them to know about this book?
The intended audience is anyone training in transfusion medicine, at any level. Review/question books in transfusion medicine are few in number, and I’m delighted that there’s now another one available. I think the most important thing for the audience to know about this book is that it is a testament to the hard work and productivity of the CAP TACT Committee. If they are impressed by the content of the book, they should consider joining the TACT Committee, or any CAP committee. CAP committees are a great way to learn, to network, and to contribute.
What can you tell us about the book’s many contributors?
The book’s 19 contributors include members of the CAP TACT Committee, both past and present. These contributors are all leaders in our field, and I’m so humbled to call them my friends and colleagues. It’s an amazing group of authors.
How does the book differ from other transfusion medicine books on the market?
This book differs in two ways. First, this is the only review/question book that is drawn from proficiency testing Surveys. These cases are previously published and the questions have already been answered by Survey participants. These cases and questions are well written and they are worthy of publication in a more permanent format. Second, this is the only review/question book with such an impressive set of authors. It’s not every day that a review/question book has 19 contributors, all of whom are nationally and internationally recognized blood bank/transfusion medicine professionals.
To order (PUB228), call 800-323-4040 option 1 or go to www.cap.org (Shop tab) ($68 for CAP members, $85 for others). If you are interested in writing a book, contact Caryn Tursky at ctursky@cap.org.
Here, from the new book Transfusion Medicine: A Compendium of Educational Cases, is case No. 14, from the red blood cells section. Dr. Karafin is medical director, Medical Sciences Institute, Versiti; associate investigator, Blood Research Institute, Versiti; and associate professor, Department of Pathology and Clinical and Translational Science Institute, Medical College of Wisconsin.
Anti-G and Anti-C
Matthew S. Karafin, MD, MS
Case History: Part 1
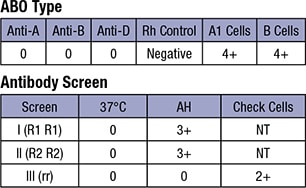
A 28-year-old African American female presented in the obstetric clinic after a miscarriage at 17 weeks gestation. A blood sample was sent with a request for Rh immune globulin (RhIg) for intramuscular injection. Per report, she has never had a previous transfusion but has had two full-term uncomplicated pregnancies. According to previous records, she received appropriate RhIg prophylaxis for these previous pregnancies. The blood bank initial results are shown on next page.
Case History: Part 2
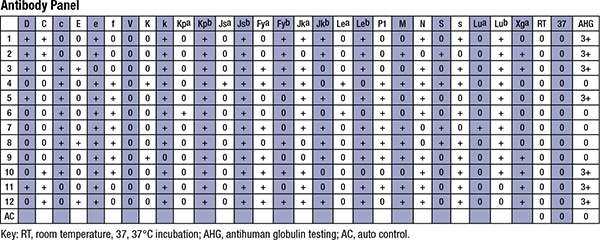
The following additional tests were performed by the blood bank. See Panel A and Panel B below.
Case Questions
1. What blood type is this patient?
a. O D-
b. O D+
c. AB D-
d. AB D+
e. B D-
2. Based on the provided clinical history, the reactivity seen on the antibody panel suggests which of the following?
a. The patient has anti-D. No additional testing is needed.
b. The patient potentially has anti-C. Additional testing is needed.
c. The patient potentially has anti-C and anti-D. Additional testing is needed.
d. The patient has anti-D and anti-C. No additional testing is needed.
e. The patient potentially has anti-D and anti-C. Additional testing is needed.
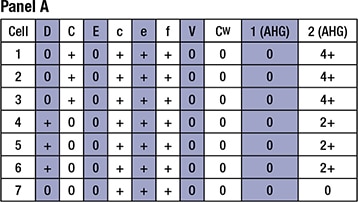
Panel A represents the patient’s serum sample adsorbed with r’ (Ce) RBCs. Column 1 represents the patient’s serum sample after adsorption, and column 2 represents the eluate prepared from the r’ (Ce) RBCs.
3. Based on the findings, what further testing would you do?
a. Adsorption of the patient’s serum
b. Adsorption and elution of the patient’s serum
c. Kleihauer-Betke test
d. Donath-Landsteiner test
e. No further testing is needed
4. If this patient were to need a transfusion now, what would be the safest blood to transfuse at this time?
a. Crossmatch-compatible O D-, C- blood
b. Crossmatch-compatible O D+ blood
c. Crossmatch-compatible O D- blood
d. Crossmatch-compatible O D+, C- blood
e. Crossmatch-compatible AB D-, C- blood
5. Should this patient receive RhIg?
a. Yes
b. No
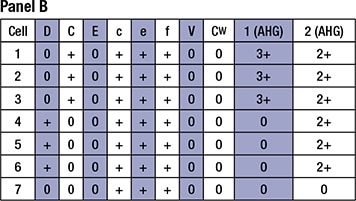
Panel B represents the patient’s serum sample adsorbed with R0 (Dce) RBCs. Like Panel A, column 1 represents the patient’s serum sample after adsorption, and column 2 represents the eluate prepared from the R0 (Dce) RBCs.
6. Based on the work above, what anti-body(ies) does the patient actually have?
a. Anti-C
b. Anti-G
c. Anti-D and anti-C
d. Anti-G and anti-C
e. Anti-D, anti-C, and anti-G
7. If a patient is identified with an anti-G and requires an RBC transfusion, how would you select units for compatibility testing?
a. Crossmatch D-negative, C-negative units
b. Crossmatch D-negative, C-negative, E-negative units
c. Crossmatch D-negative, E-negative units
d. Screen D-negative units with anti-G, then crossmatch D-negative, G-negative units
e. Obtain G-negative units from the blood provider
8. A serum sample that appears to contain anti-D and anti-C is adsorbed with R0 (Dce) RBCs. What antibodies may be present in the adsorbed serum?
a. Anti-C
b. Anti-D
c. Anti-G
d. Anti-D and anti-G
e. Anti-C and anti-G
9. Which of the following patients is eligible for RhIG?
a. A patient with anti-D
b. A patient with anti-C and anti-G
c. A patient with anti-C, anti-D, and anti-G
d. A patient with anti-C and anti-D
e. A patient with anti-D and anti-G
10. Which of the following Rh phenotypes is most likely G-negative?
a. R0R0 (Dce)
b. r’r” (CcEe)
c. r”r” (cE)
d. R1R1 (DCe)
11. Which is the correct order of potential severity of hemolytic disease of the newborn due to the following antibodies, from most severe to least severe?
a. Anti-D > anti-G > anti-C
b. Anti-G > anti-D > anti-C
c. Anti-D > anti-C > anti-G
d. Anti-C > anti-G > anti-D
12. Which of the following patients who have allo–anti-D and allo–anti-C by general antibody panel would need an adsorbtion/elution study?
a. A 65-year-old female who needs a transfusion due to symptomatic anemia
b. A 19-year-old nonpregnant female who needs blood because she was shot
c. A 26-year-old female who needs blood due to severe postpartum bleeding
d. A 75-year-old male who needs blood intraoperatively while receiving a liver transplant
e. An 18-year-old male who needs blood and was in a car accident
Case Discussion
This case describes a 28-year-old multiparous female who presented for possible RhIg after a miscarriage. The goal of this case was to describe the clinical and serologic difficulties that arise in a patient with an anti-G. Distinguishing between anti-C and anti-D versus anti-G is important in order to determine if the woman requires RhIg (to prevent anti-D formation). This scenario requires an adsorption and elution study.
Question 1 requires an understanding of forward and reverse typing. The forward type tests the recipient’s red cells with reagent anti-A, anti-B, and anti-D. The present example reveals that no agglutination was observed with any of the reagents, suggesting that the patient has no A, B, or D antigens on her red cells. This finding is confirmed by the reverse typing. Consequently, our patient has an O blood type. The reverse type does not check for anti-D in the patient’s serum as anti-D is not a naturally occurring antibody, and its presence is investigated by the red cell antibody screen (see question 2).
Question 2 tests multiple concepts. First, this question requires an appropriate interpretation of the antibody screen and antibody panel. In both the antibody screen and antibody panel, the patient’s serum (or plasma) is tested using a panel of group O red cells of known antigenic composition. The antibody screen determines the presence of a potentially clinically significant red cell antibody, and the antibody panel contains more O red cells such that the antibody specificity can be determined. In the present example, screening cell I (R1R1) and II (R2R2) are positive, indicating that a potentially significant alloantibody is present. The subsequent antibody panel reveals that the antibody is most likely an anti-D and an anti-C. This question also requires an understanding of anti-G. The G antigen is present on almost all D- or C-positive red cells and is absent from those cells that are C and D negative. The G antigen epitope is on Rh proteins (Ser103) expressing the D or C antigen. After transfusion of Rh-negative, C-positive blood, Rh-negative patients can make an anti-G, which appears to be an anti-C and anti-D on an antibody panel. Anti-G is clinically significant and is known to cause hemolytic transfusion reactions and hemolytic disease of the fetus and newborn. The antibody panel shown is not capable of differentiating anti-D and anti-C from anti-G, so additional tests are warranted in this case.
Question 3 requires an understanding of the testing required to identify an anti-G and the situation in which this testing is warranted. Sequential adsorption and elution studies are needed to determine whether anti-G is present and if anti-C and/or anti-D are also present in the patient’s serum. Since the G antigen may mask the presence of anti-D on standard antibody panels, sequential adsorption and elution studies are needed when administration of Rh immune globulin for anti-D prevention is considered important. Consequently, D-negative women who are pregnant, like the patient in this case, will need adsorption and elution studies.
Question 4 requires an understanding of the G antigen. As noted in the answer to question 2, the G antigen is present mainly on red cells with the D or C antigens. Consequently, blood that is negative for D and C antigens will also be negative for G. Moreover, as this patient may also have anti-D or anti-C in addition to anti-G, providing ABO-compatible blood that is negative for D and C antigens should provide her with the safest possible transfusion.
Question 5 requires a correct interpretation of the adsorption and elution studies, and a correct understanding of the utility of RhIg. RhIg is used in pregnant women who are at risk for becoming alloimmunized to the D antigen. Differentiating anti-G from anti-D is important in pregnancy because a woman who has made an anti-G but not an anti-D is still a candidate for RhIg prophylaxis. Panel A represents the patient’s serum sample adsorbed with r’ (Ce) RBCs. As noted above, column 1 represents the patient’s serum sample after adsorption, and column 2 represents the eluate prepared from the r’ (Ce) RBCs. Based on the reactivity shown, this patient has an anti-C and/or anti-G (agglutination seen on the eluate made by a D- C+ red cell), but no anti-D, as the serum sample (column 1) showed no agglutination when D-positive red cells were tested. As noted above, panel B represents the patient’s serum sample adsorbed with R0 (Dce) RBCs. Like panel A, column 1 represents the patient’s serum sample after adsorption, and column 2 represents the eluate prepared from the R0 (Dce) RBCs. Since panel A demonstrated that our patient had no anti-D, the reactivity from the eluate in panel B (column 2) is inferred to be from the anti-G. However, unlike panel A, the serum remaining after the adsorption shows reactivity to C-positive D-negative red cells, indicating that this patient has both anti-C and anti-G, but no anti-D. Consequently, this patient is still a candidate for RhIg.
Question 6 requires a correct interpretation of the adsorption and elution studies. See question 1 for the explanation of the correct interpretation.
Question 7 requires an understanding of the G antigen. The G antigen is present on C-positive or D-positive red cells. Consequently, cells that are negative for D and C will be negative for G. Screening specifically for G-positive or G-negative units for the crossmatch is excessively time consuming and unnecessary. Moreover, there is no commercially available anti-G typing serum available. Consequently, requesting antigen-negative units from the blood provider would be costly and impractical. Lastly, the E antigen is unrelated to the G antigen.
Question 8 requires an understanding of the G antigen and the antibodies that can form from exposure to G-antigen–positive blood. A D-negative C-negative person could form antibodies to G, C, or D. If R0 (Dce) RBCs are used for an adsorption of such a patient’s serum, then anti-D and anti-G would be removed by the R0 cell, and only a possible anti-C would remain in the adsorbed serum, which is the correct answer.
Question 9 requires a correct understanding of the utility of RhIg. RhIg is needed in D-negative pregnant women who are not yet alloimmunized to the D antigen (do not yet have anti-D). The correct answer is the only choice that does not have an anti-D.
Question 10 requires an understanding of the G antigen. The G antigen on red cells is present in individuals who are D or C antigen positive. Conversely, virtually all individuals who are D and C antigen negative are also G negative. Consequently, the person least likely to have the G antigen is someone with the r” (cE) phenotype.
Question 11 requires an understanding of the immunogenicity of the D, C, and G antigens. D is highly immunogenic, and anti-D is well known to cause severe hemolytic disease of the fetus and newborn. Anti-C is less common, and while HDFN has been reported, its effects are usually mild, and serious hemolytic disease is rare. Lastly, anti-G has been rarely reported to reach a high titer and thus is reported to be less of a threat than either anti-D or anti-C.
Question 12 requires a clinical application of the concepts addressed above. Anti-G can cause hemolysis due to an incompatible transfusion and/or HDFN. Rh-negative patients with a possible anti-G can be safely transfused with D-negative C-negative blood because the G antigen is absent from D-negative C-negative blood. Consequently, adsorption and elution studies to detect anti-G are unnecessary in most clinical situations. The only time that anti-G should be definitively evaluated is in situations where the use of RhIg is in question. As noted previously, anti-G presents as an anti-C and anti-D on standard antibody panels. Since the use of RhIg in D-negative pregnant women depends on the presence or absence of anti-D, the use of the adsorption-elution study is needed to uncover the possible underlying anti-D. Patients who are males, not of child-bearing age, or not pregnant will not need additional workup.
Reference
1. Shirey RS, Mirabella DC, Lumadue JA, Ness PM. Differentiation of anti-D, -C, and -G: clinical relevance in alloimmunized pregnancies. Transfusion. 1997;37:493-496.
Answers to questions: 1a, 2e, 3b, 4a, 5a, 6d, 7a, 8a, 9b, 10c, 11c, 12c.