[one_third]
[/one_third]
[one_third]
[/one_third]
[one_third_last]
[/one_third_last]
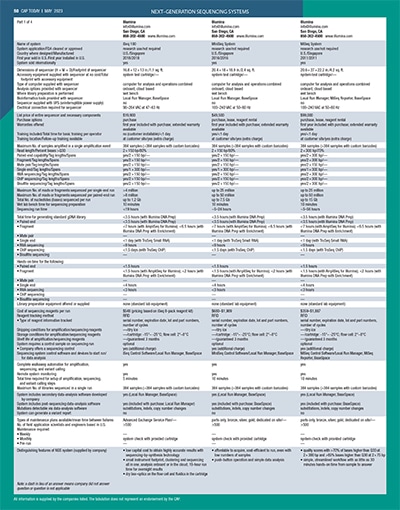
[wpdatatable id=81]
[one_third]
[two_third_last]
The outlook for in-house next-generation sequencing
Bringing next-generation sequencing in-house was at the center of a March 27 roundtable led by CAP TODAY publisher Bob McGonnagle, with costs, reimbursement, equity, and the electronic health record part of the conversation. Jeremy Segal, MD, PhD, of the University of Chicago, explains why the Genomics Organization for Academic Laboratories was formed. “By lowering barriers and encouraging cooperation,” he said, “we’ve seen our labs increase the pace of development and the quality of the assays they’re bringing on.”
There’s increasing interest in bringing next-generation sequencing in-house as opposed to sending it to a reference laboratory. While there are costs associated with bringing it in-house, if you look at total cost of the test and turnaround time, it’s an even horse race or slightly preferable to bring it in-house. Luca Quagliata, would you agree with that?
Luca Quagliata, BCMAS, PhD, VP and global head of medical affairs, Thermo Fisher Scientific: I do agree. Cost is an important component of being able to bring these tests in-house. We often think cost is driven only by reagents, but it’s not the component that most impacts overall expenditure. Cost of personnel to perform the analysis is equally important. With solutions that are going toward fully automated or semiautomated systems, you can free hands, better utilize the lab workforce, and effectively maximize budget.
The other part of the equation is the cost of treatment. A long turnaround time to results usually means placing the patient on any available treatment, which is not necessarily the best option. If a suboptimal treatment decision is made and subsequently a treatment change needs to be forced, the patient will pay for it in terms of outcomes. The health care system will probably also bear additional costs because those patients usually experience more side effects, which means rehospitalization costs and a prolonged process.
Fiona Nohilly, can you comment on the movement to in-house and how you look at the total cost of performing the test for the benefit of patients?
Fiona Nohilly, senior manager, product marketing, Americas regional marketing, Illumina: We are thinking about the total cost of ownership from sample and library preparation through sequencing and analysis. Since we last met for this roundtable, we have launched three new platforms.
Continue reading …
[/two_third_last]