Editors: Olga Pozdnyakova, MD, PhD, Geoffrey Wool, MD, PhD, David Bernard, MD, PhD & Raul S. Gonzalez, MD
Submit your pathology-related question for reply by appropriate medical consultants. CAP TODAY will make every effort to answer all relevant questions. However, those questions that are not of general interest may not receive a reply. For your question to be considered, you must include your name and address; this information will be omitted if your question is published in CAP TODAY. Submit a question.
Q. When should plasma mixing tests be performed in the coagulation laboratory, and how are they best interpreted?
A. March 2025—Plasma mixing studies, also known as mixing tests or inhibitor screens, can be used to help determine if a prolonged clotting time or decreased activity assay is due to a factor deficiency or inhibitor. They are also used in the algorithm to detect a lupus anticoagulant and to screen for an inhibitor in hereditary or acquired factor deficiencies.
Mixing studies are typically conducted by combining equal portions of patient plasma with normal pooled plasma (NPP) and then immediately performing the clotting time or activity assay. Typically, correction into the normal reference range or to a predetermined cut-off indicates a factor deficiency, while lack of correction, or not meeting a predetermined cut-off, indicates an inhibitor. Mixing tests are of no clinical value when baseline clotting time or activity is normal. They should not be performed when the patient sample contains anticoagulant drugs or if the patient has received fresh frozen plasma, cryoprecipitate, factor replacement therapy, Feiba (anti-inhibitor coagulant complex), or emicizumab. It is acceptable to vary the ratio of patient plasma to NPP (i.e. four parts patient plasma to one part NPP) or incubate the mix at 37°C for one to two hours, although the latter is only recommended with activated partial thromboplastin time (APTT) and not prothrombin time (PT) mixing tests.
Published studies of mixing tests are limited as the studies often use only one instrument/reagent platform or limited sample types (i.e. only lupus anticoagulants). In 2023, results of a small field study that evaluated multiple instrument/reagent manufacturers, varied sample types, and 11 mixing results interpretation methods were published.1 This study as well as expert opinion and good laboratory practice were used to draft a 2024 International Council for Standardization in Haematology guidance document on the performance and interpretation of APTT and PT mixing tests.2 All ICSH guidance documents and standards, including those on hematology and hemostasis, are open access and available through the ICSH website, icsh.org.
There is no one mixing test interpretation method that can consistently differentiate a factor deficiency from an inhibitor with 100 percent certainty (Table). In the aforementioned field study, subtraction and ratio methods tended to have high degrees of inaccuracy. In general, subtraction and ratio methods that perform well in detecting a factor deficiency perform poorly for detecting inhibitors and vice versa. The field study results demonstrated that calculation methods, either percent correction (a greater than 70 percent threshold was used to demonstrate correction) or the Rosner index (a value less than 15 percent was used to demonstrate correction), performed better than any subtraction or ratio method alone. As no single interpretation method is 100 percent accurate, an algorithmic approach for mixing study result interpretation using one of the two calculation methods followed by an NPP subtraction method (test result – NPP result [correction is defined as less than zero]) should be considered.
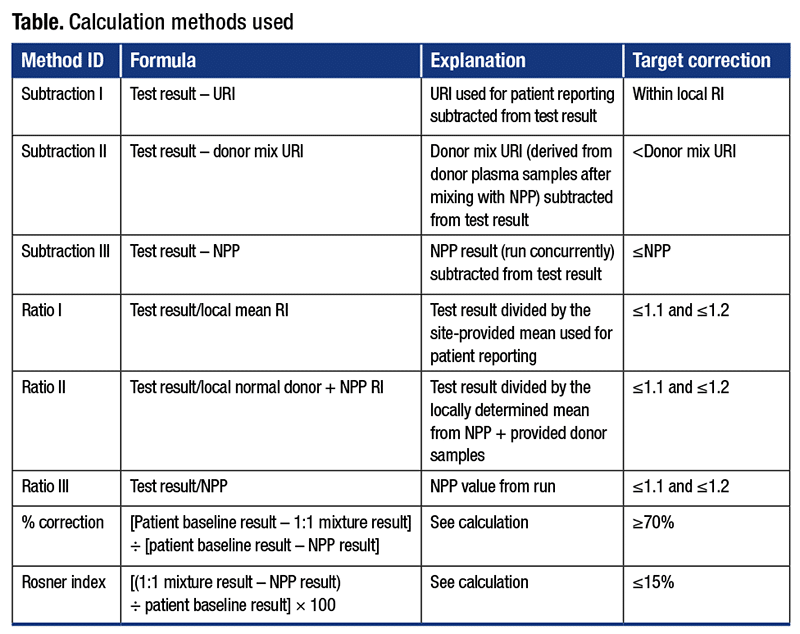
Laboratories can choose their preferred mixing test interpretation method but only if they validate the method used. Laboratories should also verify or validate their own thresholds for calculation methods.
- Gosselin RC, Moore GW, Kershaw GW, Montalvão S, Adcock DM. International Council for Standardization in Haematology field study evaluating optimal interpretation methods for activated partial thromboplastin time and prothrombin time mixing studies. Arch Pathol Lab Med. 2024;148(8):880–889.
- Adcock DM, Moore GW, Kershaw GW, Montalvão SAL, Gosselin RC. International Council for Standardization in Haematology (ICSH) recommendations for the performance and interpretation of activated partial thromboplastin time and prothrombin time mixing tests. Int J Lab Hematol. 2024;46(5):777–788.
Dorothy M. Adcock, MD
Chief Medical Officer, Labcorp, Retired
Q. How important is it for a laboratory to perform whole mount specimen collection and examination?
A. Although not used routinely, this novel method can be used for serial pathologic sectioning to help determine margin status and optimize cross-sectional visualization of whole resected specimens. The technique is commonly used for radical prostatectomies and involves placing a full cross-section of a specimen on a large slide. This method improves examination and shows the architecture of the gland, and it can associate molecular imaging, ultrasound detection, and functional MRI imaging with the resected specimen.
Whole mount processing requires special techniques and equipment and does not typically fit into routine daily histopathology. It is used more readily in research and academic institutions.
Cimadamore A, Cheng L, Lopez-Beltran A, et al. Added clinical value of whole-mount histopathology of radical prostatectomy specimens: a collaborative review. Eur Urol Oncol. 2021;4(4):558–569.
Duan L, Liu Z, Wan F, Dai B. Advantage of whole-mount histopathology in prostate cancer: current applications and future prospects. BMC Cancer. 2024;24:448.
Elizabeth Sheppard, MBA, HT(ASCP)
Global Pricing and Market Access Director
Oncology Diagnostics
AstraZeneca
Gaithersburg, Md.
Vice Chair, CAP/NSH Histotechnology Committee

