Sherrie Rice
May 2025—Fetal RhD prediction by genotyping can prevent unnecessary RhD treatment in some patients and conserve anti-D immunoglobulin. Other countries long ago adopted a targeted antenatal approach to RhD genotyping, and in Canada change is underway.
“We have had, in countries with access to Rh immune globulin, a really effective tool for reducing RhD alloimmunization and therefore hemolytic disease of the fetus and newborn,” said Gwen Clarke, MD, a clinical professor in the Department of Laboratory Medicine and Pathology, University of Alberta. “The success of the prophylactic program in North America until now has to some extent made it a little more difficult to change strategies.”
Nearly all other countries with a significant population of Rh-negative individuals are doing this type of testing, she said. “In North America, we are late to this party.”
Dr. Clarke was a presenter in March with Nancy Robitaille, MD, vice president of transfusion medicine, Héma-Québec, and clinical professor, Department of Pediatrics, University of Montreal, in a CAP TODAY webinar (https://bit.ly/CT_031125-WOD) made possible by a special educational grant from Swedish biotech company Devyser.

The timeline for routine testing and anti-D immune globulin prophylaxis remains what it has been since 1978, as established by clinical trials, said Dr. Clarke, a hematopathologist and transfusion medicine physician with Island Health, Victoria, BC. At about 12 weeks gestational age, an ABO/Rh type and antibody screen are done. In many pregnant patients, the two procedures are repeated at about 24 to 26 weeks to confirm no anti-D has developed in the interim. At 28 weeks all RhD-negative patients receive Rh immune globulin as a matter of routine. After delivery, a targeted dose of RhIG is given to only those who are shown by postnatal cord testing to have an Rh-positive neonate. They are “those at risk for anti-D development because the baby’s cells could immunize them,” Dr. Clarke noted.
Additional doses of RhIG may be given for episodes of potential fetomaternal hemorrhage during pregnancy, such as with an intervention like amniocentesis or in cases of trauma or threatened abortion or vaginal bleeding after 12 weeks.
Targeted use of postpartum RhIG in those long-ago clinical trials was found to reduce the 15 percent alloimmunization rate to about 1.6 percent, Dr. Clarke said. That 1.6 percent rate declines further, to 0.5 to 0.8 percent, when the routine dose is given antenatally at about 28 weeks. Anti-D-related hemolytic disease of the fetus and newborn has not been eliminated owing to RhIG failures and missed or inadequate doses, with the prevalence at about one in 21,000 live births, she said. “Of those, severe anemia occurs in a minority, about 10 percent of those at risk, meaning those where the mom has an anti-D and the fetus is D-positive.”
In high-resource countries where intrauterine transfusion is available, mortality is about 0.5 per thousand live births.
While the postnatal dose is typically targeted, the antenatal dose is not. “It’s given to everybody who’s RhD-negative without an anti-D.”
Genotyping for the RHD gene using noninvasive prenatal testing from cell-free DNA in maternal plasma is the path to even better care, Dr. Clarke said. Dennis Lo, DM, DPhil, and colleagues found in the late ’90s that maternal blood contains cell-free fetal DNA and that it is detectable as early as seven weeks gestation.
Not long after those discoveries, Dr. Clarke said, it was established that genotyping could be used for many disorders, for which it’s in common clinical use. “But for fetal RhD type prediction in particular, it was shown to be as sensitive as postnatal serology as early as 2008.”
“If we could figure out whether the baby was RhD-positive or -negative,” Dr. Clarke said, “we could change the routine prophylactic dose [at 28 weeks] to a targeted dose to only those women who have an RhD-positive baby,” because those who have an RhD-negative baby are not at risk for alloimmunization.
Denmark implemented national fetal RhD genotyping in 2010, the Netherlands in 2011, England in 2013, and Finland in 2014. Regional testing programs were in place in Sweden, Belgium, and France even earlier, in 2009. “All these places have established large-scale programs, and this is just the tip of the iceberg,” Dr. Clarke said, with many other countries testing also.
“And many external quality assessment workshops that compare the results of these various programs demonstrate that, despite different approaches taken to perform the clinical assays, fetal RhD genotyping is a reliable test that can guide the use of targeted Rh immune globulin prophylaxis in a clinical setting,” she said. “It’s available, it works.”
The diagnostic sensitivity of the various assays used in the other countries’ regional and national programs exceeds 99.9 percent, with narrow confidence intervals (Clausen FB. Immunohematology. 2024;40[1]:15–27). The RHD targets range from one to four exons, and testing is done at a variety of gestational ages—some as early as 11 weeks, others as late as 27 to 29 weeks and everywhere in between.
The systematic reviews and meta-analyses of the published studies from the national and regional programs are “very positive” with respect to the test efficacy, she said (Alshehri AA, et al. Transfus Med Rev. 2021;35[2]:85–94; Runkel B, et al. BMC Pregnancy Childbirth. 2020;20:83; Yang H, et al. BMC Med. 2019;17:37; Mackie FL, et al. BJOG. 2017;124[1]:32–46). “They agree the test is safe to use from 11 weeks onward. In other words, there’s enough cell-free DNA with particular assays to carry out this testing from an early gestational age.” They agree, too, that the tests in general have high sensitivity and specificity and that the testing has improved over time, “and that in general these tests are sufficiently accurate for widespread use,” Dr. Clarke said. “So we’re on a solid foundation with respect to introducing this change and a strategy for targeting Rh immune globulin prophylaxis.”
Many studies correlate the use of RHD genotyping with a decrease in the use of RhIG, she said (among other studies are the following examples: Darlington M, et al. BMC Pregnancy Childbirth. 2018;18:496; Haimila K, et al. Acta Obstet Gynecol Scand. 2017;96[10]:1228–1233; Papasavva T, et al. BMC Res Notes. 2016;9:198).
“Across all studies, which would involve populations with different prevalences of RhD positivity, in 25 to 39 percent of all genotyped pregnancies, unnecessary Rh immune globulin use was avoided,” she reported.
“So you compare reduced Rh immune globulin use and cost with the performance of this test, and to some extent at least, depending on the test used and the health care system economics where you live and work, you can offset the cost of the test with a reduction in Rh immune globulin use.”
In Canada in 2017, a group of transfusion and fetal medicine physicians, obstetricians, and scientists wrote a guideline “that was intended as a call to action for blood suppliers and funders, which in Canada are provincial governments, to take a look at routine noninvasive prenatal prediction of fetal RhD genotype,” Dr. Clarke, a member of the Canadian National Rh Working Group, said (Johnson J, et al. J Obstet Gynaecol Can. 2017;39[5]:366–373).
Their work was effective in attracting the funders’ attention, and several pharmacoeconomic analyses were done. (The pandemic delayed what had begun a few years prior, she noted.)
In the clinical practice guideline, issued by the Society of Obstetricians and Gynecologists of Canada, the authors explain the reasons RHD noninvasive prenatal testing should be done. The first is ethics—reducing the use of an unnecessary drug. “Is it ethically acceptable to continue giving Rh immune globulin when an alternative is available? We know there’s a substantial proportion of RhD-negative individuals at 28 weeks who get this drug and who don’t need it because their fetus is RhD-negative and there’s no chance of alloimmunization,” Dr. Clarke says.
Other reasons:
- RhIG is produced by immunizing blood donors. “Reducing the need for this might be an improvement,” she said.
- It’s a globally responsible approach in that RhIG worldwide is in short supply, and Rh disease continues to have devastating effects in some countries. “If we could make more Rh immune globulin available, more individuals could benefit from protection from alloimmunization.”
- It would mean joining international partners in providing state-of-the-art testing to support optimal transfusion practice. In addition to conserving Rh immune globulin, the numbers of laboratory tests and clinic visits could be reduced. And this would be in compliance “with the spirit of Choosing Wisely: Don’t transfuse blood if other non-transfusion therapies would be just as effective,” Dr. Clarke said.
- It would reduce the theoretical risk of transmissible viral and prion infections to patients and other adverse events. RhIG use has been safe over its 50 years, she notes, but there’s still a theoretical risk of infection and “we would eliminate that risk in those who don’t get the drug.”
- It would allow women to make an informed choice about RhIG administration and may improve adherence to anti-D treatment.
Dr. Robitaille called the SOGC clinical practice guideline “a turning point for noninvasive prenatal testing for fetal RHD in Canada” and described how its implementation got off the ground in Quebec.
In many European countries in which NIPT for fetal RHD is in place, blood establishments offer the testing, and “Canada is going in the same direction,” she said. Héma-Québec will provide the test for Quebec, and Canadian Blood Services will do so for the rest of Canada, and both plan to begin offering testing within the next 12 to 18 months. “Both organizations work in close collaboration, and we will be each other’s contingency plan,” Dr. Robitaille said.
The scope of any such program is one of the main factors to consider: Will it be a national or a local hospital-based program? Others to consider are the local laws and regulations, funding, and the health care structure. In Canada, with its universal care coverage, the Ministry of Health will pay for NIPT for fetal RHD for every pregnant patient, she said.

The Héma-Québec executive board in 2019 approved the business plan Dr. Robitaille and colleagues had developed once they decided Héma-Québec should offer the testing. Héma-Québec’s decision was based on the following: It was within the immunohematology reference laboratory’s mandate, and Héma-Québec’s team had the scientific and medical expertise, equipment, transportation logistics, IT and QA and other support, and close collaboration with hospital transfusion medicine services and the Ministry of Health. “We knew it would answer an unmet need requested by our medical community and that physicians would be willing to collaborate with us,” and that Canadian Blood Services and Héma-Québec “could rely on each other,” Dr. Robitaille said.
An evaluation by the Health Quality Council is required for any new test funded by the Ministry of Health, and the council’s report was delayed by the pandemic and not published until early 2021. “At first, the decision was disappointing,” Dr. Robitaille said. The council recommended that fetal RHD genotyping using maternal plasma not be offered. However, they said this test could be offered to the target population in the event that the method chosen, either a commercial kit or an in-house test, could be specified and that its performance and limitations are determined prospectively through a validation study carried out in real-world care settings. The report also said: “[B]ased on this new data, the offer of service is recognized as being effective and safe and that the costs it entails are acceptable relative to the clinical benefits obtained.”
“Early on,” Dr. Robitaille said, “we decided we would not develop an in-house assay, although our research and development department could have done it, due to all the licensing requirements.”
Three commercial RHD PCR kits were on the market in 2019, none of which was licensed by Health Canada. Two targeted a combination of exons 5, 7, and 10; the third kit targeted only exon 4.
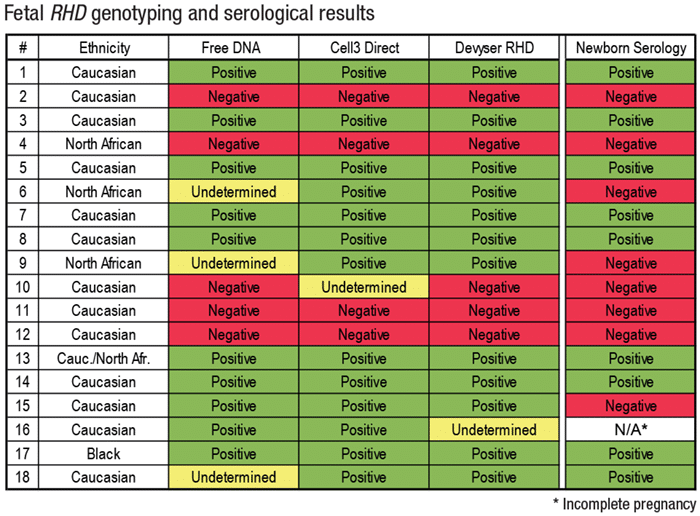
Her team evaluated the three assays—Free DNA Fetal Kit RhD (BioRad label), Cell3 Direct (Nonacus), and Devyser RHD—and compared the results with newborn serology (see “Fetal RHD genotyping and serological results,” above). When a result was undetermined, they requested a buccal swab of the newborn and performed an RHD genotype. “Serology RhD results were concordant with the RHD prediction for 15 of the 18 individuals,” Dr. Robitaille reported. “Two newborns from mothers of North African origin were found RhD-negative by serology” (cases six and nine), while the cell-free DNA analysis predicted an RhD-positive or undetermined result. Newborn genomic DNA was subsequently analyzed.
The triplex PCR analysis targeting various exons found case No. 6 was indeed RhD-negative due to the absence of amplification of all exons tested, she said, noting sequencing analysis wasn’t possible. For case No. 9, sequencing and multiplex PCR analysis found one pseudogene and one RHD-CE-D hybrid allele. One Caucasian newborn, case No. 15, was found to be RhD-negative while all kits predicted RhD positivity. Subsequent genetic analysis of the newborn RHD wasn’t possible.
“Given that the recommendation for positive or undetermined results would be to administer RhIG, safety would be ensured,” Dr. Robitaille summed up. The Ministry of Health issued its mandate in 2022.
Implementation of the test is planned for 2026.
The project timeline consists of 20 steps and five phases. Planning is phase No. 1. “Planning will avoid going back and forth and prevent unpleasant surprises,” Dr. Robitaille said. Phase No. 2 is acquiring and validating the equipment and method. No. 3 is IT solution development configuration and validation. No. 4 is change management—“knowledge translation to help professionals in the community, as well as regulatory training of Héma-Québec employees,” leading to the launch, Dr. Robitaille said. Closure of the project is the final phase.
She expanded on phases two and three. First, when choosing an assay, ask the following: Will you use a commercial or an in-house assay? What’s available, and is your laboratory familiar with the technology and equipment? Is the test used elsewhere and, if so, what is the experience with it? What are its technical requirements? “Take a close look at performance and stability criteria in considering the transportation time of your samples,” she said. Depending on test volume, automation and high-throughput analysis might be required. And cost is a factor.
Héma-Québec’s assay will be the Devyser RHD, which targets exon 4 and is licensed by Health Canada, Dr. Robitaille said. Fetal DNA will be extracted from maternal plasma using Qiagen’s QIASymphony. Thermo Fisher’s QuantStudio will be used for amplification of DNA and real-time PCR detection, and results will be reported via the Quebec health data repository.
Testing will be offered from week 11 of the pregnancy. All blood samples are stable for six days at room temperature and for two weeks when frozen between -18°C to -28°C, per the package insert. “We will ask that a sample be sent from the collection site to Héma-Québec within five days to respect the six-day stability at room temperature,” Dr. Robitaille said. “However, considering that Quebec covers a large territory and that winter weather conditions can be extreme, we may ask remote sites to centrifuge the plasma and freeze the sample if the delays cannot be respected.” Another option is to use a Streck tube, she added.
Post-implementation quality indicators are, among others, turnaround time, assay specificity and sensitivity, repeat sampling rate, indeterminate results rate, and external QA competency/proficiency testing, which “are now available and strongly recommended,” she said, citing the following validation and QA recommendation (Clausen FB, et al. Vox Sang. 2022;117[2]:157–165).
Dr. Robitaille said the challenges and barriers are many but that she would limit herself to a few, and the main challenge has been the IT component.
“As of now, the Héma-Québec LIS is not integrated with the health care system LIS. We currently use faxes to send our immunohematology reference lab results, which is not viable for high-volume analysis.” They expect to perform about 12,000 analyses per year. “Thus, we need a secure connection to download the result to the electronic health data repository,” she said.
The first unexpected barrier was legislation prohibiting Héma-Québec from downloading data to the repository, but they were successful in getting the legislation modified. Now, a cybersecurity certificate is required for the LIS, and an interface must be developed and validated to download results. “This is underway, but it’s a huge development that will take many months,” Dr. Robitaille said.
Regulation could have been another barrier were it not for the foresight in Canada’s regulation for blood and blood components. Its 2020 RhIG standard says all Rh-negative women should receive RhIG at 28 weeks gestation and within 72 hours after delivery, abortion, amniocentesis, or any other event or procedure that can cause fetomaternal hemorrhage. It adds that all RhD-negative women at 20 weeks or more gestation who experience an event or procedure that can cause FMH shall be tested by a method that will detect FMH requiring more than a single dose of RhIG for effective prophylaxis. “Exceptions shall be if the fetus or neonate is tested to be RhD negative (by genotype or by serology). . . .”
“So no modification is required,” Dr. Robitaille said, “but it’s an important aspect to consider.”
She and colleagues are looking forward to achieving success, which she sums up this way: Targeted antenatal RhIG prophylaxis is provided only to those who need it based on the prediction of fetal RHD. NIPT from fetal RHD is available to all RhD-negative pregnant people in Canada. Results are reported clearly and accurately to clinicians and transfusion services. “Finally,” Dr. Robitaille said, “RhIG use decreases and the alloimmunization rate remains very low.”
Sherrie Rice is editor of CAP TODAY. The Devyser assay is cleared in Europe, Canada, and Australia. Devyser’s CAP-accredited laboratory in Atlanta offers the genotyping service.

