November 2025—Tosoh Bioscience has received FDA 510(k) clearance for its Tosoh Automated Glycohemoglobin Analyzer HLC 723-GR01, intended for monitoring the long-term blood glucose control of people who have diabetes. The GR01 uses Tosoh’s proprietary nonporous ion exchange high performance liquid chromatography column technology to deliver A1c results in 50 seconds. It also detects the most common hemoglobin variants. The system delivers precision with coefficient of variations less than or equal to 1.1 percent and features automatic buffer change, a compact footprint, and walkaway operation.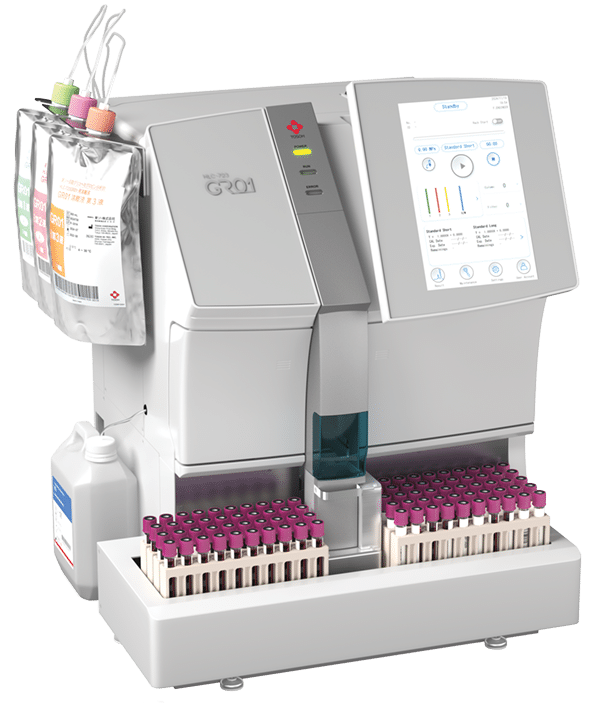
Tosoh Bioscience, 800-248-6764

