Karen Lusky
February 2016—Musician Lauryn Hill has been quoted as saying, “Reality is easy. It’s deception that’s the hard work.” That viewpoint just might resonate with pathologists who sometimes have to diagnose deceiving-looking skin lesions.
In a talk at CAP ’15, Deborah L. Cook, MD, professor of pathology and director of dermatopathology at the University of Vermont, shared several case examples that illustrate that investigative effort. All of them involve non-melanocytic malignancies known to mimic benign entities and the converse—“the proverbial wolf in sheep’s clothing” and “sheep in wolf’s clothing,” as she puts it.
In one case, a woman in her 50s developed a firm, mildly tender nodule on her flank. The clinician thought the nodule was a cyst or dermatofibroma. At low magnification, it did look like a dermatofibroma, particularly because the clinician had suggested a diagnosis of dermatofibroma, Dr. Cook says. The growth pattern showed “small cells percolating through the dermis of the skin.” At higher magnification, however, the pattern was fairly typical of lobular breast cancer.
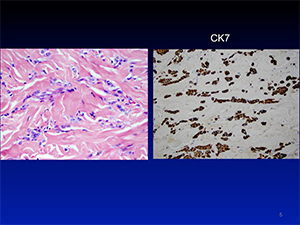 Immunohistochemistry testing helped confirm the diagnosis. “CK7 [cytokeratin 7] was positive. It isn’t specific for breast cancer but it is for epithelial cells. A dermatofibroma would not stain with CK7,” she says. “This showed it was at least epithelial and then there was the pattern, and we did do additional studies that at least suggested breast cancer.”
Immunohistochemistry testing helped confirm the diagnosis. “CK7 [cytokeratin 7] was positive. It isn’t specific for breast cancer but it is for epithelial cells. A dermatofibroma would not stain with CK7,” she says. “This showed it was at least epithelial and then there was the pattern, and we did do additional studies that at least suggested breast cancer.”
They also subsequently found out that the patient had a history of breast cancer. “The use of electronic health records allows us to more easily access our patients’ previous history that we have here so we had that information,” she says.
Dr. Cook notes that it’s not that common for a skin metastasis to be the first sign of breast cancer. What’s probably more common, she adds, is to initially diagnose an internal malignancy, such as renal cell carcinoma, in a skin metastasis, which usually presents as a firm nodule. “Renal cell carcinoma can sometimes be clinically silent in the kidney itself but metastasize, often to the skin.”
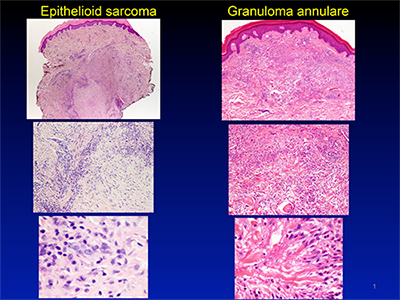
In another case, a male in his early 20s had a small, slowly growing, asymptomatic pink nodule on his dorsal wrist. At low to medium magnification, the nodule looked similar to granuloma annulare, Dr. Cook says. In this common and benign inflammatory process, the cells consist of macrophages or histiocytes and have a rather palisaded appearance, she explains. When viewed at a higher magnification, however, the cells no longer resembled histiocytes. “They looked more atypical.”
“What really cinched it,” she says, is IHC testing that showed cytokeratin positivity. “The histiocytes in granuloma annulare would be CD68 or CD163 positive and cytokeratin negative.”
The diagnosis? Epithelioid sarcoma, which Dr. Cook notes also has a palisaded appearance under the microscope. The malignancy is aggressive, she says, but “early detection does help.”
An epithelioid sarcoma isn’t a distinctive lesion clinically, Dr. Cook cautions. There were two clues: the patient’s age, as the cancer affects young adults, and the tumor’s location on a distal extremity where it typically occurs.
Another case involved what was likely a long-standing cancer. A middle-aged man sought evaluation for a 20 × 15 cm violaceous, fixed, firm growth that had been on his back for several years. It was enlarging and becoming more painful. A limited needle core biopsy was reported as “CD34-positive, spindle cell proliferation, favor solitary fibrous tumor; no features of malignancy seen.” The man had the mass removed when an MRI showed that it was large and growing.
 Dr. Cook says she and colleagues had a broad differential that included neurofibroma owing to its myxoid background and bland spindle cell morphology. So they used an S-100 protein immunostain, which helps distinguish “tumors of neural crest origin, such as peripheral nerve sheath tumor and melanoma,” she says. The S-100 protein was used “more to rule out a nerve sheath neurofibroma, which isn’t a malignancy.” S-100 protein was negative.
Dr. Cook says she and colleagues had a broad differential that included neurofibroma owing to its myxoid background and bland spindle cell morphology. So they used an S-100 protein immunostain, which helps distinguish “tumors of neural crest origin, such as peripheral nerve sheath tumor and melanoma,” she says. The S-100 protein was used “more to rule out a nerve sheath neurofibroma, which isn’t a malignancy.” S-100 protein was negative.
CD34 was positive, however, which would be expected to be seen in a dermatofibrosarcoma protuberans (DFSP), which is malignant, Dr. Cook says. “[CD34] marks progenitor cells but it’s also positive in a variety of lesions so you really have to interpret it with morphology. It is also positive in stem cells in the bone marrow and normal cells too.”
Dr. Cook says there was another key to cracking the case: Although the patient had the myxoid variant of DFSP, which is unusual, they found focal areas in the tumor that were more classic for the cancer. In addition, cytogenetic testing showed the tumor had an abnormal karyotype that is seen in DFSP.
Why didn’t the needle core biopsy pick up indications of malignancy? “Unfortunately, this particular malignancy is made up of very bland cells,” Dr. Cook says. “So if you just look at the cells in a limited specimen or in cytology needle aspirate, it will appear very bland and won’t have typical features of malignancy. But then when you look at a tissue section and see the growth pattern and other features, it’s malignant.”
Dr. Cook makes it clear that she’s not criticizing the decision to start with the needle core biopsy. The case in question included numerous entities in the differential diagnosis, she says, “and needle core biopsy is an excellent first diagnostic tool to try to eliminate [those] and direct further investigational pathways.”
Dr. Cook also discussed two patients who had benign lesions that have the potential to become malignant or may be interspersed with cancerous lesions.
For the first case, Dr. Cook showed a picture of a rather angry-looking, well-circumscribed verrucous keratotic plaque on the shin of a woman in her 60s. The patient said the plaque had been present for several months. The differential diagnosis included squamous cell carcinoma.
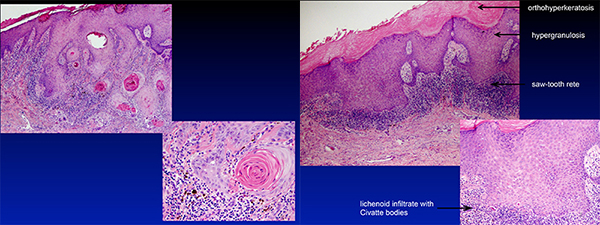
“Both the clinical appearance and the histopathology were alarming, so it was worrisome for cancer,” Dr. Cook says. Upon closer inspection, only part of the lesion’s histology was disturbing. “In the center where it was very thick, it had the kind of mimicking features for cancer, but then when you looked out to the sides away from the thickest area, that’s where you saw the inflammatory changes characteristic of lichen planus.”
The final diagnosis: hypertrophic lichen planus, a benign condition. “The lesions of hypertrophic LP can vary,” she says. “Some are entirely composed of atypical epithelium with lichenoid inflammation. Others, like the case that I showed, have areas with less hyperplasia and atypia that make it somewhat easier to make the diagnosis.”
While the pathology requisition didn’t say so, the patient had pruritic lesions on her entire leg—information that “actually gave more credence to it being lichen planus and not cancer.”
Squamous cell carcinomas and keratoacanthomas can occur in these lesions, she cautions. So patients with the condition would require follow-up to treat the inflammatory process and identify and biopsy lesions with suspicious changes.
In the other case, an African-American woman in her early 30s had a 6.0 × 5.0 cm, non-tender, slowly growing plaque on her left upper arm. Initially the clinical information provided by the clinician just said “indurated plaque.”
Dr. Cook says that even though the plaque was a little more deeply pigmented than the surrounding skin, she didn’t think it was a melanoma due to the patient’s age and the lesion’s location. “In African-Americans, usually melanomas arise on the palms and soles—not that they couldn’t be seen on other areas.” But the most striking clinical feature was the lesion’s firmness.
She did consider that the lesion could be subcutaneous panniculitis-like T-cell lymphoma, but testing for T-cell gene rearrangements was negative. Instead, the patient had lupus panniculitis, a form of lupus. She, in fact, had a lupus diagnosis, although Dr. Cook says she doesn’t think they had that information initially. They didn’t know up front that the patient had other skin lesions, as well as ones involving her scalp with alopecia.
 “There have been two fairly recent papers showing that some patients [with lupus panniculitis and lymphoma] overlap. So it may not be two distinct entities but on a spectrum.” That theory is still evolving, she notes, with one of the papers published in 2015 (Bosisio F, et al. Am J Surg Pathol. 2015;39[2]:206–211).
“There have been two fairly recent papers showing that some patients [with lupus panniculitis and lymphoma] overlap. So it may not be two distinct entities but on a spectrum.” That theory is still evolving, she notes, with one of the papers published in 2015 (Bosisio F, et al. Am J Surg Pathol. 2015;39[2]:206–211).
Thus, for patients who have the differential of lupus panniculitis versus lymphoma, “it’s important to look at various areas of a single lesion, sample multiple lesions if present, and follow the patient over time with biopsy of lesions showing progression.”
Some malignancies mimic each other. In one case, a 66-year-old woman had what the requisition said was a new pigmented lesion under her arm. The clinician thought it looked like an atypical nevus and wanted to rule out melanoma.
The lesion was, however, pigmented Paget’s disease, “which is basically an adenocarcinoma,” Dr. Cook says. Paget’s disease is separated into two forms: mammary and extra-mammary, with the former occurring on or near the breast. “The extra-mammary tends to be more on the genitals predominantly,” she says, and is also adenocarcinoma.
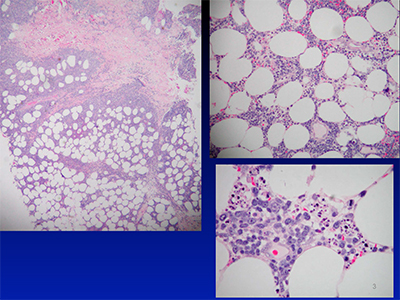 Thus, the location of the patient’s lesion in the axillary area was a clue of sorts, Dr. Cook says. “With the H&E, the nested pattern was a little bit different than what melanoma usually shows, and the cell cytology of the cell areas that were pigmented was more suggestive of Paget’s. The cells had more voluminous wispy cytoplasm and vesicular nuclei.” There were also coarse melanin granules within the cytoplasm of some of the Paget’s cells.
Thus, the location of the patient’s lesion in the axillary area was a clue of sorts, Dr. Cook says. “With the H&E, the nested pattern was a little bit different than what melanoma usually shows, and the cell cytology of the cell areas that were pigmented was more suggestive of Paget’s. The cells had more voluminous wispy cytoplasm and vesicular nuclei.” There were also coarse melanin granules within the cytoplasm of some of the Paget’s cells.
She says that generally in cases with this differential, they perform a panel of immunohistochemistry stains, including adenocarcinoma markers (CK7, CEA, GCFDP-15), melanoma markers (S-100 protein, Mart-1, or HMB45), and possibly p63 for Bowen’s disease (SCC in situ).
The opposite can occur where a melanoma imitates a non-melanocytic lesion. In an interview with CAP TODAY, Clay Cockerell, MD, of Cockerell Dermatopathology Laboratory in Dallas, said he recalls a case that looked almost exactly like a neurofibroma. “It didn’t look anything like a melanoma clinically and looked totally benign under the microscope.” Nevertheless, it was desmoplastic melanoma that became evident when it recurred, says Dr. Cockerell, who is a clinical professor of dermatology and pathology and director of dermatopathology, University of Texas Southwestern Medical Center.
“Desmoplastic melanoma is one of the harder things we have to evaluate in dermatopathology as the cells may be very bland in appearance with minimal atypia, and in some cases there is only minimal, if any, epidermal involvement, which can lead to under-diagnosis. Most clinicians are not aware of the diagnosis, either,” he says.
Dr. Cockerell (who wasn’t a presenter) recalls a case he shared often during teaching conferences: A patient who had a lesion biopsied that was initially diagnosed as squamous cell carcinoma. “It turned out that the individual actually had cutaneous leishmaniasis, a rare parasitic infection that may produce a skin lesion that clinically mimics squamous cell carcinoma,” he says. The infection is usually treated with oral medication.
Dr. Cook says it’s important for clinicians to know about a potential pitfall “and say that what we diagnosed didn’t really fit, and give us a little more clinical information.”
Pathologists who don’t have access to patients’ electronic health records have to decide on a case-by-case basis when to contact the clinician for more information, she adds.
Lori Lowe, MD, professor of dermatology and pathology at the University of Michigan, in an interview said she knows pathologists who would prefer making an objective diagnosis without being influenced by the clinical scenario. “This would be very difficult to do in dermatopathology, as so much depends on clinical-pathologic correlation. The age of the patient, for example, is hugely important when considering a histopathologic diagnosis.
“My gut is that it is very easy, even for dermatopathologists, to overcall hypertrophic lichen planus as squamous cell carcinoma if they don’t have sufficient clinical information,” she says. “It’s also important to know if a patient is immunosuppressed because the differential diagnosis of many skin lesions needs to be expanded to include opportunistic infections.”
For a melanocytic lesion, Dr. Lowe thinks pathologists and dermatologists should want to know the size and whether it’s asymmetrical or has an irregular border, has changed in size or color, is itching or bleeding. “Most of the time, we think of pigmented lesions being brown but melanomas can have shades of brown and/or black, or they can be red and white because of inflammation or regression, respectively. Even something as simple as itching is a really common sign of melanoma.” Knowing that a patient has a family history of melanoma is also helpful.
“It’s always helpful to know if the patient has a past history of melanoma or any other malignancy,” she stresses, noting that the scalp is a common place for metastases from an internal malignancy to appear. Knowing that a patient had received radiation would, of course, be important because some tumors, such as malignant fibrous histiocytoma or angiosarcoma, are more likely to arise in irradiated sites, she says.
“It’s nice if you can get a whole picture of the patient rather than just the bump or lump,” Dr. Lowe says. “It helps paint the picture and make the story make sense.”
[hr]
Karen Lusky is a writer in Brentwood, Tenn.

