For myelodysplastic neoplasms or syndromes, the fifth edition of the World Health Organization classification of hematolymphoid tumors and the International Consensus Classification created additional genetically defined categories.
(For WHO: MDS with biallelic TP53 inactivation. For ICC: MDS with mutated TP53. And ICC has MDS with mutated SF3B1; it omitted MDS with ring sideroblasts as a distinct category.)
In the WHO classification are novel morphologic categories (hypoplastic MDS, MDS with fibrosis), optional designation of unilineage versus multilineage dysplasia, and nomenclature changes. The authors of the WHO fifth edition review write, “The classification introduces the term myelodysplastic neoplasms (abbreviated MDS) to replace myelodysplastic syndromes, underscoring their neoplastic nature and harmonizing terminology with MPN” (Khoury JD, et al. Leukemia. 2022;36[7]:1703–1719).
In the ICC is the novel MDS/AML entity, defined by 10 to 19 percent blasts in the bone marrow or peripheral blood and no AML-defining genetics. The entity does not apply to pediatric patients (Arber DA, et al. Blood. 2022;140[11]:1200–1228).
Within AML there are novel AML-defining genetic alterations with increased blasts (> five percent, WHO; 10 percent, ICC): KMT2A, MECOM, NUP98 rearrangements; ICC only: NPM1 mutation, CEBPA mutation. Only the bZIP domain of CEBPA is considered class-defining in the ICC; the WHO classification includes biallelic (biCEBPA) and bZIP.
Other significant changes are the AML myelodysplasia-related subclassification (AML-MR) with its defining somatic mutations and, in ICC, the myeloid neoplasms with TP53 mutations. The latter include the following: MDS, multi-hit, <10 percent blasts; MDS/AML, 10–19 percent blasts, any TP53 mutation with VAF >10 percent; AML, ≥20 percent blasts, any mutation with VAF >10 percent.
Dr. Mirza shared the case of a 51-year-old woman who underwent genetic testing for breast cancer predisposition, which identified a TP53 mutation (p.Arg248Gln) with a VAF of 34 percent.
The patient was referred for further testing for suspicion of Li-Fraumeni syndrome, but germline genetic testing performed on her skin fibroblasts was negative for the mutation.
“The first question is whether this patient has cytopenias,” he said.
The patient’s CBC showed normal leukocyte and absolute neutrophil counts (6.5 × 109/L and 4.57 × 109/L). She had anemia (Hgb:10.9 g/dL). Her MCV was normocytic (96 fL). RDW-CV was 13.2, and platelet count was within normal range (366 × 109/L).
The patient has trilineage hematopoiesis, as seen in the clot particle in Fig. 1 (left). “It’s likely reasonably cellular for the patient’s age,” Dr. Mirza noted. In the aspirate smear (right) “there is evidence of nice, normal maturation to mature neutrophils and no significant increase in blasts. So there’s no dysplasia that is quantitively significant, the cellularity seems normal, and the only finding in the blood and bone marrow is her isolated anemia,” he said. He noted, too, the negative p53 stain (top right). “The high VAF did not necessarily show up in the stain, so we have a 34 percent VAF where the p53 stain is negative.”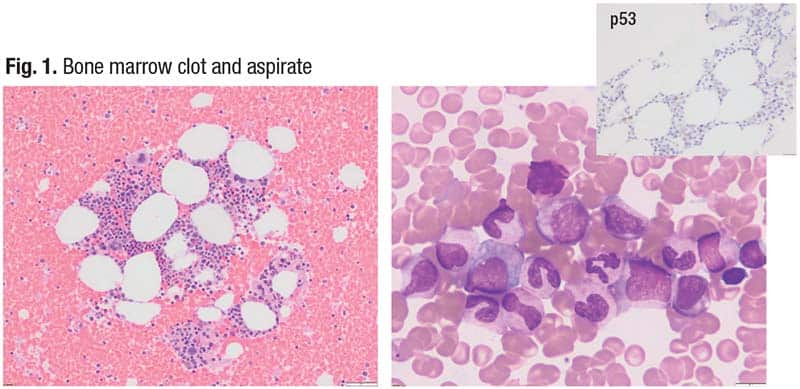
The spectrum for myeloid neoplasia is CHIP to clonal cytopenia of undetermined significance to MDS to AML (Loghavi S, et al. Mod Pathol. 2024;37[2]:100397). The patient’s cytopenia puts her in the CCUS category, Dr. Mirza said, noting the MDS category would require the presence of morphologic dysplasia.
What does the CCUS diagnosis mean for the patient? Weeks, et al., reported two years ago on their clonal hematopoiesis risk score, of which the prognostic variables are the presence or absence of a single DNMT3A mutation, presence or absence of high-risk mutation(s), mutation number, VAF, red cell distribution width–coefficient of variation, mean corpuscular volume, cytopenia, and age (Weeks LD, et al. NEJM Evid. Published online April 25, 2023. doi:10.1056/EVIDoa2200310).
The authors write, “We leveraged analysis of genetic, laboratory, and MN [myeloid neoplasm] outcomes data from 438,890 U.K. Biobank participants to definitively identify features of CHIP/CCUS that predict rise of MN.” In clinical cohorts, they found, most MN events occurred in high-risk patients with CHIP/CCUS. The patient’s score of 11 put her into the intermediate risk category (10–12).
In another article, Abelson, et al., reported on the prediction of AML in healthy individuals (Abelson S, et al. Nature. 2018;559[7714]:400–404). They looked at TP53 and other mutations in the control population versus the pre-AML population. Said Dr. Mirza, “The difference between the control and the pre-AML population was statistically significantly different in the presence of TP53 mutations. TP53 is an important finding that we need to take into account when subclassifying our patients with myeloid neoplasms.”
Dr. Mirza presented a second case, that of a 60-year-old woman whose WBC count is within normal range and whose ANC is without abnormality (3.3 × 109/L; 1.6 × 109/L, respectively). Hemoglobin is normal (12.7 g/dL), MCV is 97 fL, and platelet count is a little on the low side (45 × 109/L).
The bone marrow aspirate and biopsy (Fig. 2) show slightly hypocellular bone marrow for the patient’s age, Dr. Mirza said, “but interestingly within this the patient had an iron stain, and we counted 15 percent ring sideroblasts and only one percent blasts.” She had a diploid karyotype, and her SF3B1 mutation had a VAF of six percent. She also had a DNMT3A mutation at a VAF of five percent.
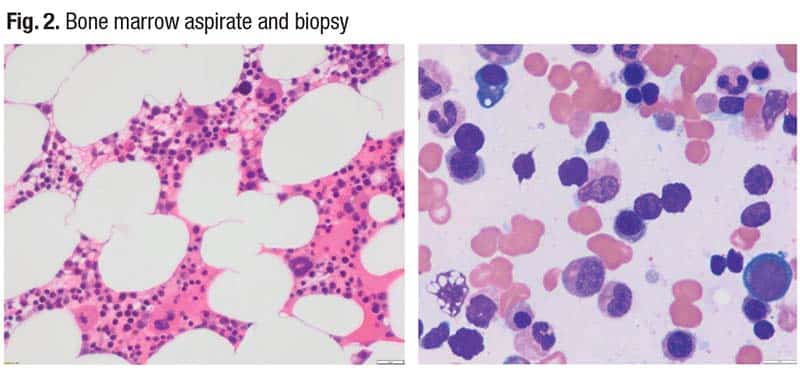
“The six percent VAF of the SF3B1 mutation puts this case into two different subclassifications” in the WHO and ICC, Dr. Mirza said. “The WHO fifth edition would qualify this case to be a myelodysplastic neoplasm with SF3B1 mutation, and the ICC would classify this as a myelodysplastic syndrome (NOS) with multilineage dysplasia. Effectively the patient has two different classification names for what we believe is one biological disease.”
Because there are two classifications in this case, the International Prognostic Scoring System (IPSS) is used to determine the risk stratification (https://mds-risk-model.com/). “Based on this model—which takes into account a variety of factors, such as the cytopenias, blast count, and mutations—this patient falls into a category of low risk,” with a score of -1.40.
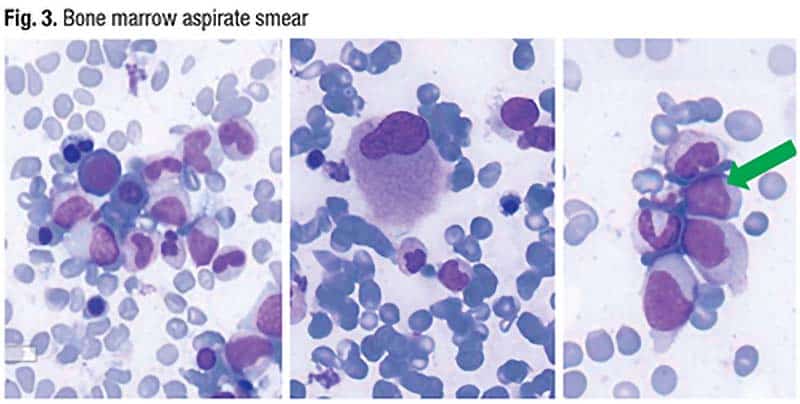
Dr. Mirza presented a complementary case: an 86-year-old male patient with a WBC of 3.9 × 109/L and normal ANC (2.6 × 109/L), Hgb of 11.4 g/dL, MCV of 98 fL, and a normal platelet count (163 × 109/L). There is multilineage dysplasia (Fig. 3) and there are 50 percent ring sideroblasts and four percent blasts (patchy on the aspirate smear). The patient has a diploid karyotype. The next-generation sequencing panel reveals the patient has KRAS, U2AF1, IDH2, NF1, ASXL1, and SF3B1 mutations at reasonably high VAFs (KRAS = 2, U2AF1 = 21, IDH2 = 42, NF1 = 5, ASXL1 = 44, and SF3B1 = 6).
Does this case present the same dilemma: MDS with SF3B1 or MDS, NOS with multilineage dysplasia? The answer, Dr. Mirza said, is that the diagnostic subcategorization for this patient is the same, “but when we plug the values into the International Prognostic Scoring System, this patient falls in the moderate risk category. So this becomes way more important therapeutically or prognostically.”
“SF3B1-mutated MDS is one of those subclassifications within the WHO and ICC that have differed in our way of diagnosing them,” he added. The CCUS and CHIP classifications are the same, “but in this particular disease entity there will be changes in the understanding of how we subclassify them.”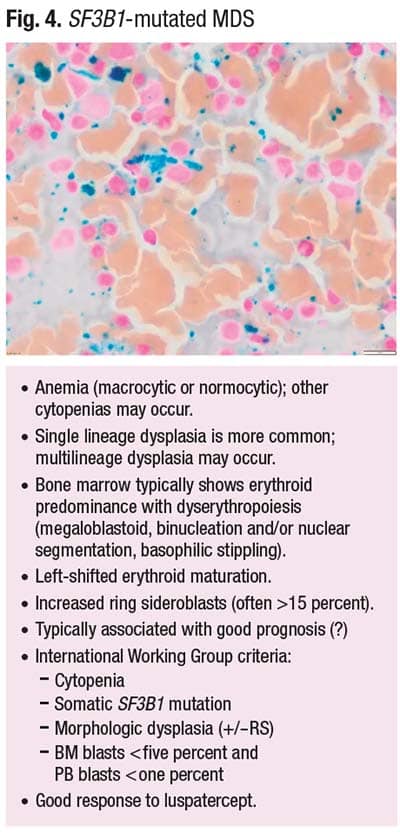
SF3B1-mutated MDS typically presents with anemia that could be macrocytic or normocytic. However, other cytopenias may occur. Single-lineage dysplasia is more common, but multilineage dysplasia is also seen, Dr. Mirza said. The bone marrow typically shows erythroid predominance with dyserythropoiesis, which can include megaloblastoid maturation, binucleation and/or nuclear segmentation, and basophilic stippling.
In Fig. 4 are left-shifted erythroids with increased ring sideroblasts, which are often more than 15 percent. SF3B1-mutated MDS typically has been thought to be associated with a good prognosis, “but that’s questionable,” Dr. Mirza said, “because depending on the IPSS [score], the same disease subclassification may fall into different risk scores.”
The International Working Group criteria include cytopenias, somatic SF3B1 mutation, morphologic dysplasia (+/-RS), and the bone marrow blast count (< five percent, and peripheral blood blasts < one percent). Response to luspatercept is good.
The IPSS risk score is a useful tool in determining the timing of transplant, Dr. Mirza said. “From a therapeutic perspective, when there is absence of cytopenias and you’re considering use of hypomethylating agents, the disease subclassification won’t help as much as the IPSS.” Incorporating the molecular data into the IPSS molecular model (IPSS-M) alters the transplant timing for patients compared with the traditional model. “If patients fall in a higher risk category, even though subclassification is the same, they would probably benefit from earlier transplant compared with those who fall into a lower risk category,” Dr. Mirza explained.
The two classifications and their different terminologies, he said, are largely an issue of semantics, because “biologically the disease and its ability to transform or progress and how we approach it by looking at transplant is the more important feature for cases.”
Amy Carpenter is CAP TODAY senior editor.

