Amy Carpenter
September 2025—In a CAP session last year on timely topics in thyroid tumors, Zubair W. Baloch, MD, PhD, shed light on poorly differentiated and differentiated high-grade follicular-derived carcinomas.
“This is something new in the WHO 5th edition of the thyroid tumor classification scheme, and I think it’s about time that we have a different category and not really split them too much,” said Dr. Baloch, an editor and author of the 5th edition of the WHO Classification of Endocrine and Neuroendocrine Tumors.
Why the category is new and what to know was part of the timely topics session, for which he paired up with Nikolina Dioufa, MD, PhD, MSc, who spoke about noninvasive follicular thyroid neoplasm with papillary-like nuclear features and follicular variant of papillary thyroid carcinoma. (See part one in the August issue.)
Dr. Baloch is a professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania, Perelman School of Medicine at UPenn, and director of the hospital’s endocrine pathology service.
He opened with a case: a 54-year-old euthyroid patient with no history of clinical thyroid disease or hypo- or hyperthyroidism, whose 3.5-cm left thyroid mass had irregular margins and was hypoechoic. “Typical of tumors that are infiltrative,” Dr. Baloch said, noting a lateral neck extension. There was associated left cervical lymphadenopathy.
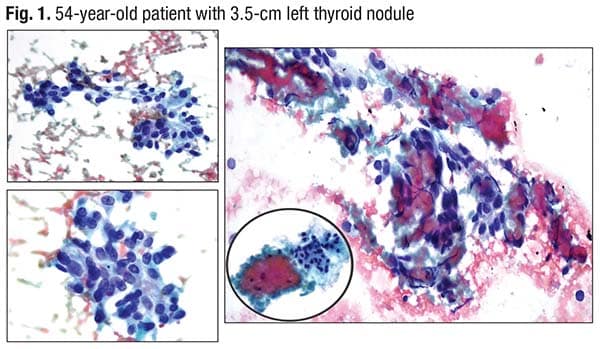
An almost papillary-forming group was seen in the cytology (Fig. 1). The elongated nuclei with nuclear features are suspicious for or diagnostic of papillary thyroid carcinoma. The problem, Dr. Baloch said, is the necrotic debris clinging to the cells (right image).
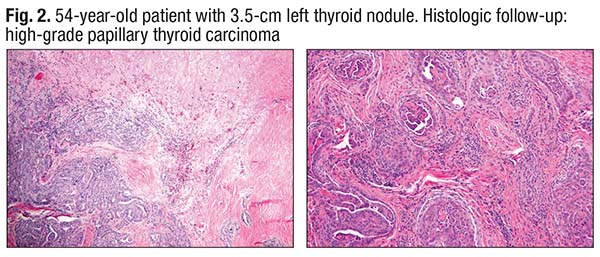
A colleague, Tom Hu, MD, signed the case out as papillary thyroid carcinoma with focal necrosis and noted there could be a high-grade transformation in this carcinoma. “Dr. Hu was right,” Dr. Baloch said, noting “a good amount of necrosis, which has a lot of cell debris” (Fig. 2). Papillary formation, which has the nuclear features of papillary thyroid carcinoma, is seen at the bottom. “No anaplastic carcinoma,” Dr. Baloch said, “but definitely geographic necrosis, which was very concerning in this case.”
When necrosis is seen in a thyroid tumor, he said, “you think, could this be anaplastic thyroid carcinoma? It’s something to think about because this is papillary carcinoma. It does have tall-cell features, but this is not anaplastic carcinoma.”
The diagnosis: high-grade papillary thyroid carcinoma, tall-cell variant.
Case No. 2 was one that came to Dr. Baloch in 2024. A 59-year-old was found in 2019 to have a 2.7-cm TR 4 right thyroid nodule.
 The other clinical history was unrelated to thyroid disease, and there was no family history of thyroid cancer and no history of external beam radiation therapy to the head or neck before age 20.
The other clinical history was unrelated to thyroid disease, and there was no family history of thyroid cancer and no history of external beam radiation therapy to the head or neck before age 20.
The patient’s nodule was fairly solid, hypoechoic, and separated from the surrounding thyroid gland, Dr. Baloch said, noting a lot of peripheral vascularity. “It was a hypoechoic lesion with [a few] irregular margins in the back.”
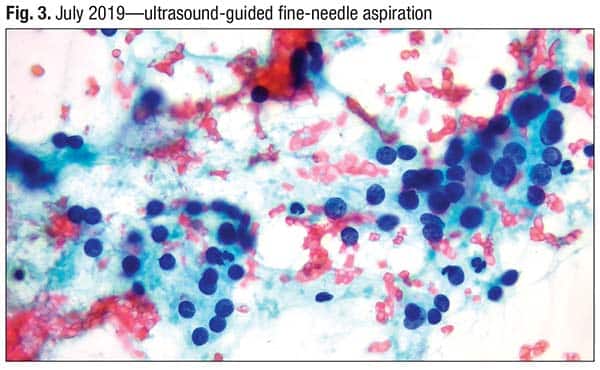
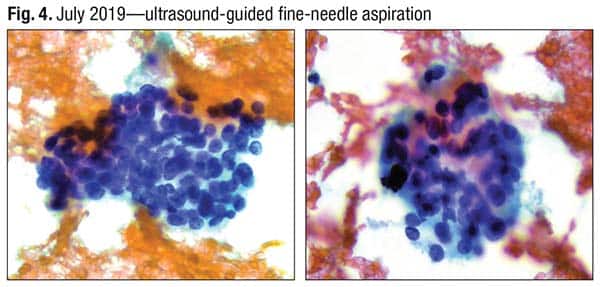
In a 2019 FNA, a monotonous population of cells was seen. There are no nuclear features of papillary carcinoma (Fig. 3). Nuclear overlapping and crowding are seen in Fig. 4. There was little cytoplasm, Dr. Baloch said. “When you have groups like this, and if you’re smearing it very gently, you will get a hint of cytoplasm.”
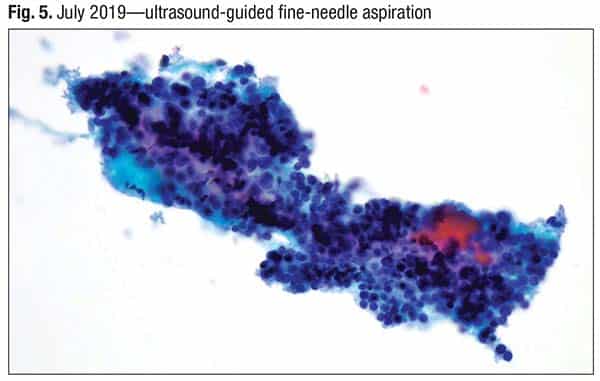
Thick colloid is seen in the ThinPrep specimen (Fig. 5), “but there’s a very monotonous population of the cells,” he said. The nodule was classified as atypia of undetermined significance—Bethesda III (Bethesda System for Reporting Thyroid Cytopathology). “But this could also be a good case of follicular neoplasm—Bethesda IV.”
 The specimen was sent for molecular analysis, which identified a PAX8/PPARG alteration. The probability of cancer or noninvasive follicular thyroid neoplasm with papillary-like nuclear features was intermediate-high, and the highest risk of malignancy by this test was about 70 percent.
The specimen was sent for molecular analysis, which identified a PAX8/PPARG alteration. The probability of cancer or noninvasive follicular thyroid neoplasm with papillary-like nuclear features was intermediate-high, and the highest risk of malignancy by this test was about 70 percent.
The PAX8/PPARG rearrangement is created by a translocation between chromosomal regions 2q13 and 3p25. This translocation results in a fusion transcript wherein most of the coding sequence of PAX8 (2q13) is fused in frame with the entire coding exons of PPARγ1 (3p25). The PAX8/PPARG gene fusion results in production of a PAX8-PPARγ fusion protein (PPFP) and is found in about 30 to 35 percent of follicular thyroid carcinomas, as well as a subset of follicular variant of papillary thyroid carcinomas. In vitro and in vivo evidence indicate that PPFP can act as an oncoprotein.
The patient refused the recommended surgical excision owing to family obligations during the pandemic.
The patient presented to an ENT clinic in July 2024 with headache, nausea, and balance problems; the right thyroidal mass had progressed and was causing airway compression. An MRI in the ED showed a bulky soft tissue mass centered within the central skull base, with aggressive features, including bony destruction, extension into the middle and anterior cranial fossa, prepontine cistern, involving the cavernous sinus and Meckel’s cave, and elevating and likely compressing of the optic chiasm. The differential included atypical aggressive appearance of macroadenoma, metastatic focus, lymphoma, and (less likely) chordoma or nasopharyngeal carcinoma.
“No one was thinking that this could be related to the thyroid because this is a very atypical presentation for a thyroid tumor,” Dr. Baloch said.
The patient was intubated and underwent biopsy of the skull base mass, and core needle biopsy of the neck mass.
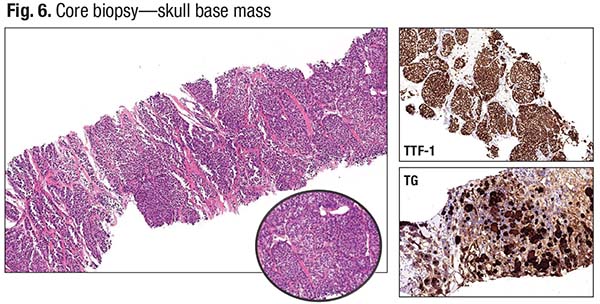
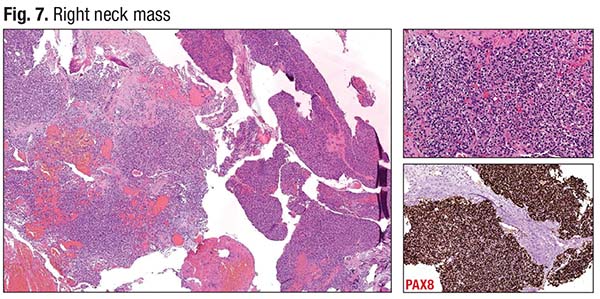 The core biopsy of the skull base mass is seen in Fig. 6. All neuroendocrine stains were negative, but positive for cytokeratin and focally positive for synaptophysin. The immunostains for thyroglobulin and TTF-1 were also positive. “So this was a primary thyroid tumor. Given the solid growth pattern, it’s reasonable to suspect a poorly differentiated carcinoma with metastasis. It’s an atypical presentation,” Dr. Baloch said. The neck mass core biopsy (also FNA) showed a very solid, nested growth pattern, typical for a thyroid primary, with an intermediate risk of tumor recurrence. The neck mass was PAX8 positive initially and was morphologically similar to the skull base tumor (Fig. 7).
The core biopsy of the skull base mass is seen in Fig. 6. All neuroendocrine stains were negative, but positive for cytokeratin and focally positive for synaptophysin. The immunostains for thyroglobulin and TTF-1 were also positive. “So this was a primary thyroid tumor. Given the solid growth pattern, it’s reasonable to suspect a poorly differentiated carcinoma with metastasis. It’s an atypical presentation,” Dr. Baloch said. The neck mass core biopsy (also FNA) showed a very solid, nested growth pattern, typical for a thyroid primary, with an intermediate risk of tumor recurrence. The neck mass was PAX8 positive initially and was morphologically similar to the skull base tumor (Fig. 7).
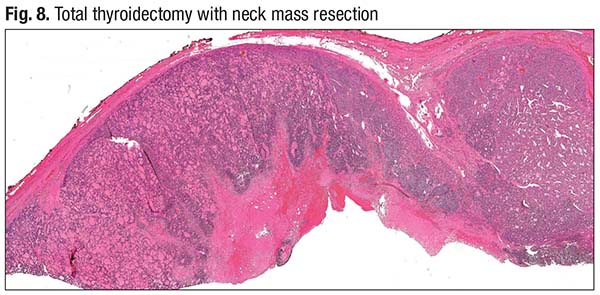
The neck mass completely replaced the entire lobe of the thyroid. On microscopic examination, it was encapsulated and showed a cellular architecture with a solid and trabecular growth pattern (Fig. 8, upper right). At low power, there’s a diagnostic clue: “If you see empty spaces between tumor nests with an almost staghorn-like appearance, start considering that you may be dealing with something beyond a simple well-differentiated follicular pattern, potentially a tumor with high-grade morphology.” The necrotic center of the tumor and the periphery of the tumor, encapsulated, are seen in Fig. 9. “It is invading the blood vessel,” Dr. Baloch said (left side of image).
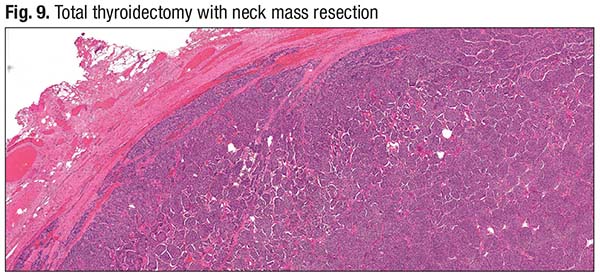 “This was diagnosed as a poorly differentiated [thyroid] carcinoma, arising in association with invasive encapsulated follicular variant of papillary [thyroid] carcinoma,” he said. It has extensive vascular invasion (more than four vessels) and extensive extrathyroidal extension.
“This was diagnosed as a poorly differentiated [thyroid] carcinoma, arising in association with invasive encapsulated follicular variant of papillary [thyroid] carcinoma,” he said. It has extensive vascular invasion (more than four vessels) and extensive extrathyroidal extension.
These tumors can be encapsulated or nonencapsulated and may or may not be associated with a well-differentiated component, according to the Turin criteria for poorly differentiated thyroid carcinoma, applicable in the 5th edition of the WHO classification. Their growth pattern is solid, insular, or trabecular, “and that is why they give rise to the look of a neuroendocrine tumor,” Dr. Baloch said. Papillary thyroid carcinoma nuclei are absent.
One of the following three diagnostic criteria must be met: convoluted nuclei, mitotic count ≥3 per 2 mm2 (about 10 high-power fields on most microscopes), or necrosis.
“These are not diagnostic difficulties,” Dr. Baloch said.
The question, he added, is how much of a tumor has to be poorly differentiated to call the entire tumor a poorly differentiated carcinoma. “Once you have a poorly differentiated carcinoma component, you have to mention that in your diagnosis,” he said. He follows the one-third rule: “If it is 30 percent or more, I will call the whole thing a poorly differentiated carcinoma. These tumors carry intermediate to high risk of loco-regional recurrence and distant metastases.”
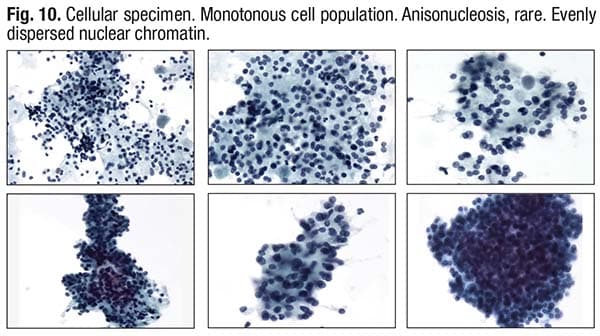 Poorly differentiated thyroid carcinomas are RAS-mutation-predominant tumors with an increased rate of distant metastasis. “When looking at cytology, you have to be sensitive to when you encounter a very cellular specimen with nuclear overlapping and crowding” (Fig. 10). In such scenarios, he said, one must also carefully search for tumor necrosis, which usually appears as debris and “clings” to the lesional cells, and, if needed, process the whole specimen.
Poorly differentiated thyroid carcinomas are RAS-mutation-predominant tumors with an increased rate of distant metastasis. “When looking at cytology, you have to be sensitive to when you encounter a very cellular specimen with nuclear overlapping and crowding” (Fig. 10). In such scenarios, he said, one must also carefully search for tumor necrosis, which usually appears as debris and “clings” to the lesional cells, and, if needed, process the whole specimen.
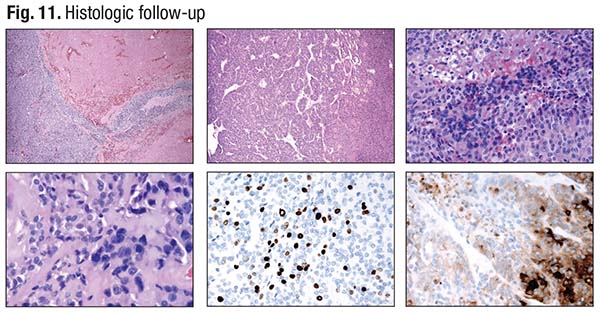 In Fig. 11 is a classic case of a poorly differentiated carcinoma with an insular and solid growth pattern. Dark, convoluted nuclei are in the background (bottom left), and in the bottom center is a high Ki-67 index. “You can perform thyroglobulin immunostain, which will be strongly expressed in the well-differentiated component,” Dr. Baloch said (bottom right). The poorly differentiated component will show comparatively weaker expression. “However, if the thyroglobulin was completely negative, you may have to rule out anaplastic thyroid carcinoma.”
In Fig. 11 is a classic case of a poorly differentiated carcinoma with an insular and solid growth pattern. Dark, convoluted nuclei are in the background (bottom left), and in the bottom center is a high Ki-67 index. “You can perform thyroglobulin immunostain, which will be strongly expressed in the well-differentiated component,” Dr. Baloch said (bottom right). The poorly differentiated component will show comparatively weaker expression. “However, if the thyroglobulin was completely negative, you may have to rule out anaplastic thyroid carcinoma.”
In the WHO classification of thyroid neoplasms 5th edition, the category of malignant neoplasms of follicular cell origin includes well-differentiated carcinomas, which can be papillary, follicular, or oncocytic (this category includes follicular thyroid carcinoma, invasive encapsulated follicular variant papillary carcinoma, papillary thyroid carcinoma, oncocytic carcinoma); high-grade follicular-derived nonanaplastic carcinomas; and anaplastic thyroid carcinoma. The category of high-grade follicular-derived nonanaplastic carcinoma includes differentiated high-grade thyroid carcinoma and poorly differentiated thyroid carcinoma.
The current WHO classification of thyroid tumors of follicular cell origin is supported by the molecular profile of these tumors, and, interestingly, it reveals “molecular gradation” from a very low-risk neoplasm to high-risk tumors, which will include poorly differentiated or high-grade well-differentiated tumors and anaplastic carcinomas. The well-differentiated follicular cell-derived thyroid tumors can be divided into RAS-like tumors, which are follicular patterned tumors, and BRAF-like tumors, which have readily identifiable nuclear features of papillary carcinoma. RAS-like molecular alterations result in tumors that are often encapsulated and have an expansile pattern of growth and subtle/less florid nuclear atypia, whereas BRAF-like molecular alterations give rise to infiltrative tumors with florid nuclear atypia.
Even within well-differentiated carcinoma groups, some tumor subtypes are associated with aggressive clinical behavior, frequent local recurrences, or distant metastasis, Dr. Baloch said, “the poster child being the tall-cell subtype. This is important because clinicians treating thyroid carcinomas use risk stratification to manage these tumors.”
The risk stratification process generally starts postoperatively with histopathologic staging. This does not, however, accurately assess the risk of recurrence/persistence. Patients initially considered to be at high risk may ultimately do very well, yet be burdened by frequent unnecessary clinical and/or radiological follow-up.
“Thus clinical risk stratification is a dynamic process, not a static process,” Dr. Baloch said (Tuttle RM, et al. J Clin Endocrinol Metab. 2019;104[9]:4087–4100).
 Before edition eight of the American Joint Committee on Cancer/tumor, node, metastasis staging system, any form of extrathyroidal extension, even microscopic, upstaged the patient to T3b, “which suffers from observer variability,” Dr. Baloch said. “Even though we were upstaging those patients based on mere presence of microscopic extrathyroidal extension,” their tumors were behaving like an encapsulated tumor or one confined to the thyroid gland. Therefore, per edition eight, the gross extrathyroidal extension seen or documented by the surgeon is required for stage T3b.
Before edition eight of the American Joint Committee on Cancer/tumor, node, metastasis staging system, any form of extrathyroidal extension, even microscopic, upstaged the patient to T3b, “which suffers from observer variability,” Dr. Baloch said. “Even though we were upstaging those patients based on mere presence of microscopic extrathyroidal extension,” their tumors were behaving like an encapsulated tumor or one confined to the thyroid gland. Therefore, per edition eight, the gross extrathyroidal extension seen or documented by the surgeon is required for stage T3b.
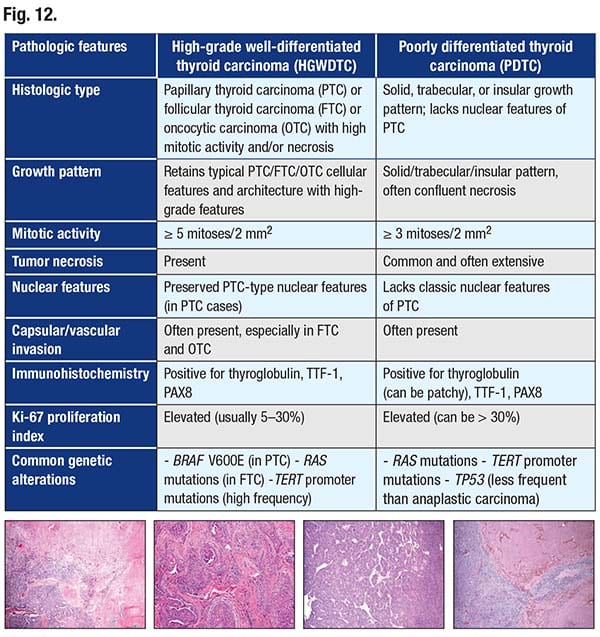
In Fig. 12 are the criteria for poorly differentiated thyroid carcinoma and differentiated high-grade thyroid carcinoma.
Counting mitoses is a recent change, with the WHO 5th edition preferring the mitotic count to be reported as mitoses per 2 mm2 instead of per 10 high-power fields (Cree IA. Endocr Pathol. 2022;33[1]:3–5; Cree IA, et al. Mod Pathol. 2021;34[9]:1651–1657).
“I stay away from the areas of necrosis and from the areas of FNA track because then you may note mitoses per 2 mm2 is a different way of reporting,” Dr. Baloch noted. “It lessens variability.”
 As for the diagnostic challenges, he said these high-grade transformations can be seen in infiltrative or encapsulated neoplasms, along with the well-differentiated component. Importantly, he said, if something looks like a poorly differentiated carcinoma because of the solid growth pattern, and there is mitosis but no necrosis and it is encapsulated, and invasion cannot be documented by doing deeper sections or by submitting the entire periphery of the tumor, it cannot be called poorly differentiated thyroid carcinoma encapsulated. “There’s no noninvasive subtype of poorly differentiated thyroid carcinoma—as per WHO 5th edition, that is follicular adenoma,” Dr. Baloch said, noting it’s been shown in two studies that such tumors follow an indolent clinical course.
As for the diagnostic challenges, he said these high-grade transformations can be seen in infiltrative or encapsulated neoplasms, along with the well-differentiated component. Importantly, he said, if something looks like a poorly differentiated carcinoma because of the solid growth pattern, and there is mitosis but no necrosis and it is encapsulated, and invasion cannot be documented by doing deeper sections or by submitting the entire periphery of the tumor, it cannot be called poorly differentiated thyroid carcinoma encapsulated. “There’s no noninvasive subtype of poorly differentiated thyroid carcinoma—as per WHO 5th edition, that is follicular adenoma,” Dr. Baloch said, noting it’s been shown in two studies that such tumors follow an indolent clinical course.
Gubbiotti, et al., in a study on which Dr. Baloch is a coauthor, found that invasive poorly differentiated thyroid carcinoma tumors lacking a capsule demonstrate greater rates of disease-related death despite showing similar adverse histologic features to invasive encapsulated tumors (Gubbiotti MA, et al. Hum Pathol. 2023;136:96–104). Wong, et al., found that truly encapsulated poorly differentiated thyroid carcinoma tumors without capsular invasion lack adverse histologic features and illustrate excellent long-term outcomes in terms of recurrences, metastases, and survival (Wong KS, et al. Thyroid. 2019;29[9]:1255–1261).
“It has been documented in multiple studies,” Dr. Baloch said, “that encapsulated and poorly differentiated carcinomas behave very differently than infiltrative tumors.”
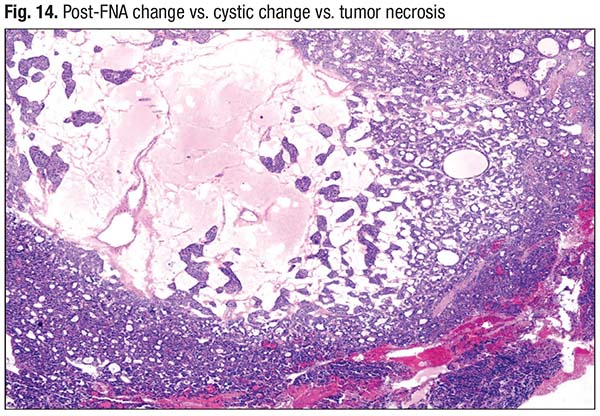
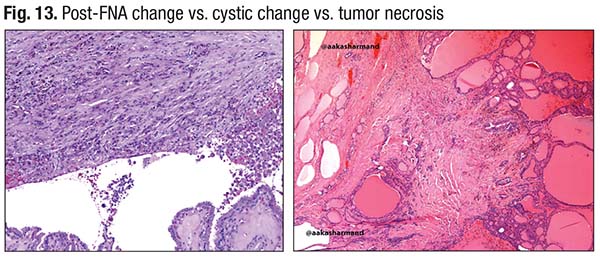
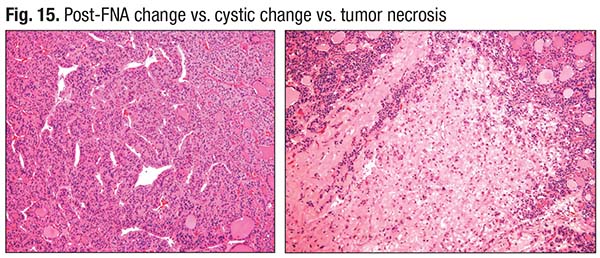 Necrosis, too, is a challenge, he said. “You have to avoid areas of FNA and associated fibrosis because FNA track can be mistaken for necrosis, especially in oncocytic tumors. FNA can cause infarction in oncocytic tumors and be mistaken for tumor necrosis”(Fig. 13). He has seen adenomatous and cystic change in these tumors mistaken for necrosis (Fig. 14). An adenoma with its solid growth pattern with central edematous change can be interpreted as having tumor necrosis. Fig. 15 shows what Dr. Baloch describes as true necrosis: “There is significant nuclear debris, consistent with the solid growth pattern.” On the left is a poorly differentiated carcinoma; the right side shows a more well-differentiated area that also exhibits necrosis.
Necrosis, too, is a challenge, he said. “You have to avoid areas of FNA and associated fibrosis because FNA track can be mistaken for necrosis, especially in oncocytic tumors. FNA can cause infarction in oncocytic tumors and be mistaken for tumor necrosis”(Fig. 13). He has seen adenomatous and cystic change in these tumors mistaken for necrosis (Fig. 14). An adenoma with its solid growth pattern with central edematous change can be interpreted as having tumor necrosis. Fig. 15 shows what Dr. Baloch describes as true necrosis: “There is significant nuclear debris, consistent with the solid growth pattern.” On the left is a poorly differentiated carcinoma; the right side shows a more well-differentiated area that also exhibits necrosis.
“Necrosis and mitotic activity are important features to watch for. I believe this will significantly impact the way we approach head and neck, as well as endocrine pathology,” he said, “because these tumors behave differently.”
FNA is not ideal for diagnosing differentiated high-grade thyroid carcinoma (Torous VF, et al. Cancer Cytopathol. 2024;132[8]:525–536). In their multi-institutional study of 40 such cases with prior FNA, Dr. Baloch and coauthors reported that more than 97 percent of cases were classified as Bethesda category IV (follicular neoplasm or oncocytic follicular neoplasm), category V (suspicious for malignancy), or category VI (malignant). About 25 percent of differentiated high-grade thyroid carcinoma cases showed cytologic features that included marked cytologic atypia, increased anisonucleosis, large oval nuclei, mitotic activity, or necrosis (p < .05). “And that is something to watch for in these large-size tumors or the tumors that are infiltrating,” he said. Sixty-eight percent of differentiated high-grade thyroid carcinoma cases were associated with high-risk molecular alterations, and TERT mutations occurred in 41 percent, of which 89 percent were associated with a second mutation, usually RAS or BRAF p.V600E.
The diagnosis of thyroid carcinoma today follows a “lasagna model,” Dr. Baloch said. Multiple layers—clinical presentation, ultrasound risk stratification, molecular profiling (pre- or postoperative), histologic classification, and clinical risk assessment—overlap and complement each other to provide a comprehensive and accurate evaluation.
Amy Carpenter is CAP TODAY senior editor.

