Charna Albert
February 2022—How to classify gastric intestinal metaplasia, when to classify it, and the implications of a GIM diagnosis were the focus of a CAP21 presentation in a session on advances in gastric neoplasms.
The big question, said presenter Namrata Setia, MD, associate professor of pathology, University of Chicago School of Medicine, is, “Why are we suddenly talking about classifying intestinal metaplasia in the stomach? I haven’t gotten a call about this topic until recently.” The reason, she said, is in 2020, the American Gastroenterological Association published the first U.S.-based clinical practice guidelines on the management of GIM (Gupta S, et al. Gastroenterology. 2020;158[3]:693–702).
According to the literature, among patients with GIM who had biopsies taken from both the gastric antrum/incisura and body, extensive GIM versus limited involvement (involvement of at least the gastric body versus GIM of the antrum and/or incisura, respectively) was associated with a twofold higher pooled relative risk of incident gastric cancer (RR, 2.07; 95 percent CI: 0.97– 4.42). Having incomplete versus complete GIM was associated with a threefold increased risk for incident gastric cancer on follow-up (RR, 3.33; 95 percent CI: 1.96 –5.64), and having a family history of a first-degree relative with gastric cancer was associated with a 4.5-fold increased risk for gastric cancer among patients with GIM (RR, 4.53; 95 percent CI: 1.33 –15.46).
But notably, Dr. Setia said, “none of the studies used to calculate the provided relative risks were from the United States, indicating the lack of data from the United States. So the guidelines’ coauthors published another paper in the same edition of Gastroenterology urging pathologists to classify intestinal metaplasia, and hence the recent uptick in classification requests” (Shah SC, et al. Gastroenterology. 2020;158[3]:745 –750). In their guidelines, Gupta, et al., write, “Anecdotally, U.S. pathologists rarely report presence of incomplete vs complete GIM as part of routine GIM diagnosis,” which “raises concerns as to whether the histologic subtype of GIM can be feasibly utilized as part of risk stratification in the United States without a substantial educational initiative for pathologists.”
Helicobacter pylori-associated GIM, Dr. Setia said, originates at the incisura, involves the antrum, and then spreads to the corpus. “But it doesn’t have to involve the entire antrum before extending to the corpus.” Thus, to classify GIM histologically, the extent, or number of sites involved in the stomach, and the grade, or severity of GIM involvement of the biopsied fragments, should both be measured. The subtype, an additional histologic classification for GIM, is based on distinct microscopic appearances. “For the purpose of using intestinal metaplasia to identify high-risk individuals, the AGA guidelines recommend using the extent and subtype.”
In determining whether the extent of GIM is limited or extensive, the antrum and incisura are considered one site; the corpus, including the body mucosa, is considered another site. The European guidelines (published in 2012 and revised in 2019) classify GIM as limited if one site—antrum/incisura or corpus—is involved, and as extensive if both sites are involved (Pimentel-Nunes P, et al. Endoscopy. 2019;51[4]:365 –388). The AGA takes a different approach. GIM is considered limited if the antrum/incisura alone is involved. “But as soon as you see intestinal metaplasia in the corpus, even if you don’t see it in the biopsies from the antrum/incisura, it’s classified as extensive,” Dr. Setia said. “And their rationale is intestinal metaplasia may not form a visible lesion and can easily be missed in endoscopy.” So intestinal metaplasia seen only in the body but not in the antrum, according to the AGA guidelines, doesn’t mean it’s absent, she said. “They presume it was there but not sampled.”
Dr. Setia presented two sets of gastric corpus and antral biopsies: one obtained from a 66-year-old, in which GIM could be seen in the corpus and in the antrum at multiple foci, and one from a 72-year-old, in which GIM could be seen in the corpus but not the antrum. The former would be considered extensive by both the AGA and European guidelines (known as MAPSII), she said, as both sites are involved. But the latter would be considered limited by the MAPSII guidelines and extensive by the AGA guidelines. For the AGA, the absence of GIM in the antral biopsies could indicate it was present but not sampled. “The other possibility is the patient may have autoimmune gastritis,” she noted. The MAPSII guidelines recommend performing an endoscopy every three to five years to surveil patients with autoimmune gastritis. But the AGA guidelines do not make recommendations regarding autoimmune gastritis, she said, because of the lack of definitive evidence or data from the U.S. supporting surveillance.
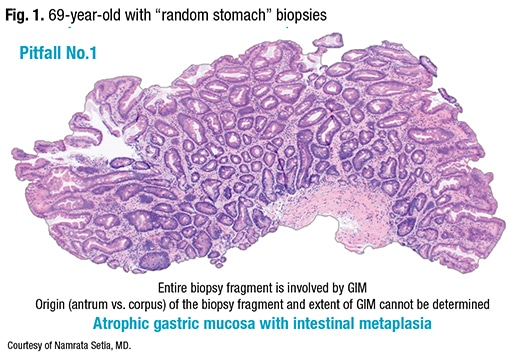
[dropcap]D[/dropcap]r. Setia warned of two significant pitfalls, the first of which is “random stomach” biopsies (total number of biopsies not specified, biopsies not separated and labeled by location). In Fig. 1, “The entire fragment is involved by gastric intestinal metaplasia. One can guess but never be sure whether this biopsy was from the antrum or corpus.” This case, she said, would be signed out as atrophic gastric mucosa with intestinal metaplasia. “You’re unable to determine the extent using this fragment, which is completely involved by intestinal metaplasia. The important point is we should discourage our clinicians from sending random biopsies if we’re expected to classify intestinal metaplasia.”
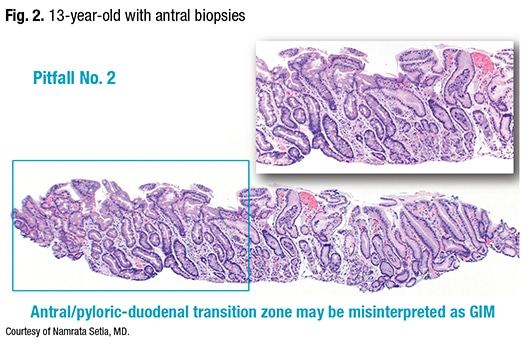
The second pitfall: the antral/pyloric-duodenal transition zone may be misinterpreted as GIM. In antral biopsies obtained from a 13-year-old (Fig. 2), “we see the zone of intestinal glands,” Dr. Setia said, “but otherwise this child had pristine GI biopsies, so these glands clearly represent the pyloric-duodenal junction. The point from this case is one has to be careful not to misinterpret the pyloric-duodenal junction as antral intestinal metaplasia.”
Although the AGA does not recommend using the grade to determine GIM’s risk of progression, Dr. Setia said, grading may be used in routine practice. In grading, each biopsy fragment is given an estimated decile score for percentage involvement by GIM, and subsequently, each site is given an average score for percentage involvement. “It is then classified as mild GIM if the percentage is between one and 30 percent, moderate if it is between 31 and 60 percent, and severe if it is more than 60.” But, she said, there’s a bit of confusion on this: “I’ve seen the term ‘focal intestinal metaplasia’ used for mild GIM, and ‘extensive’ used for severe.” She admits “extensive” sounds better than “severe” intestinal metaplasia, but according to the AGA guidelines, the term extensive should be used only for extent, not severity.
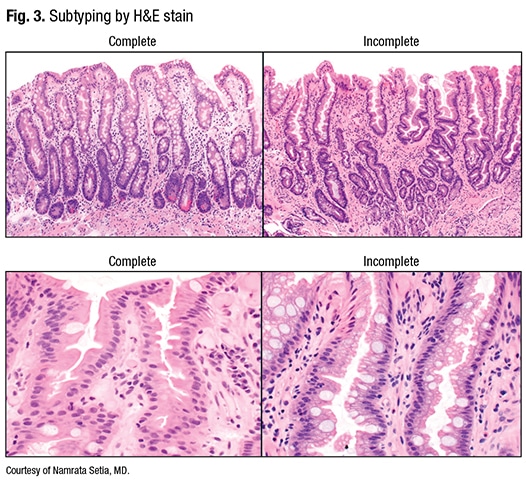
GIM subtyping, Dr. Setia said, can be performed on H&E slides, by using special stains (high-iron diamine), or with mucin IHC stains. H&E subtyping classifies GIM as complete or incomplete: “If you’re able to see the brush border and Paneth cells in the small intestinal mucosa, then the intestinal metaplasia is complete,” Dr. Setia said. “But if the intestinal metaplasia does not have a brush border or Paneth cells, it is incomplete.” The incomplete type, she said, has a distinctive hybrid epithelium, with goblet cells interspersed within the gastric foveolar cells (Fig. 3).
The well-documented special stain used for GIM subtyping—i.e. high-iron diamine/Alcian blue stain—is not used routinely because of the toxicity of the reagents. The stain divides GIM into types I, II, and III, with type I corresponding to complete GIM and types II and III corresponding to incomplete GIM. Type III has the highest reported association with progression to gastric cancer, she said. “This type is characterized by its sulphomucin content, i.e. colon-type mucin, which stains the goblet cells and the background columnar cells brown.”
H&E may be the best stain for subtyping, she said, but the issue with H&E subtyping is that GIM is rarely fully complete or fully incomplete. “Mostly it’s composed of different proportions of complete and incomplete, and, yes, you can report it as ‘mixed predominantly complete’ or ‘mixed predominantly incomplete,’” for example, “but the cutoffs for the mixed types are arbitrary at this point.”
[dropcap]A[/dropcap]dequate sampling is a major prerequisite for classifying GIM, Dr. Setia said. “And that is incumbent on your gastroenterologist to provide when they ask you for intestinal metaplasia classification.” The AGA and MAPSII guidelines again differ in their recommendations, with the AGA recommending the Sydney system five-biopsy protocol, and the MAPSII guidelines a four-biopsy protocol. The Sydney protocol indicates one biopsy from the incisura, two from the antrum—one from the lesser and one from the greater curvature—and two from the corpus (also from lesser and greater curvature). “While it seems a bit much, I’ve noticed that most of our gastroenterologists follow the Sydney protocol. So it’s not impossible to expect adequate sampling from our gastroenterologists.” This reflects the University of Chicago experience, she tells CAP TODAY, not necessarily the pattern in the community.
The MAPSII-recommended protocol involves obtaining two antral biopsies from the lesser and greater curvature, and two from the corpus, also from the lesser and greater curvature, and it’s in the 2019 version of the guidelines, not the 2012 version. “The change in recommendation was based on a large study of around 4,000 biopsies, where they found the yield of the four-biopsy protocol to be the same as the five-biopsy protocol,” Dr. Setia explained.
How should GIM be surveilled? It depends, Dr. Setia said, on whether sampling was adequate and if the patient is at high risk clinically or pathologically. By the AGA guidelines, the groups with higher risk clinically are racial and ethnic minorities with a known high gastric cancer incidence, immigrants from regions with a high incidence of gastric cancer, and those with a first-degree relative who has had gastric cancer. Pathologically, extensive or incomplete-type GIM is considered high risk.
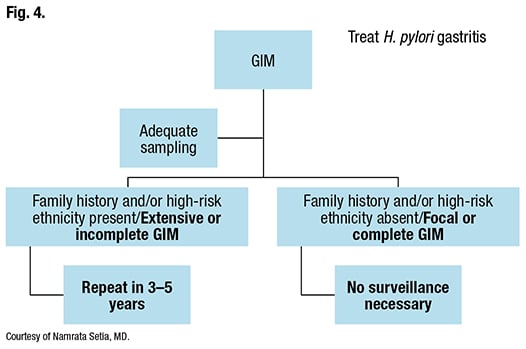
Dr. Setia provided an example of how a patient with GIM should be surveilled and stratified, according to the AGA guidelines. “You see intestinal metaplasia in a patient’s antral biopsies, but because the body mucosa wasn’t sampled, sampling was inadequate. You sign it out as atrophic antral gastritis with intestinal metaplasia.” A gastroenterologist calls to ask what should be done next. Ask the gastroenterologist if the patient is at high risk for gastric cancer per the AGA guidelines, she said. If so, the AGA recommendation is to perform a repeat endoscopy within a year if the patient elects to do so, “to ensure you have an optimal baseline with adequate sampling.”
If sampling is adequate and the patient has high-risk clinical or pathologic features, the AGA’s conditional recommendation (low-quality evidence) is to perform a repeat upper endoscopy with careful mucosal visualization in three to five years, if the patient elects to do so. Gastric biopsies of the antrum and body and any concerning lesions also should be taken. If no high-risk features are present and the GIM is focal or complete, surveillance is not necessary. H. pylori-associated gastritis, irrespective of sampling and classification, is treated (Fig. 4).
The AGA guidelines emphasize the lack of quality evidence on GIM from the United States. In response, Dr. Setia and colleagues at the University of Chicago are conducting a prospective study to evaluate the risk of gastric cancer among patients with GIM. “I’m hopeful that in the next five years,” she said, “we’ll have meaningful data on intestinal metaplasia follow-up, at least from the Chicagoland area.”
Charna Albert is CAP TODAY associate contributing editor.

