From the CAP Cytopathology Committee;
Jordan Paul Reynolds, MD, Derek B. Allison, MD, Editors
Mohammad Al-Attar, MD
M. Lisa Zhang, MD
May 2025—Claudin 18.2 (CLDN18.2), an isoform of claudin-18, is a transmembrane tight junction protein essential for maintaining barrier function and cell polarity in normal gastric and pancreatic epithelium. In malignant epithelial cells, the loss of polarity exposes the CLDN18.2 epitope, leading to its increased expression in gastric and pancreatic adenocarcinomas.1,2 CLDN18.2 has drawn attention as a therapeutic target, particularly with the development of the monoclonal IgG antibody zolbetuximab (Vyloy, Astellas Pharma).3 Zolbetuximab exerts its antitumor effects via both antibody-dependent and complement-mediated cytotoxicity and has demonstrated significantly improved progression-free and overall survival when combined with standard chemotherapy in two pivotal phase three clinical trials (SPOTLIGHT and GLOW).4,5 These data validated CLDN18.2 as a promising target in advanced, HER2-negative gastric and gastroesophageal junction (GEJ) adenocarcinoma.
In October 2024, the FDA approved zolbetuximab in the front-line setting for treatment of locally advanced unresectable or metastatic HER2-negative gastric/GEJ adenocarcinoma. This approval was granted alongside the companion diagnostic immunohistochemistry assay Ventana CLDN18 (43-14A) RxDx (Ventana Medical Systems, Roche Diagnostics). Both trials defined CLDN18.2 positivity as moderate to strong membranous staining in greater than or equal to 75 percent of tumor cells, with approximately 38 percent of tumors meeting this threshold.
While clinical trials have largely focused on surgical specimens (core biopsies and resections), data on CLDN18.2 testing in cytology preparations are scarce. To date, only one study6 has specifically examined the feasibility of CLDN18.2 IHC on cell blocks from effusion cytology samples (Fig. 1) in patients with gastric adenocarcinoma, demonstrating relatively high concordance (83.7 percent) between effusion cytology and tissue specimens—though they employed a less stringent positivity cutoff (≥40 percent of tumor cells with moderate to strong expression) than that used in the zolbetuximab trials.
A recent international panel of expert gastrointestinal pathologists published recommendations for CLDN18.2 IHC evaluation, suggesting that at least 50 viable malignant cells (preferably 100) be evaluated in effusion cytology.7 However, additional studies are needed to refine cytology-specific testing guidelines, including optimal thresholds for tumor cellularity, preanalytical variables (e.g. collection media, fixatives, and preservatives), and postanalytical considerations that may affect antigenicity and staining interpretation in cell blocks.
Given the emerging requests from oncologists for CLDN18.2 status—and with a growing array of CLDN18.2-based therapies (e.g. bispecific CLDN18.2/CD3 antibodies) under investigation for a broader range of tumors including pancreatic ductal adenocarcinoma—cytopathologists will increasingly encounter this immunostain. Establishing familiarity with its staining patterns and performance characteristics in limited cytologic samples will be important as testing expands beyond traditional surgical specimens.
In summary, CLDN18.2 has rapidly become a key biomarker for novel targeted therapies in advanced gastric/GEJ adenocarcinoma. Cytopathologists can expect a rising demand for CLDN18.2 interpretation and have the potential to play an important role in the management of patients for whom cytology/cell block and small biopsy samples represent the initial diagnostic material in patients presenting with advanced disease, ultimately broadening treatment opportunities for patients with these challenging cancers. As a result, additional studies using cytology material are warranted to ensure accurate results are reported for optimal therapy selection.
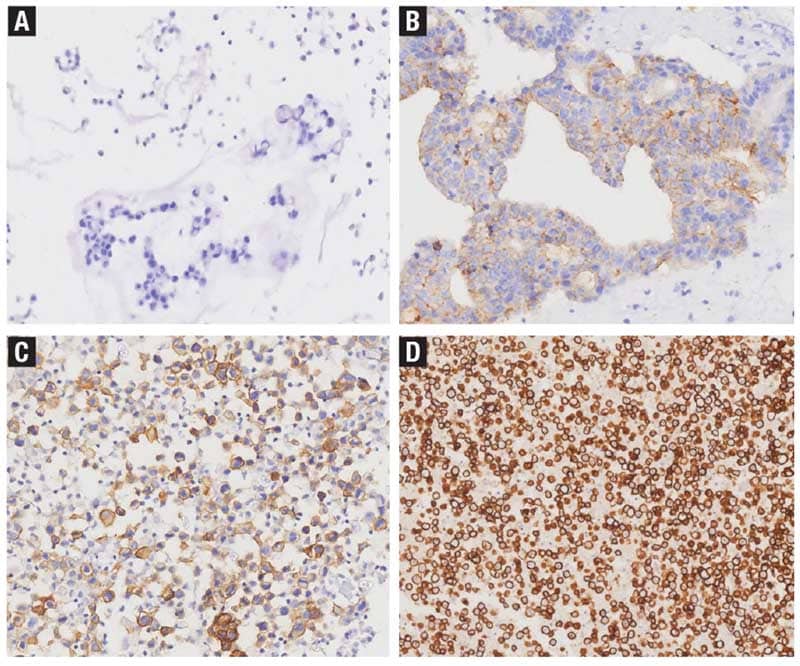
Reprinted from Dai J, et al. Claudin18.2 expression and clinicopathological features in cytology effusion specimens from gastric adenocarcinoma: A comparative study with tissue specimens. Cancer Cytopathology. 2023;131(6):365–372, with permission from John Wiley & Sons. © 2025 American Cancer Society.
- Yang YJ, Jeng YM, Yang CY, Hu HW. Claudin-18 immunohistochemical staining facilitates the identification of metastatic carcinoma of gastric or pancreatic origin in effusion specimens. Appl Immunohistochem Mol Morphol. 2022;30(1):8–13.
- Isidro RA, Abukhiran I, Dunseth CD, et al. Strong annexin A10 expression supports a pancreatic primary and combined annexin A10, claudin 18, and SOX2 expression supports an esophagogastric origin in carcinomas of unknown primary. Am J Surg Pathol. 2023;47(4):440–452.
- Nakayama I, Qi C, Chen Y, Nakamura Y, Shen L, Shitara K. Claudin 18.2 as a novel therapeutic target. Nat Rev Clin Oncol. 2024;21(5):354–369.
- Shitara K, Lordick F, Bang YJ, et al. Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial. Lancet. 2023;401(10389):1655–1668.
- Shah MA, Shitara K, Ajani JA, et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat Med. 2023;29(8):2133–2141.
- Dai J, Zheng H, Jin J, Cheng Y, Xu H. Claudin18.2 expression and clinicopathological features in cytology effusion specimens from gastric adenocarcinoma: a comparative study with tissue specimens. Cancer Cytopathol. 2023;131(6):365–372.
- Fassan M, Kuwata T, Matkowskyj KA, Röcken C, Rüschoff J. Claudin-18.2 immunohistochemical evaluation in gastric and gastroesophageal junction adenocarcinomas to direct targeted therapy: a practical approach. Mod Pathol. 2024;37(11):100589.
Dr. Al-Attar is a fellow in cytopathology, Mass General Brigham. Dr. Zhang is a cytopathologist and gastrointestinal pathologist, Massachusetts General Hospital, and assistant professor of pathology, Harvard Medical School. Dr. Zhang is a member of the CAP Cytopathology Committee.

