Editor: Frederick L. Kiechle, MD, PhD
Submit your pathology-related question for reply by appropriate medical consultants. CAP TODAY will make every effort to answer all relevant questions. However, those questions that are not of general interest may not receive a reply. For your question to be considered, you must include your name and address; this information will be omitted if your question is published in CAP TODAY.
[button size=”small” link=”http://www.captodayonline.com/q-a-submission/”]Submit a Question[/button]
Q. Due to an ever-changing workforce, many new and inexperienced technologists are working in the microbiology lab and appear to be having difficulty interpreting cultures and troubleshooting when an organism in question may not be significant. As an example, a scant growth of Micrococcus was isolated and reported from a cerebrospinal fluid culture; it was not seen in the Gram stain and was negative for leukocytes. Contaminants had been noted on some of the media plates at this time as well, but many of these inexperienced technologists do not have the confidence to ignore obvious contaminants or suggest the possibility of contamination. Is there some guidance or troubleshooting tools for these situations?
A. This is indeed a significant matter that we are all facing with our seasoned technologists retiring and new, less experienced personnel coming into the laboratory. The determination of contamination is something that must be taught and learned by experience, which is how we all learned it. The importance of using prominent examples of such situations to train new members of the laboratory team cannot be overstated. Teaching what is usual resident flora and what are considered contaminants under normal situations must also be included when onboarding new staff members. Many of our common textbooks1 have chapters on what is usual resident flora and those that are often considered contaminants in normally sterile specimen sources. Again, this must be emphasized in the training of newly minted microbiology technologists. Less experienced technologists should also be encouraged to ask a more senior coworker if they have suspicions of a contaminating organism; this will help them recognize these situations going forward. There is much we can initially teach our new microbiologists, but, as has always been true, experience is the best teacher.
- Sharp S, Loeffelholz M. The Human Microbiome. In: Jorgensen JH, Pfaller MA, Carroll KC, et al., eds. Manual of Clinical Microbiology. 11th ed. Washington, DC: ASM Press; 2015:226–237.
Susan E. Sharp, PhD, Director of Microbiology, Kaiser Permanente, Northwest Region Laboratory, Portland, Ore., Member, Microbiology Resource Committee
[hr]
Q. What is the latest recommendation for lot-to-lot quality control crossover? Some sources recommend a 20 points/20 days correlation and some say a minimum of 10 points/10 days.
A. The 2016 revision of CLSI document C24, “Statistical Quality Control for Quantitative Measurement Procedures: Principles and Definitions,” provides excellent guidance on this point. When changing to a new lot of QC material, assuming there is no change in the measurement procedure, the target value for the new QC lot should be established using a minimum of 10 measurements on 10 different days. Ten days allows some day-to-day sources of variability to be represented in the mean value. It is important to recognize that other sources of variability, such as recalibrations and maintenance procedures, may not have occurred in the 10 days. Consequently, it is recommended that the cumulative mean value be monitored for a few weeks to months and that the target value of the QC material be adjusted, if needed, to better reflect most sources of variability that can influence the mean value.
The standard deviation for the previous lot of QC material should have been established as a cumulative value over a long period of use and thus properly represents most sources of variability in the measurement procedure that are likely to be encountered. Since the precision of a measurement procedure rarely changes with a new lot of QC material, the SD already established for the measurement procedure at the approximate concentration of the new lot of QC material can be used for the new lot. If the new QC material has a very different concentration than the preceding lot, then a different SD may be needed because the magnitude of the standard deviation can vary with concentration. In this situation, the coefficient of variation can be used to estimate the SD at the concentration of the new lot of QC material when both concentrations are within the concentration interval over which the CV is approximately constant.
It is also important to recognize that reagent lot changes can require a change in the target value for a QC material. It is well understood that the magnitude of the non-commutability bias (also called matrix bias) for a typical non-commutable QC material is frequently different for different reagent lots (Miller WG, et al. Clin Chem. 2011;57[1]:76–83).Consequently, a shift in QC values following a reagent lot change cannot be assumed to represent a defect in the new reagent nor that erroneous results for patient specimens will be produced. The recommended approach for reagent lot changes is to verify that the new reagent lot gives consistent results for patient specimens between the old and new reagent lots using, for example, an approach such as that in CLSI document EP26-A, “User Evaluation of Between-Reagent Lot Variation.” Once the results for patient specimens verify the suitability of the new reagent lot, the QC target value is adjusted to reflect any shift in values that may have occurred when using the new reagent lot.
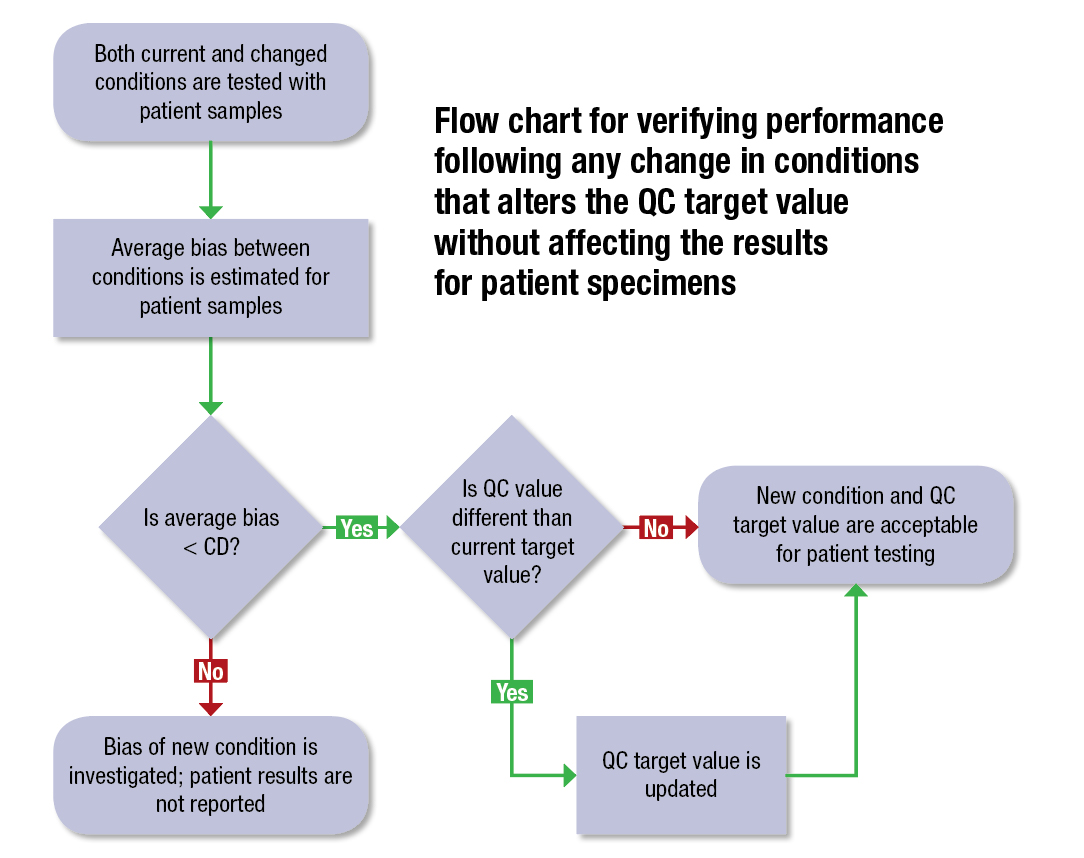
A diagram from CLSI C24 illustrates the recommended approach for assessing QC target values following a change in conditions that does not affect patient specimen results. A change in conditions could be a new reagent lot, a component replacement, maintenance, or other procedure that may affect the QC differently than it affects the patient specimen results.
- Clinical and Laboratory Standards Institute. C24: Statistical Quality Control for Quantitative Measurement Procedures: Principles and Definitions, 4th ed.; 2016.
- Miller WG, Erek A, Cunningham TD, Oladipo O, Scott MG, Johnson RE. Commutability limitations influence quality control results with different reagent lots. Clin Chem. 2011;57(1):76–83.
- Clinical and Laboratory Standards Institute. EP26-A: User Evaluation of Between-Reagent Lot Variation, 1st ed.; 2013.
Greg Miller, PhD, Professor of Pathology, Director of Clinical Chemistry, Director of Pathology
Information Systems, Virginia Commonwealth, University Health System, Member, CAP Accuracy, Based Testing Committee
[hr]
Dr. Kiechle is a consultant, clinical pathology, Cooper City, Fla. Use the reader service card to submit your inquiries, or address them to Sherrie Rice, CAP TODAY, 325 Waukegan Road, Northfield, IL 60093; srice@cap.org. Those questions that are of general interest will be answered.

