Amy Carpenter Aquino
June 2023—Although goblet cell adenocarcinoma can label for neuroendocrine markers, it behaves as an adenocarcinoma and is staged as such. And it’s important to distinguish goblet cell adenocarcinoma from tubular neuroendocrine tumor, a rare subtype of neuroendocrine tumor.
With disseminated appendiceal mucinous neoplasms, distinguishing between low-grade and high-grade disease is important because only the latter may benefit from chemotherapy.
That was some of what Maryam Pezhouh, MD, MSc, associate professor of pathology at the University of California, San Diego, and Jacqueline Birkness-Gartman, MD, assistant professor of pathology at Johns Hopkins University School of Medicine, presented in their CAP22 session on appendiceal neoplasms. They walked attendees through a case-based look at the lesions that began with inflammatory lesions, structural abnormalities, polyps, and gynecologic proliferations (see part one, https://bit.ly/3MYZ2mA) and moved to appendiceal mucinous neoplasms, appendiceal adenocarcinomas (NOS), goblet cell adenocarcinomas, and neuroendocrine neoplasms.
Appendiceal mucinous neoplasm grading is based on whether the neoplasm is a low- or high-grade mucinous neoplasm or signet ring cell adenocarcinoma with signet ring cells infiltrating the tissue or in mucin pools. In the low-grade appendiceal mucinous neoplasm (LAMN), Dr. Pezhouh said, KRAS and GNAS mutations are common. However, GNAS mutations are less common in high-grade mucinous tumors. No longer recommended are the terms mucinous tumor of uncertain malignant potential, mucinous cystadenoma, borderline tumor, or mucinous cystadenocarcinoma, per the World Health Organization digestive system classification of tumors, fifth edition.
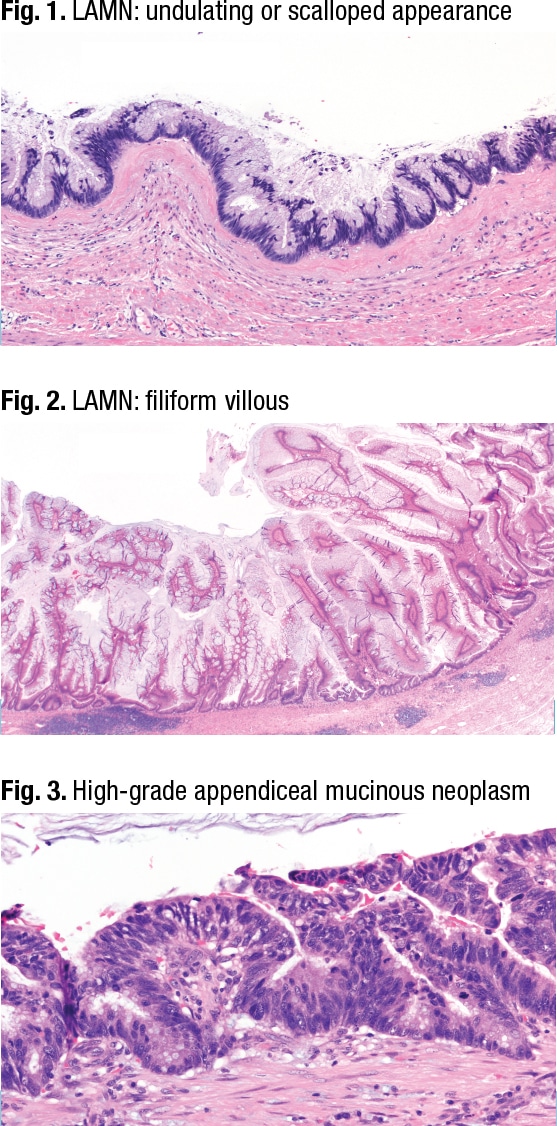
In Figs. 1 and 2 are two examples of LAMN, which can have different shapes and forms, including (not shown) an attenuated or flattened monolayer.
Cytologic features are the only difference between HAMN (high-grade appendiceal mucinous neoplasm) (Fig. 3) and LAMN, Dr. Pezhouh said. In HAMN, “the nuclei are becoming enlarged, you don’t have a lot of mucin on the surface, and you can see mitoses.”
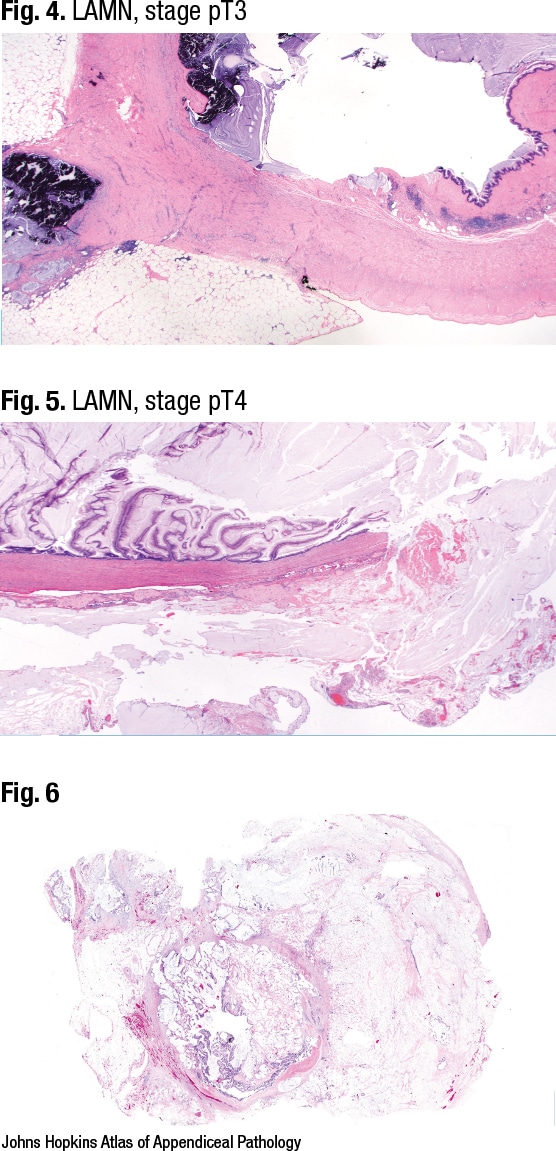
Dr. Pezhouh described T staging in mucinous neoplasms, beginning with pTis, which is “whenever your LAMN or HAMN is confined to the submucosa or muscularis propria. Whenever acellular mucin or mucinous epithelium invades into muscularis propria, it is still pTis. But when this passes the muscularis propria, and the tumor or acellular mucin extends to the subserosa, it’s pT3 [Fig. 4].” When the tumor or acellular mucin perforates and involves the serosa, it is T4 (Fig. 5).
Mucinous neoplasm prognosis is stage dependent, Dr. Pezhouh said. “When it’s limited to the appendix, it’s excellent. When it’s disseminated, it depends on the grade of peritoneal mucinous epithelium, extent of the disease, and the ability to achieve complete cytoreduction.” Data on HAMN prognosis is limited, she noted, but when it’s disseminated to the peritoneal cavity it behaves like the other counterparts.
Grade one mucinous neoplasms have low-grade cytology with a pushing border (LAMN). Grade two consists of two categories: high-grade cytology with a pushing border (HAMN) and invasive mucinous adenocarcinoma without a signet ring cell component. Grade three is defined by signet ring cells.
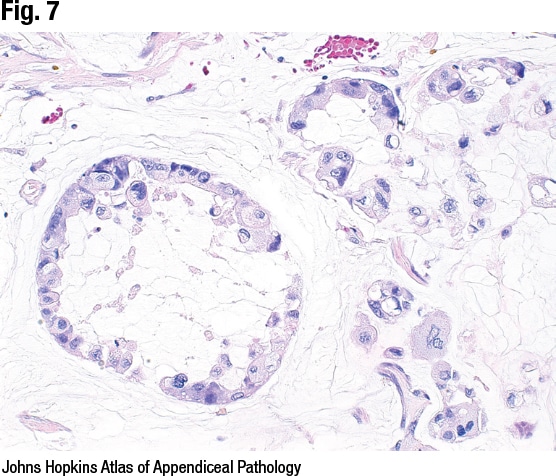
Dr. Birkness-Gartman reported on the case of a 50-year-old woman with a history of abdominal pain, increasing abdominal girth, and weight gain who was found to have an appendiceal mass. In Fig. 6 glandular epithelium can be seen in the lumen of the appendix. Small pools of mucin can be seen going through the appendix wall and out into the periappendiceal adipose tissue, and there are glands associated with the pools of mucin.
High magnification of one mucin pool with neoplastic epithelium reveals high-grade cytology, she said (Fig. 7). “Our cells are losing polarity, the nuclei are rounding up,” and there is pleomorphism. It is mucinous adenocarcinoma, WHO grade two, and “the defining feature that distinguishes this from HAMN is invasive growth,” she said. This can be angulated glands with desmoplastic stroma, as would be seen in a typical colon cancer case, or there can be a pattern that some call a small cellular mucin pool pattern. The small pools of mucin have glands or they have clusters of neoplastic epithelium, she said.
Grade two mucinous adenocarcinoma tends to behave aggressively and often has peritoneal involvement at presentation, Dr. Birkness-Gartman said, noting treatment involves a right hemicolectomy and otherwise is similar to colorectal adenocarcinoma treatment.
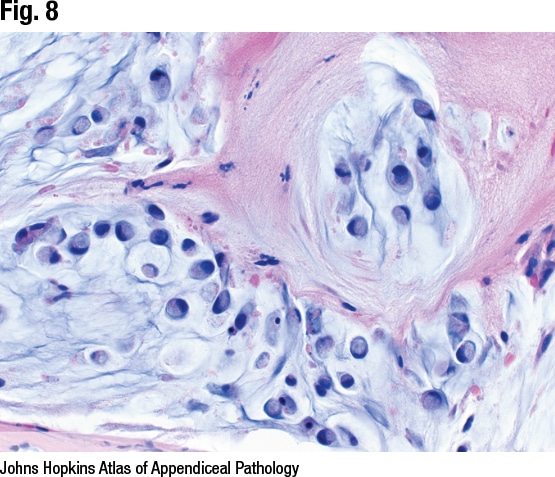
Dr. Birkness-Gartman presented a second case: a 60-year-old woman with chronic abdominal pain who underwent a CT scan that revealed an appendiceal mass and diffuse intraperitoneal disease. Typical signet ring morphology can be seen at high magnification (Fig. 8), and central mucin vacuoles are pushing the nuclei to the periphery. That it was positive for keratin proved it was signet ring cell adenocarcinoma rather than histiocytes, she said. “This is an example of mucinous adenocarcinoma with signet ring cells, WHO grade three.”
Signet ring cells can float in pools of mucin or infiltrate the tissue, she said, and they may make up the entire tumor or there may be a glandular component. “If signet ring cells make up more than 50 percent of the tumor,” she said, “then the correct diagnostic term is mucinous signet ring cell adenocarcinoma.” Peritoneal disease is commonly seen at presentation, and treatment is similar to that for colorectal adenocarcinoma. Colonic staging—T1 through T4—is used.
[dropcap]D[/dropcap]isseminated appendiceal mucinous neoplasms are clinically called pseudomyxoma peritonei, which is a term for mucin in the peritoneal cavity. In this situation, Dr. Birkness-Gartman said, typically there are many specimens from a debulking procedure and “buckets of mucin, and you need to sort through all of that, grossly and histologically.” This is most commonly of appendiceal origin, she noted, but it can be from other sites, including other sites in the tubular gastrointestinal tract and the pancreas.
For disseminated mucinous neoplasms, several grading systems have been used histologically and several systems for terminology, Dr. Birkness-Gartman said. The Peritoneal Surface Oncology Group International (PSOGI) consensus system includes terms for low-grade disease (mucinous carcinoma peritonei or disseminated peritoneal adenomucinosis) and high-grade disease (high-grade mucinous carcinoma peritonei or peritoneal mucinous carcinomatosis, with or without signet ring cells). “The fifth edition of the WHO and the eighth edition of the AJCC now include a three-tiered grading system with histologic criteria outlined for each grade,” she said.
For peritoneal metastasis, WHO classification grade one lesions are typically hypocellular at low magnification, have low-grade cytology, and lack infiltrative invasion, Dr. Birkness-Gartman said, adding that it looks like a LAMN but it’s outside the appendix. Grade two lesions are hypercellular at low magnification and have high-grade cytology and/or infiltrative invasion, often both, she said. And grade three lesions are defined by mucinous tumor deposits with signet ring cells.
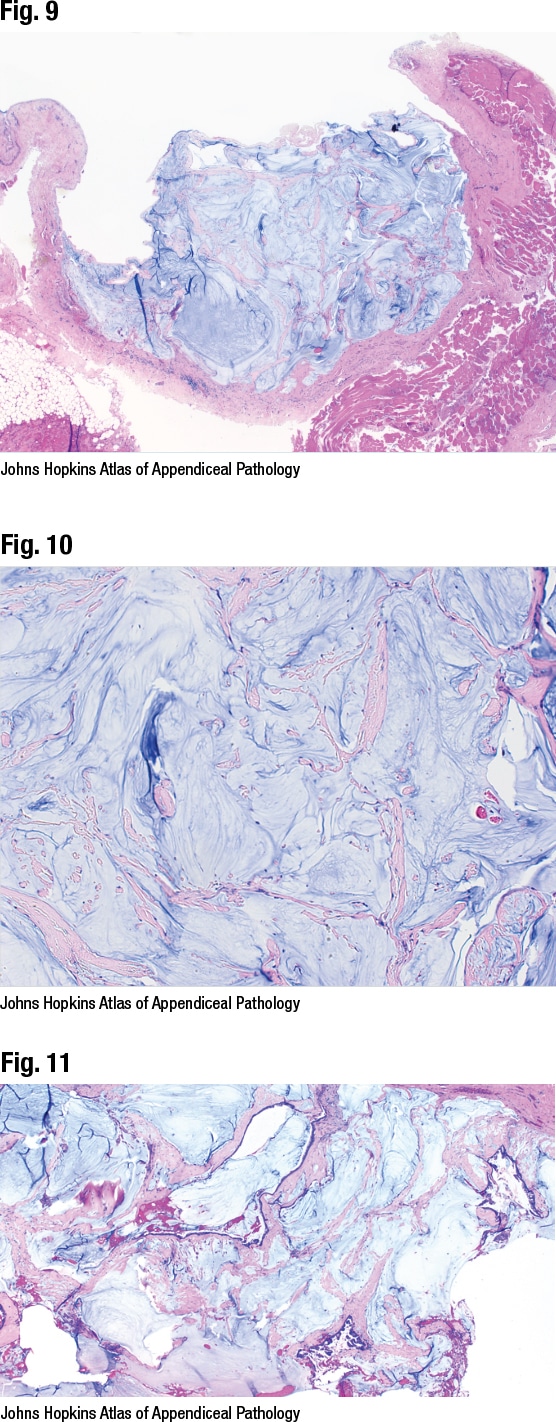
Dr. Birkness-Gartman shared the case of a 40-year-old woman with a history of LAMN and pseudomyxoma peritonei who underwent a second debulking procedure. In Fig. 9 a mucinous deposit is seen attached to the diaphragm, and skeletal muscle of the diaphragm is seen on right. In Fig. 10 pools of mucin are seen, possibly with a couple of inflammatory cells and some associated fibrosis, but no neoplastic epithelium. It’s an example of acellular mucin, Dr. Birkness-Gartman said, noting some prefer to call it mucin devoid of neoplastic epithelium. “Technically it can have some inflammatory cells in it,” she said, “but the important thing is we don’t see neoplastic epithelium.”
Foamy macrophages may be present in the mucinous deposits but shouldn’t be misinterpreted as signet ring cells, she cautioned (CD68 and AE1/AE3 or other preferred keratin can help). It’s treated with cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion. The risk of recurrence with absence of neoplastic epithelium is low, but adequate sampling is important, she said, “and make sure you sample any solid areas, anything that looks grossly different.”
Additional mucinous peritoneal deposits from the same patient were examined, and while large pools of mucin and fibrotic stroma can be seen, there is also neoplastic epithelium (Fig. 11). At low magnification the deposits have a smooth border, Dr. Birkness-Gartman noted; at higher magnification (Fig. 12) the smooth border with underlying fibrosis is seen, as is “low-grade mucinous epithelium where we have all the long axes of the nuclei lined up parallel to each other and perpendicular to the basement membrane.” It looks like a LAMN, she said, but because it’s in the peritoneum, it is an example of low-grade mucinous neoplasm, WHO grade one; by PSOGI it is low-grade mucinous carcinoma peritonei or disseminated peritoneal adenomucinosis.
Histologically, “you will see low cellularity at low magnification,” typically less than 20 percent of tumor at 20×, she said, and there should be both low-grade cytology and a pushing border—“the same criteria as for a LAMN but outside the appendix” (Valasek MA, et al. Adv Anat Pathol. 2018;25[1]:38–60). Lymph node involvement is rare. Treatment is cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion. Low-grade neoplasms typically do not respond to chemotherapy. “This is the important distinction in terms of calling low versus high grade. Low grade is not going to get chemo and high grade is.”
Experts disagree about whether it should be called a neoplasm or a carcinoma, she said. “The argument for neoplasm is that it lines up with the terminology used in the appendix” and may be less confusing for clinicians. “The argument for carcinoma is that it can behave aggressively and can cause significant morbidity and mortality,” with multiple intra-abdominal recurrences. “I see both arguments,” Dr. Birkness-Gartman said, and she recommends talking to clinicians to see which terminology they understand, with the aim of communicating, most importantly, whether it is low- or high-grade disease.
In another case, a 50-year-old woman with pseudomyxoma peritonei and suspected appendiceal adenocarcinoma underwent a debulking procedure. One of the peritoneal deposits can be seen in Fig. 13. Dr. Birkness-Gartman noted the higher cellularity at low magnification and the small cellular clusters and mucin in a fibrotic stroma, as well as some architectural complexity. Some of the architectural complexity in the cells was seen at higher magnification. “They’re starting to round up. They’re not lined up parallel to each other anymore.” It is an example of mucinous adenocarcinoma, WHO grade two (by PSOGI, high-grade mucinous carcinoma peritonei or peritoneal mucinous carcinomatosis).
Compared with a low-grade neoplasm, there is higher cellularity at low magnification—typically greater than or equal to 20 percent of tumor at 20×—and high-grade cytologic features or infiltrative invasion, typically both, she noted (Valasek MA, et al. Adv Anat Pathol. 2018;25[1]:38–60). The infiltrative invasion can include the small cellular mucin pool pattern, “so it may not be your typical angulated glands with desmoplastic stroma.” Lymph node involvement can occur (17 percent), and treatment for all is systemic chemotherapy (with additional treatment based on patient response) (Davison JM, et al. Mod Pathol. 2014;27[11]:1521–1539).
In this case, additional mucinous peritoneal deposits were examined (Fig 14). In addition to glands and nests of mucinous epithelium, single cells with signet ring morphology can be seen. The case is an example of a mucinous adenocarcinoma with signet ring cells, WHO grade three (by PSOGI, high-grade mucinous carcinoma peritonei with signet ring cells/peritoneal mucinous carcinomatosis with signet ring cells). “Histologically, as in the appendix, the signet ring cells can be seen floating in mucin or infiltrating the tissue,” Dr. Birkness-Gartman said. This is associated with a worse prognosis and a higher rate of lymph node metastasis (72 percent). Treatment is systemic chemotherapy (with additional treatment as needed) (Davison JM, et al. Mod Pathol. 2014;27[11]:1521–1539).
One pitfall is detached, degenerating cells (Fig. 15), which can mimic signet ring cells, she said. Fig. 15 is from a case involving a LAMN that disseminated to the peritoneal cavity and spread to the ovary; some of the cells resemble signet ring cells. In this case, though, “these cells were few and far between, and the rest of the neoplasm in this extensively sampled tumor was a low-grade type of neoplasm,” she said. Degenerative changes were also seen. “We ended up interpreting these as detached, degenerating cells rather than as a true signet ring cell component,” she said. “Some authors propose using a 10 percent signet ring cell cutoff for a true signet ring cell component because this produces better interobserver reproducibility” (Davison JM, et al. Mod Pathol. 2014;27[11]:1521–1539).
In staging disseminated appendiceal neoplasms, the M stage depends on the location of the neoplasm and whether there is neoplastic epithelium or just acellular mucin, Dr. Birkness-Gartman said. pM1a is intraperitoneal acellular mucin only without neoplastic epithelium, pM1b is intraperitoneal involvement with neoplastic epithelium, and pM1c is spread outside the peritoneum.
[dropcap]A[/dropcap]ppendiceal adenocarcinoma, not otherwise specified, can be similar to colorectal adenocarcinoma, Dr. Birkness-Gartman said, and may have an associated precursor lesion, such as an adenoma. Subtypes include mucinous adenocarcinoma with at least 50 percent mucin or a signet ring cell adenocarcinoma with at least 50 percent signet ring cells. “This can behave aggressively and is treated similarly to colorectal adenocarcinoma.”
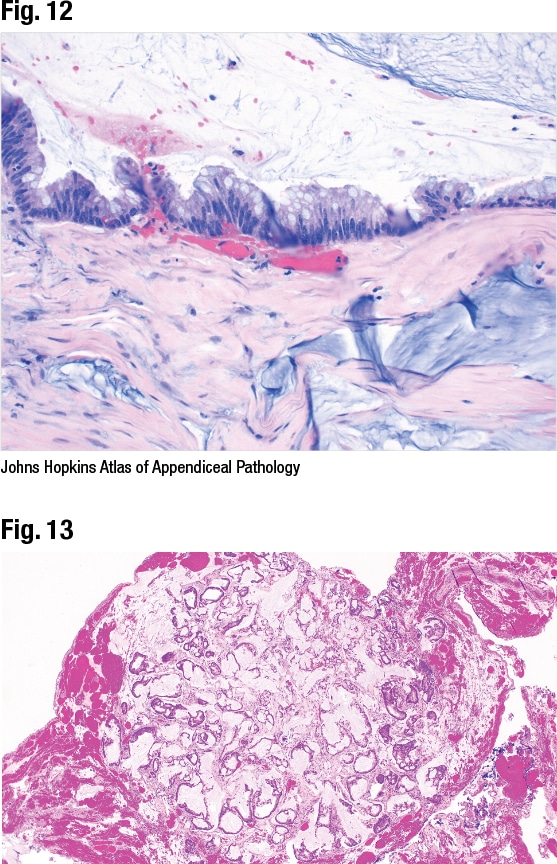
She presented the case of a 90-year-old woman with a diagnosis of presumed urachal adenocarcinoma involving the bladder who underwent an en bloc resection of the bladder dome, appendix, urachus, and umbilicus. In Fig. 16 is a bladder biopsy seen by her GU pathology colleagues. “They did not call this urachal carcinoma. They gave a descriptive diagnosis, explaining that it was intestinal-type adenocarcinoma and giving a differential that it could be coming from the gastrointestinal tract or it could be a primary bladder tumor.” The two cannot be distinguished well with immunohistochemistry, she said, “but clinically this was thought to be a urachal adenocarcinoma,” and the patient underwent a resection.
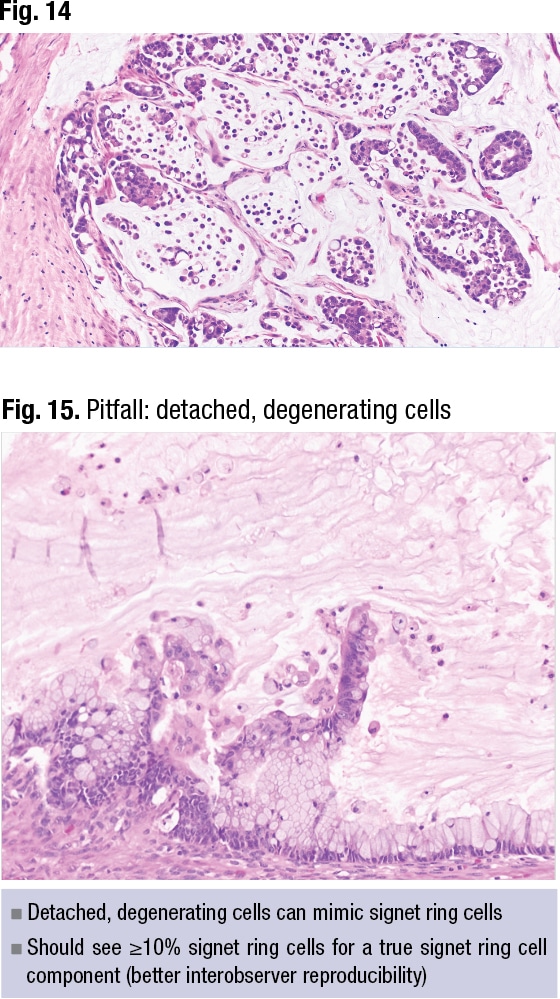
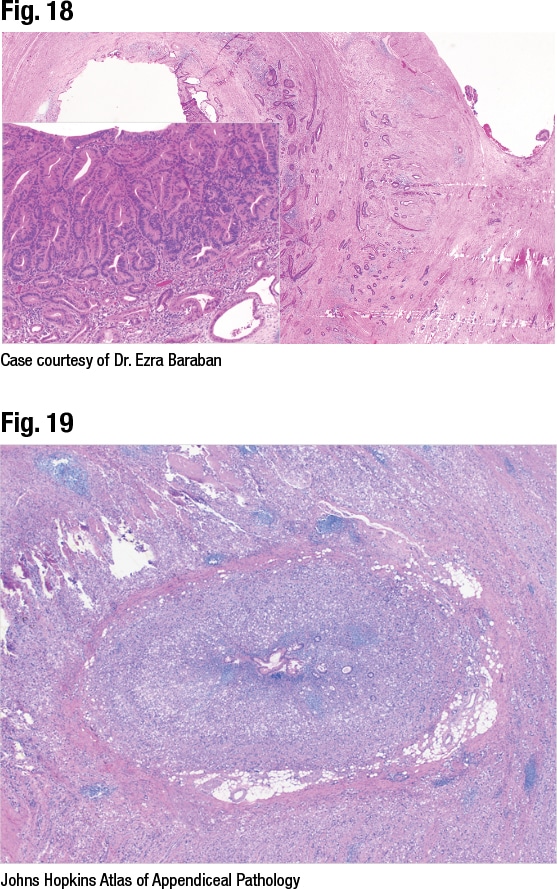
In Fig. 17 from this case is a sessile serrated adenoma or lesion involving the circumference of the appendix. There is evidence of dysplasia and an invasive carcinoma extending through the appendix wall (Fig. 18) and bladder invasion (not shown). The case was signed out as appendiceal adenocarcinoma, moderately differentiated, invading the bladder wall, with mention of the precursor sessile serrated adenoma/lesion with dysplasia. It was staged as T4b because it directly invaded the bladder, she said.
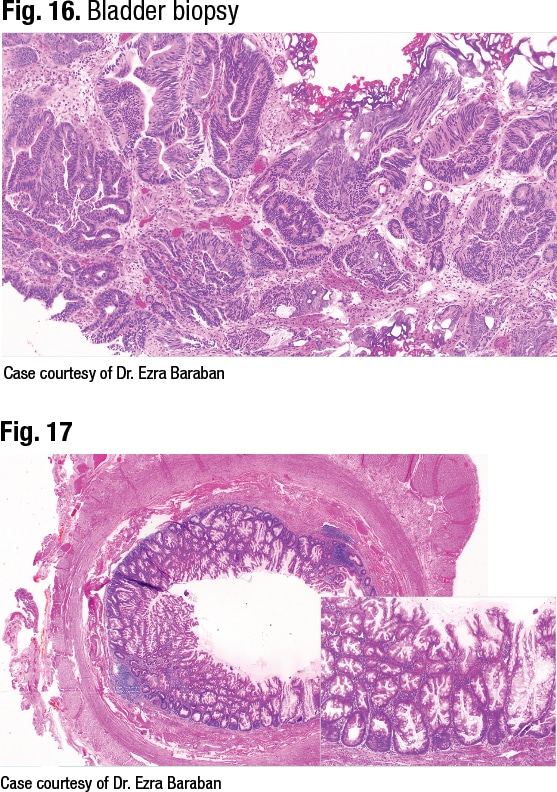
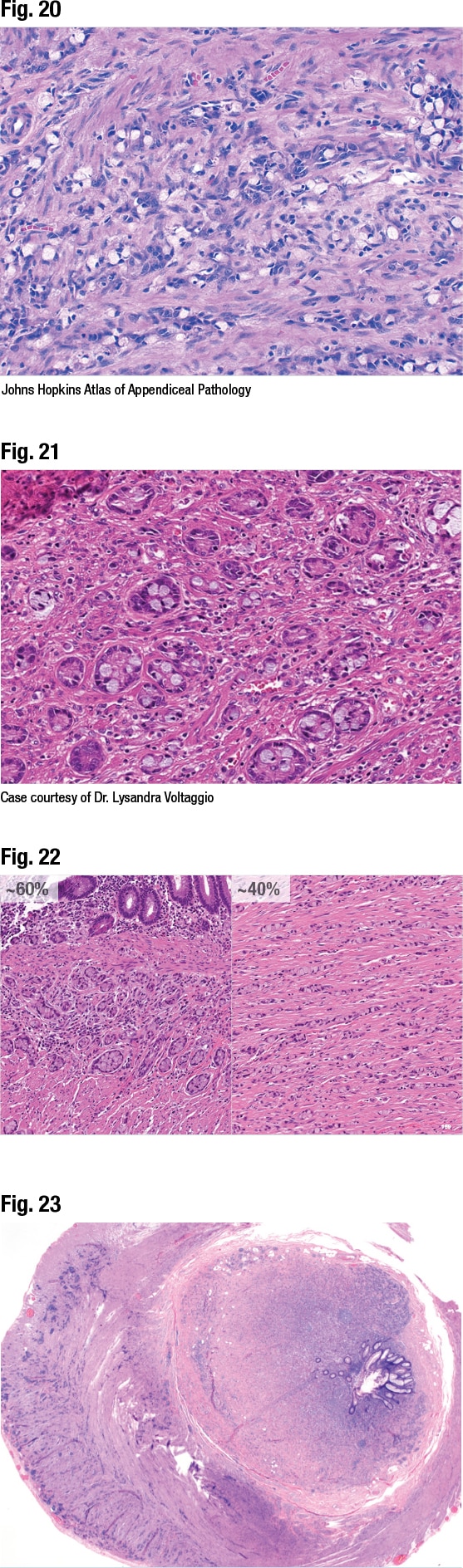
In another case, a 65-year-old woman with an appendiceal mass was treated with a right hemicolectomy. In Fig. 19 is a small portion of the normal appendiceal lumen, “but otherwise the whole appendix is overrun by a signet ring cell proliferation,” Dr. Birkness-Gartman said. On higher magnification signet ring cells are seen infiltrating the muscle as single cells (Fig. 20)—an example of signet ring cell adenocarcinoma of the appendix.
[dropcap]T[/dropcap]he newer WHO term goblet cell adenocarcinoma replaced the prior terms goblet cell carcinoid and adenocarcinoma ex goblet cell carcinoid. “This was previously thought of as a type of neuroendocrine neoplasm, but it’s now classified as an adenocarcinoma due to aggressive clinical behavior,” Dr. Birkness-Gartman said.
To make this diagnosis, histologically “you need to see, at least focally, tubules or clusters with goblet cell morphology,” she said. “This often shows focal labeling for neuroendocrine markers, but this is not necessary or sufficient for the diagnosis.” She therefore typically goes more with the morphology instead of staining.
The WHO’s current grading system for goblet cell adenocarcinoma is based on the percentage of low- versus high-grade patterns seen in the tumor. Low-grade pattern consists of the typical tubules or clusters with goblet cell morphology, “but this can show some variance such as limited tubule fusion or trabecular growth if the structures are cut tangentially,” she said. It may have associated acellular mucin and, rarely, bland tubules with oncocytic cytoplasm. High-grade pattern can have various morphologies: fusion of goblet cell clusters or tubules with complex architecture, large aggregates of goblet cells in mucin, sheet-like growth, single infiltrating cells, or conventional adenocarcinoma (Yozu M, et al. Am J Surg Pathol. 2018;42[7]:898–910).
In a case of an incidental appendiceal neoplasm, a 50-year-old woman presented with acute appendicitis, and the neoplasm was identified in an appendectomy specimen. At low power (not shown) the neoplasm was seen penetrating the appendix wall. The low-grade goblet cell adenocarcinoma morphology was seen at higher magnification—nests of cells with goblet cells in them (Fig. 21). Since this made up the majority of the tumor (> 75 percent low grade), it was graded as WHO grade one, Dr. Birkness-Gartman said.
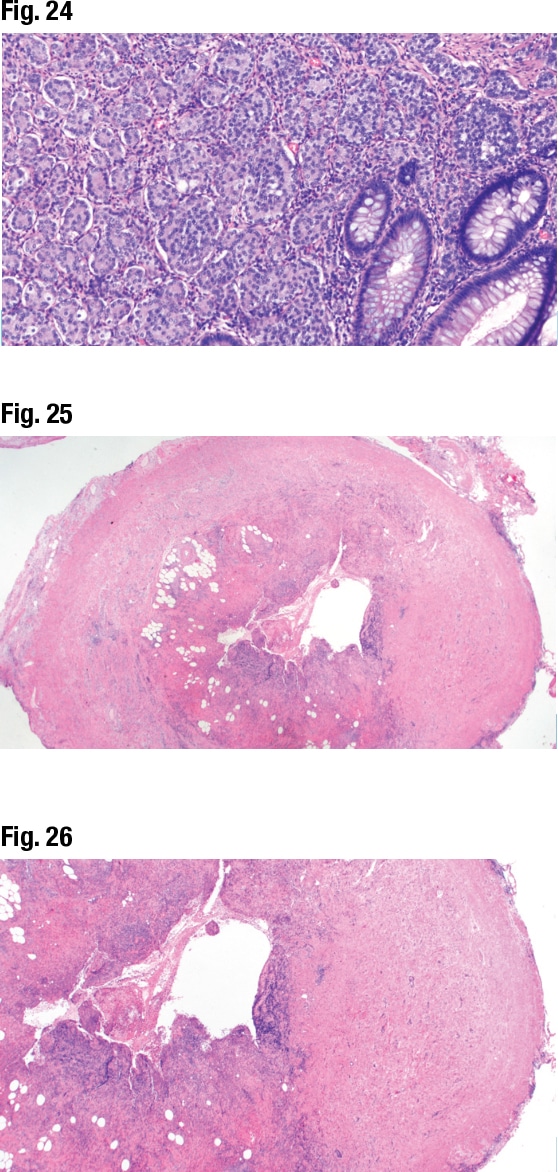
She also presented the case of an 85-year-old woman with acute appendicitis whose incidental neoplasm was identified in an appendectomy specimen. A proliferation was seen going through the appendix wall. Two patterns can be seen at higher magnification (Fig. 22): a low-grade goblet cell adenocarcinoma pattern with well-formed clusters of goblet cells making up about 60 percent of the tumor (on left) and single cells forming little lines resembling a signet ring cell adenocarcinoma making up about 40 percent of the tumor (on right). Based on the percentages of each pattern, she said, the tumor was WHO grade two (50 to 75 percent low grade).
[dropcap]D[/dropcap]r. Pezhouh began her section on neuroendocrine neoplasms with the case of a 29-year-old male who presented with abdominal pain not responsive to painkillers. A CT scan at an outside institution revealed acute appendicitis and appendicolith, she said, and on presentation at her institution, the patient’s pain was minimal, but he underwent an appendectomy because of the CT scan findings. In Fig. 23 a neoplasm can be seen expanding the layers of the appendix, extending through the muscularis propria and to the subserosa. In Fig. 24 a nested pattern can be seen with cells with a lot of cytoplasm and salt-and-pepper chromatin. It was a well-differentiated neuroendocrine tumor. IHC revealed the tumor to be positive for chromogranin and synaptophysin, she said.
In well-differentiated neuroendocrine tumors, the subtypes and grading are the same as for other parts of the GI tract, Dr. Pezhouh said. Grade one has a mitotic rate less than two and a Ki-67 index under three percent, grade two has a mitotic rate of two to 20 and a Ki-67 index of three to 20 percent, and grade three has a mitotic rate greater than 20 and a Ki-67 index greater than 20 percent. The 29-year-old patient had a grade one well-differentiated neuroendocrine tumor.
Neuroendocrine carcinomas are classified as large- or small-cell carcinomas or mixed adenoneuroendocrine, Dr. Pezhouh said. Staging is based mainly on tumor size and infiltration of the serosa/mesoappendix.
Prognosis of appendiceal well-differentiated neuroendocrine tumors is often excellent, she said. “Metastasis occurs rarely—1.4 to 8.8 percent, typically to regional lymph nodes. Distant organs are rare. Size is the most reliable predictor; usually when it’s less than one centimeter, risk of metastasis is low.” At 1 to 2 cm the risk of metastasis is 18 to 44 percent, and if larger than 2 cm the risk of metastasis is 30 to 86 percent. The European Neuroendocrine Tumor Society distinguishes invasion of more than 3 mm into the mesoappendix as an indication for extended resection, she said.
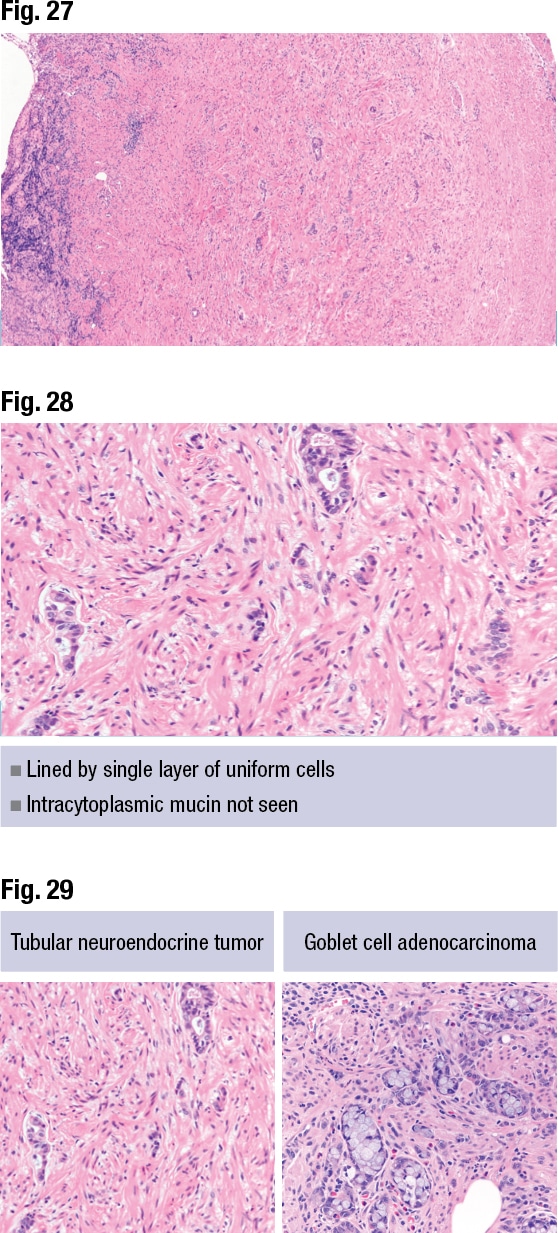
Dr. Pezhouh shared the final case: a 32-year-old male who presented with abdominal pain not responsive to painkillers, for whom a CT scan revealed acute appendicitis and an appendectomy was performed. It’s a case of acute appendicitis, she said, with loss of the mucosal layer and inflammation (Fig. 25). The tumor can be seen infiltrating the muscle (Fig. 26). In Fig. 27 some areas resemble a form of glandular formation, and in Fig. 28 there is no sign of intracytoplasmic mucin. It is a tubular neuroendocrine tumor, she said. “Make sure you distinguish this from goblet cell adenocarcinomas, which have a lot of mucin.”
Tubular neuroendocrine tumor, previously known as tubular carcinoid, is a rare subtype, Dr. Pezhouh said. Its clues: salt-and-pepper chromatin, lined by a single layer of uniform cells, true glands with intracytoplasmic mucin not seen, and a negative mucicarmine stain. In Fig. 29 the tubular neuroendocrine tumor is on left and the goblet cell adenocarcinoma on right.
Amy Carpenter Aquino is CAP TODAY senior editor. Images are courtesy of Drs. Pezhouh and Birkness-Gartman, as well as Ezra Baraban, MD, and Lysandra Voltaggio, MD. Dr. Birkness-Gartman’s images published in the preceding article appear in the Johns Hopkins Pathology Atlas iPad app on appendiceal pathology, released in 2022 (https://apps.pathology.jhu.edu/all). Dr. Birkness-Gartman is first author on the app; Toby Cornish, MD, PhD, Norman Barker, MA, MS, and Ralph Hruban, MD, are series editors.

