Key insights and diagnostic strategies
Elena Enbom, MD, PhD
Xiaoying Liu, MD
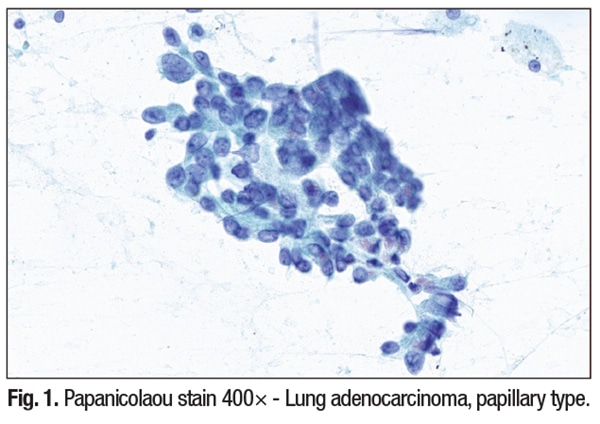
August 2025—Case summary. A 52-year-old chronic smoker with a known MLH1 mutation and Lynch syndrome presented with a pleural-based lung lesion. Fine-needle aspiration revealed a malignant neoplasm with papillary architecture, featuring enlarged overlapping nuclei, coarse chromatin, prominent nucleoli, and rare mitotic figures. Given the patient’s clinical background and cytologic findings, a broad differential diagnosis including primary and metastatic tumors from both thoracic and ab-dominal origins was considered.
Ancillary immunohistochemical studies confirmed the lesion as a metastatic adenocarcinoma of endometrial origin. Additional imaging revealed an ovarian mass and an enlarged uterus with a thickened endometrial stripe. Surgical pathology confirmed synchronous FIGO grade 2 endometrioid carcinomas of both the endometrium and ovary.
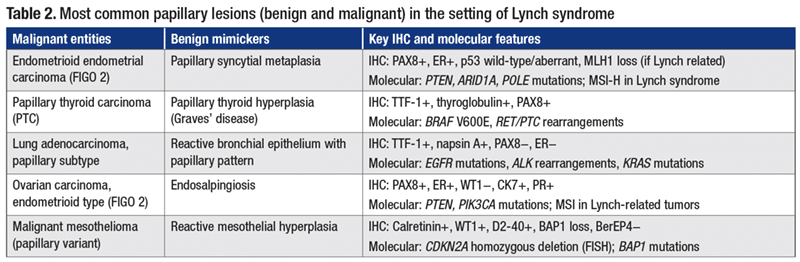
Lynch syndrome: expanding oncologic horizons. Lynch syndrome (hereditary nonpolyposis colorectal cancer, HNPCC) results from germline mutations in DNA mismatch repair genes—MLH1, MSH2, MSH6, and PMS2. While classically linked to colorectal and endometrial carcinomas, its spectrum now includes ovarian carcinoma (endometrioid and clear cell subtypes), papillary thyroid carcinoma, primary pulmonary adenocarcinoma, urothelial carcinoma, and pancreatic neoplasms, particularly intraductal papillary mucinous neoplasms (IPMNs).
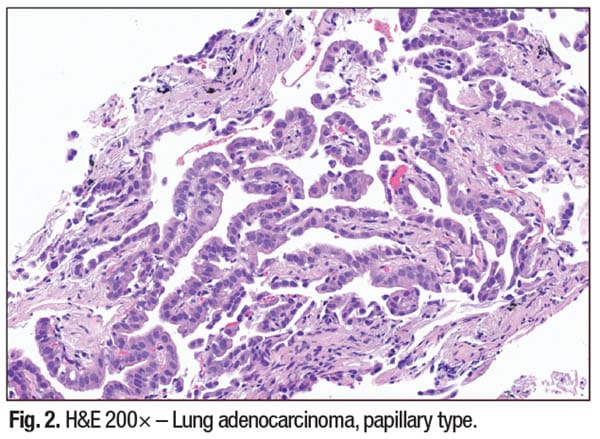
Papillary morphology: patterns and diagnostic challenges. Papillary lesions exhibit epithelial cell clusters with fibrovascular cores and variable atypia. Cytological features such as nuclear grooves, pseudoinclusions, and psammoma bodies may be seen, especially in papillary thyroid carcinoma or serous ovarian tumors. Necrosis and inflammation in the background may indicate higher-grade malignancy and could be a diagnostic clue.
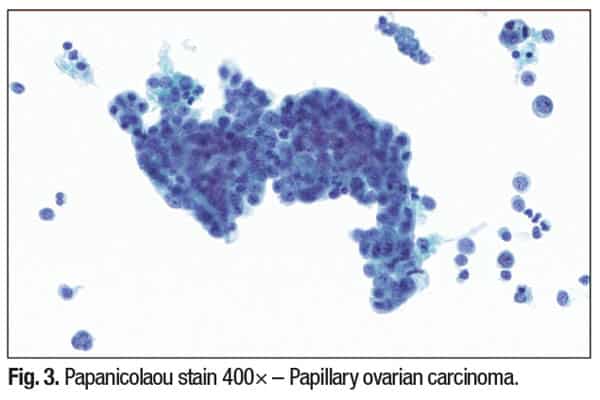
Importantly, papillary architecture is not site-specific, and distinguishing primary from metastatic disease requires integrating patient clinical history, imaging, cytomorphology, IHC profile, and sometimes molecular diagnostics to aid definitive tumor primary prognostication.
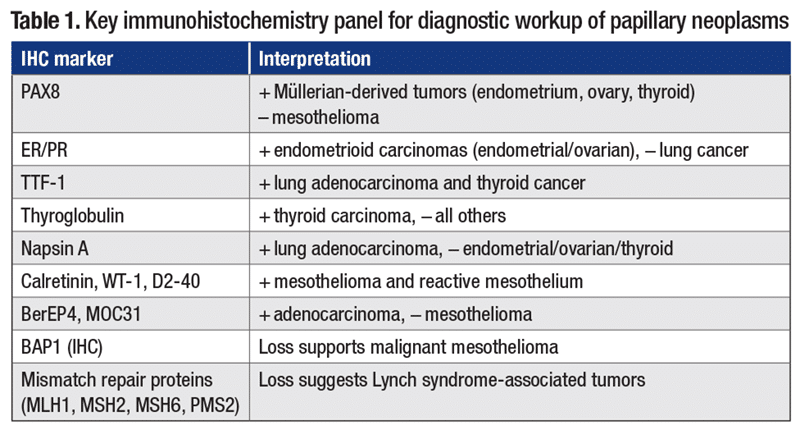
Diagnostic workup and IHC (Table 1). In our case, cytology revealed papillary clusters with stratified nuclei, coarse chromatin, and rare mitoses, raising suspicion for metastatic endometrial carcinoma. This was supported by positive staining for PAX8 and ER, negative for TTF-1, thyroglobulin, and calretinin, along with MLH1 loss on IHC workup. This profile confirmed metastatic endometrioid carcinoma, FIGO grade 2, in a patient with known Lynch syndrome.
Main differential diagnoses are presented in Table 2, including most common malignant entities as well as benign mimickers.
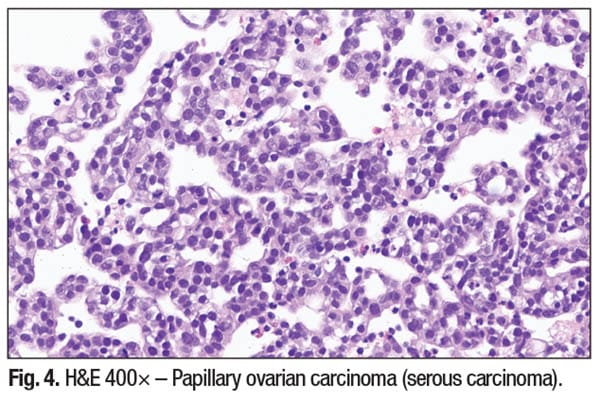
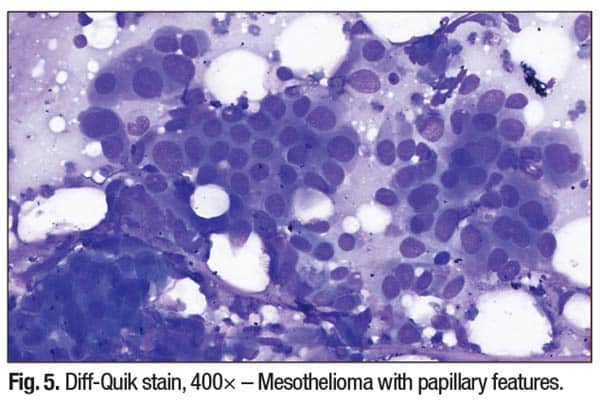
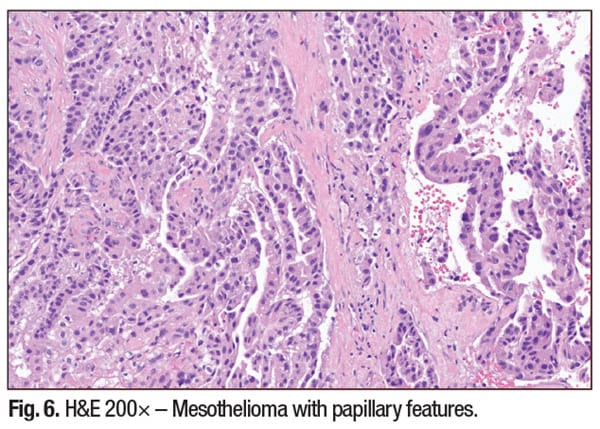
Clinical and diagnostic lessons. Papillary architecture in a neoplasm is not site-specific and can represent a range of benign and malignant lesions. Without appropriate IHC studies and molecular testing, misinterpretation is a significant pitfall. Key points to keep in mind include the fact that Lynch syndrome-associated tumors can present in unusual locations, such as pleural metastases and other distant sites. In pleural-based tumors, PAX8 and ER positivity favors a Müllerian origin over lung primaries. Site-directed molecular testing (e.g. MMR status, POLE, PTEN) further refines the diagnosis and can be helpful in guiding genetic counseling and treatment decisions. Vigilance is needed to avoid mistaking benign papillary mimics (e.g. reactive mesothelium or thyroid hyperplasia) for malignancies. Careful attention to nuclear features, supported by IHC and molecular findings, is critical for accurate diagnosis.
In patients with Lynch syndrome, the diagnostic landscape for papillary neoplasms is becoming broader and more nuanced than ever. Uncommon metastatic sites, such as the pleura, may harbor papillary tumors of gynecologic, thyroid, and pancreatic origin, as well as mesothelioma. Pathologists must maintain a high index of suspicion, deploy comprehensive IHC panels, and incorporate molecular testing to achieve accurate diagnosis. Recognizing these associations ensures the best treatment strategies and enables most appropriate genetic interventions for patients and their families.
This structured approach aims to help cytotechnologists, cytopathologists, and pathologists who handle cytology specimens, and other clinicians, to navigate complex cases with confidence, especially when cytology is the only tissue source available for diagnosis.
Breitenfelder M, Kusch E, Leichsenring M, Prontnicki A. Pulmonary nodular amyloidosis mimicking multiple pulmonary metastases of carcinoma of the corpus uteri. Article in German. Chirurg. 2001;72(9):1062–1066.
Huang YD, Hung YC, Yeh LS, Chiang IP, Zeng GC, Chang WC. Synchronous ovarian endometrioid adenocarcinoma and endocervical mucinous adenocarcinoma. Taiwan J Obstet Gynecol. 2006;45(3):264–267.
Flanagan MR, Jayaraj A, Xiong W, Yeh MM, Raskind WH, Pillarisetty VG. Pancreatic intraductal papillary mucinous neoplasm in a patient with Lynch syndrome. World J Gastroenterol. 2015;21(9):2820–2825.
Pelizzo MR, Pennelli G, Zane M, et al. Papillary thyroid carcinoma (PTC) in Lynch syndrome: report of two cases and discussion on Lynch syndrome behaviour and genetics. Biomed Pharmacother. 2015;74:9–16.
Dr. Enbom is a senior gynecological and breast pathologist and cytopathologist, Quest Diagnostics. Dr. Liu is associate professor of pathology and director of cytopathology, Dartmouth-Hitchcock Medical Center. Drs. Enbom and Liu are members of the CAP Cytopathology Committee.

