October 2025—CAP Publications has released its newest book, Forensic Pathology: Principles and Pitfalls. It comes more than 20 years after the second edition of the CAP Handbook of Forensic Pathology was published. The book’s more than 900 pages comprise 36 chapters written by 57 contributors. CAP TODAY asked the following questions of the editors—Joseph Prahlow, MD, and Erin Brooks, MD—who led the publication of the comprehensive update.
Dr. Prahlow is professor, Department of Pathology, St. Louis University School of Medicine, St. Louis, Mo., and deputy chief medical examiner, Office of the Medical Examiner—City of St. Louis. Dr. Brooks is professor of pathology and vice chair of education, Department of Pathology and Laboratory Medicine, University of Wisconsin Hospitals and Clinics.
Below is the chapter “Deaths Due to Embolism,” written by Dr. Brooks and James Gill, MD, chief medical officer, Connecticut Office of the Chief Medical Examiner, and clinical professor, Department of Pathology, Yale School of Medicine.
Who is the targeted audience for your new book?
This work was produced in order to provide a comprehensive, up-to-date, well-illustrated, and thoroughly referenced forensic pathology textbook for use by any pathologist, whether they are in training or in practice or subspecialized in the area of forensic pathology. The book may also be suitable for persons who have an interest in or interaction with forensic pathology, including medical students, death investigators, other forensic scientists, and attorneys.
What would you like CAP TODAY readers to know about the content of this new edition?
We have attempted to produce a forensic pathology book that is as comprehensive as possible, bolstered by numerous color images as well as classic and up-to-date references for each chapter.
How is this book different from other forensic pathology books on the market?
A variety of excellent forensic pathology books are available. Each has its strong points and areas of excellence. Some are particularly excellent in terms of specific features, such as providing high-quality color images or numerous references or by addressing certain less common topics within forensic pathology. At the outset of this project, we decided to produce a book that takes the best aspects of each of the existing forensic books and combine them into one all-encompassing reference work for forensic pathology. In doing so, we have provided chapters written by multiple experts, complemented by a generous array of color photographs, high-content tables, numerous references, and a comprehensive look at all aspects of forensic pathology, including the traditional classic topics as well as many of the less common matters of importance.
What can you tell us about the many contributors to this book?
In selecting the authors for the various chapters, we attempted to strike a balance between preserving input from the outstanding “old guard” within the forensic pathology community and “newer” experts within our field. We have also included experts from within various ancillary forensic science disciplines. The conglomeration of authors, both new and old, makes it possible to provide an excellent overview of the many topics presented in this textbook.
In the new CAP book Forensic Pathology: Principles and Pitfalls is the following chapter 24, “Deaths Due to Embolism,” by Erin G. Brooks, MD, and James R. Gill, MD. (The chapter’s 75 references are not published here because of space limitations.) To order the book (PUB132), go to https://bit.ly/CAP_FPbook. The price is $192 for CAP members, $240 for nonmembers (ebook: $172).
Deaths Due to Embolism
Erin G. Brooks, MD
James R. Gill, MD
Introduction
Both thrombotic and non-thrombotic emboli may be encountered by the medical examiner at autopsy. While thrombotic embolism is far more common, non-thrombotic emboli also may be associated with sudden and unexpected death. Non-thrombotic emboli include air, fat, amniotic fluid, tumor, foreign material, and septic elements (e.g., bacterial or fungal organisms, portions of hydatid cysts). In addition to causing a sudden, unexpected death, both thrombotic and non-thrombotic emboli can result from trauma, therapy, or drug use, which also would make such cases reportable medicolegal deaths.
Thrombotic Emboli
While venous thromboembolism (VTE) can result in chronic complications due to impaired thrombus resolution including postthrombotic syndrome and chronic thromboembolic pulmonary hypertension, acute pulmonary thromboembolism (PTE) remains the primary cause of mortality. Approximately 20–25% of PTEs present as sudden death; it is estimated that annually there are 60,000–100,000 deaths in the US due to fatal PTE. Despite medical advances, the diagnosis of PTE frequently remains clinically unsuspected/undiagnosed prior to autopsy. Numerous hospital/nursing home autopsy-based studies have reported that in the majority of PTE deaths (i.e., 66–87%), patients had no distinctive heralding symptoms. While PTE predominantly arises in the adult population, it can occur at any age; for instance, fatal PTE accounts for an estimated 0.05–0.73% of deaths in the pediatric population.
Epidemiology
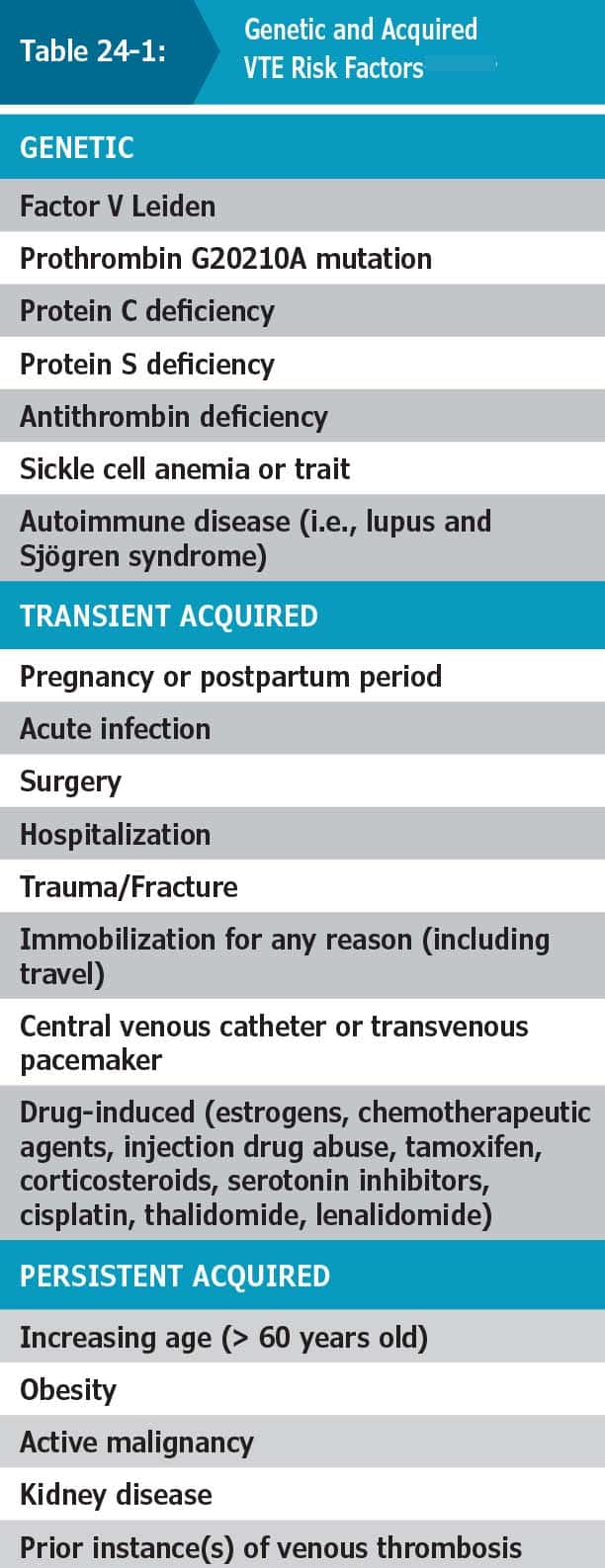
Fatal pulmonary thromboemboli are typically found more frequently in hospital (3.8–6.8%) than forensic autopsies (0.8–2.5%). This discrepancy is likely attributable to the greater number of PTE risk factors in the hospitalized vs. community populations. In roughly half of all cases, VTE is considered to have been “provoked” by transient acquired risk factors such as immobilization, trauma, surgery, or hospitalization in the preceding 3 months. Of the remaining cases, 20% are associated with persistent acquired risk factors (predominantly malignancy), and 30% are considered unprovoked with no clear risk factors. Overall, VTE appears to be a multifactorial disease. It has been associated with a variety of genetic and acquired factors, and while a single risk factor may not suffice to cause disease, additional acquired risk factors may synergistically interact to trigger thrombosis (Table 24-1). Molecular testing has allowed some thrombophilias to be detected both ante- and postmortem.
Incidence rates differ by age, race, and gender. Incidence is higher in older adults, black and white populations vs. other races/ethnicities (e.g., Hispanic or Asian), and men, except during reproductive years. Both pregnancy and the postpartum period confer an increased risk of VTE. Compared to non-pregnant women, the relative risk of VTE increases by 4-fold during pregnancy, and 21 to 84-fold in the postpartum period. Likewise, hormonal contraceptives and hormone replacement therapy can convey an increased risk. Injection drug use has also been associated with an increased risk of venous thromboembolic disease. Cigarette smoking is associated with a slightly increased risk for VTE; however, BMI may be a confounding factor in the risk estimates.
Pathophysiology
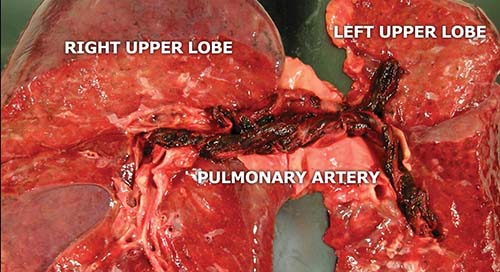
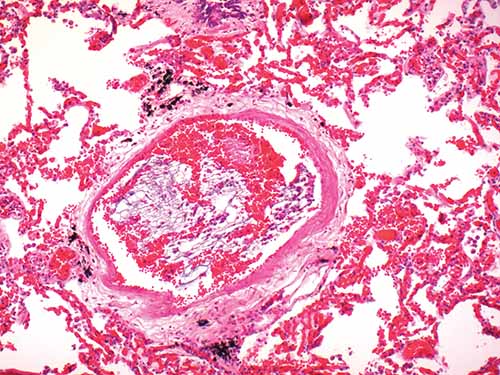
Venous thromboses typically form in the setting of hypercoagulability, blood flow stasis, and/or vascular endothelial damage (i.e., “Virchow’s Triad”). Once formed, thrombi may ultimately be lysed, organize with recanalization or resorption, propagate further, and/or embolize. While pulmonary thromboemboli can arise from any systemic vein, 70–90% of fatal PTE arise from deep venous thromboses (DVTs) originating in the legs or pelvis. Thrombi that propagate proximally into the inguinal/proximal leg veins are more likely to embolize to the lungs. In the adult population, lethal PTE tend to be large and central, occluding a main pulmonary artery or else forming a saddle thromboembolus (Figure 24-1.) By contrast, in the pediatric population, fatal PTE may cause a diffuse, peripheral (microscopic) embolization pattern rather than a large central embolus. Patients without preexisting cardiopulmonary disease typically require a pulmonary bed obstruction of at least 30-50% to develop pulmonary hypertension, though less obstruction may suffice in patients with advanced cardiopulmonary comorbidities. Pulmonary hypertension leading to right ventricular failure is considered the primary mechanism of death in these instances. An additional mechanism by which PTE may contribute to cause death is via ventilation-perfusion mismatch and global hypoxia.
Clinical Features
Clinical history suggestive of DVT includes lower extremity erythema, warmth in the area of thrombosis, unilateral leg pain/discomfort, or asymmetric limb swelling. While non-specific, clinical signs and symptoms that could suggest PTE include chest pain, dyspnea, tachycardia, and tachypnea. If the patient presents to medical attention prior to death, an elevated D-dimer level, computed tomography pulmonary angiography and/or ventilation-perfusion scans supportive of PTE, and echocardiography showing acute right heart overload can prove helpful—particularly in instances in which thrombolytic therapy was provided prior to death and PTE has thus been largely eradicated prior to autopsy. Thrombolytic agents work by activating plasminogen to form plasmin, which accelerates PTE lysis. In one study, 59% of patients infused with alteplase had moderate to marked PTE lysis as assessed by pulmonary angiography within 2 hours of treatment initiation. Heparin is used to treat DVT. Heparin does not lyse the thrombus but prevents propagation and allows the body to naturally lyse/resorb the thrombus.
Following an initial thromboembolic event, venous thrombosis commonly recurs. Impaired thrombus resolution or recurrence can lead to chronic complications such as postthrombotic syndrome (PTS) or chronic thromboembolic pulmonary hypertension (CTEPH). PTS is characterized by pain/cramps, edema, and heaviness in the affected limb; it can progress to cutaneous ulceration. Roughly 4% of patients will develop CTEPH within 2 years of an initial PTE event; CTEPH is characterized by pulmonary arterial fibrotic stenosis typically involving central pulmonary arteries. If untreated it can progress to right ventricular failure and death.
Autopsy
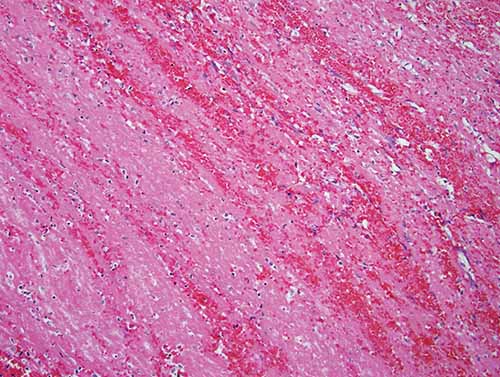
Careful assessment of gross features of PTE can help distinguish them from postmortem clot. Classic central PTE typically bulge outward from the main pulmonary arteries. Unlike postmortem clots, they may be slightly adherent to the wall of the pulmonary artery and may have a folded pattern. The majority are acute and have a red-purple color. Serial sectioning of the PTE can be helpful in revealing swirling layers of fresh red blood cells alternating with pink layers of platelets and fibrin (i.e., Lines of Zahn), indicating the PTE formed in flowing blood (Figure 24-2). Marks of venous valves from deep leg veins may be apparent on the PTE. While the central PTE does not typically assume the exact configuration of pulmonary arterial branches, prosectors should be aware that postmortem clot may form adjacent and distal to the PTE giving it the gross external appearance of a postmortem clot. Photographic documentation of PTE is advised. When dissecting the lungs, the pulmonary arterial tree is opened to demonstrate the extent of the PTE. Sectioning of pulmonary parenchyma may disclose smaller peripheral thromboemboli, which tend to protrude prominently from vessels above the cut surface of the lung.
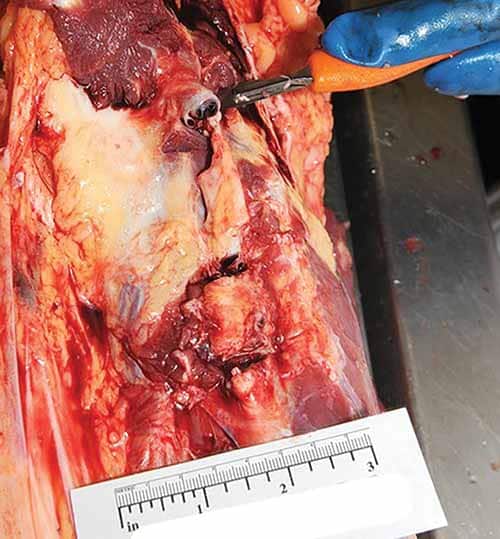
Dissection of deep veins of the pelvis and both legs is advised in all instances of PTE. This allows the pathologist to assess the causal relationship between recent events such as trauma or immobility and whether a therapeutic complication or inadequate medical care played a role. In addition, previously undocumented conditions (e.g., phlebitis, injection drug use with vascular sclerosis), which may be the underlying cause for the thrombosis, can be diagnosed. While external examination may reveal findings supportive of lower extremity DVT such as erythema and asymmetric limb swelling, lack of such findings does not exclude it. With the body in a supine position, the iliac and femoral veins can be easily accessed and dissected down the medial anterior thighs bilaterally. With the body in the prone position, a medial incision from the popliteal fossae to the ankles allows for access and dissection of the posterior tibial, soleal, and popliteal veins (Figure 24-3). Detection of thrombi in cases of PTE are reported to be particularly high in the soleal veins. Histologic sections of DVTs can be informative and should include the adjacent vein wall and muscle. Rarely, venous thromboses may start in other sites including the periprostatic/perivaginal veins, the upper extremities, the superior vena cava with invasive procedures, and the right atrium/ventricle.
Histology and Ancillary Testing
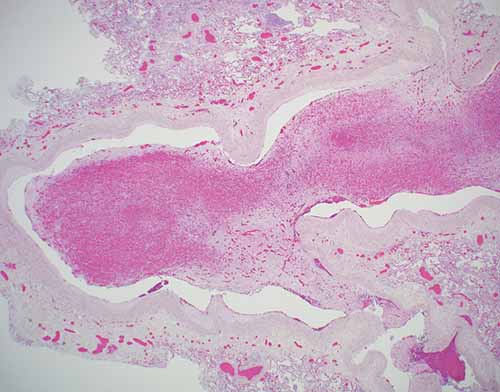
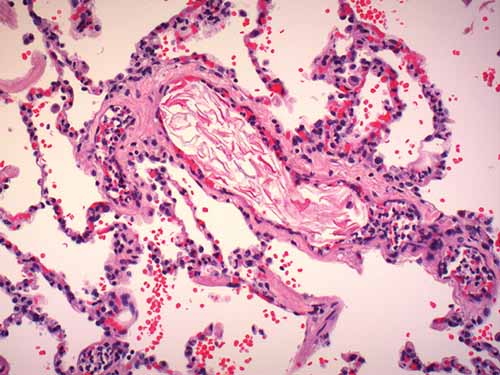
Histologic sections of thromboemboli are advised. While there may be thrombi of varying ages within the same vein, determining the oldest age is typically of most importance in medicolegal cases. Examination of the thrombo-endothelial junction can yield particularly helpful information such as whether there is hemosiderin, endothelial proliferation, collagen deposition, or neovascularization. While a variety of systems have been proposed to help date thromboemboli, caution is advised. There can be individual variation which precludes exact age determination. Providing a general categorization of thromboemboli as acute, organizing, or remote and opining as to whether this categorization seems consistent with a given clinical history may, however, be possible. Immunohistochemical stains for platelets (e.g., CD61), macrophages (e.g., CD68), leukocytes (e.g., CD45), neutrophils (e.g., CD15), and fibrinogen may be helpful in aging thromboemboli. Acute thromboemboli can be roughly characterized as having Lines of Zahn with no visible reaction between the endothelium and thrombus, and agglomeration of erythrocytes; the age is typically < 7 days. Organizing thromboemboli demonstrate fibroblast proliferation, endothelial budding, fibrinous transformation, and hemosiderin-laden macrophages; the age is roughly considered to be between 2–8 weeks (Figures 24-4 and 24-5). Remote thromboemboli are largely hyalinized with recanalization and are generally considered to be older than 2 months.
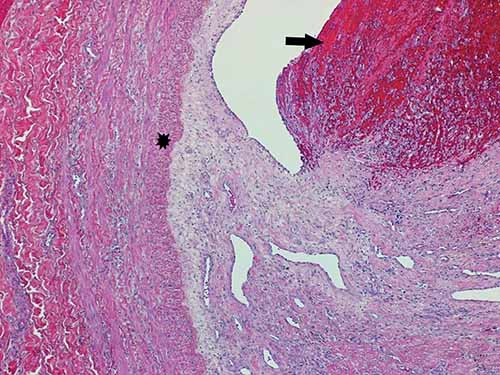
Ancillary testing for hereditary thrombophilias can be important in young decedents, decedents without clear risk factors, or those with a family history of thrombosis who die of PTE. Such genetic testing can be critical in elucidating the cause of death as well as informing living relatives of increased thrombosis risks they may face. Factor V Leiden is the most common of the hereditary thrombophilias, found in up to 21% of individuals with DVT. Other less common thrombophilias incude methylenetetrahydrofolate reductase (MTHFR) mutation, the G20210A mutation in prothrombin, antithrombin deficiency, and protein C and protein S deficiencies. A mutation in MTHFR can result in hyperhomocysteinemia, which may increase risk for thrombosis. Molecular testing is routinely used to identify the prothrombin and factor V Leiden mutations.
Nonthrombotic Emboli
Gas Embolism
Etiology
While gas embolism into arterial or venous structures may arise due to traumatic injury or scuba-related decompression sickness/pulmonary barotrauma (see Chapter 14 for further details regarding scuba deaths), most cases are iatrogenic. A wide array of common medical procedures including radiologic contrast administration, hemodialysis, laparoscopic surgery, and central line manipulation may result in gas embolism. The exact incidence is difficult to quantify as many instances are likely subclinical; for instance, 23% of patients undergoing intravenous contrast injection for CT scanning were found to have incidental air emboli in larger intrathoracic veins. Although a variety of gases including carbon dioxide, nitrogen, and nitrous oxide are used in medical procedures, air is by far the most commonly embolized gas. Gas emboli are typically categorized as either venous or arterial, with each having a distinctive etiology and clinical sequelae.
Venous Gas Embolism
Introduction of air into the venous system requires both a communication between the vein and atmosphere and a favorable pressure gradient; the amount and rapidity of gas infusion, body position, and underlying comorbidities all determine the likely lethality of the infusion. Infused volumes of air as low as 200–300 mL are sufficient to cause death. However, any volume >50 mL may be lethal if infused more rapidly or in patients with advanced underlying cardiorespiratory disease and compromised hemodynamics. Small bubbles of air infused into veins embolize to the pulmonary circulation where they may strain the right ventricle, resulting in acute cor pulmonale or asystole. Larger air volumes may interfere with the pulmonary outflow, preventing blood flow through the right heart. Medical procedures in which there is vascular elevation above the level of the heart (e.g., neurosurgery with patient in the seated position or obstetric/gynecologic surgeries with patient in the Trendelenburg position) have higher risks of venous gas embolism. The initial clinical manifestations of venous gas embolism are non-specific but may include syncope or altered mental status, cough, acute dyspnea with gasping for air, tachypnea, and chest pain.
Arterial Gas Embolism
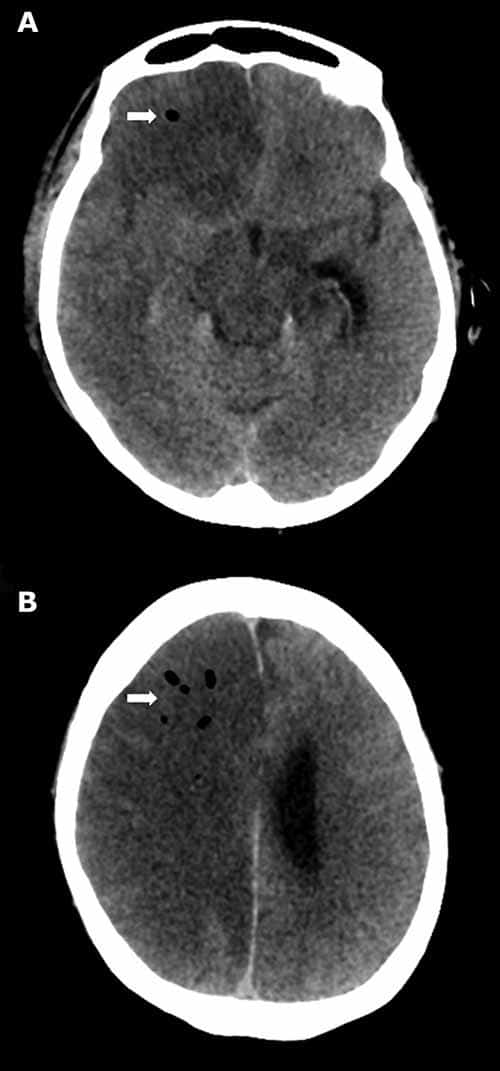
Arterial gas embolism is less common, but more poorly tolerated than venous gas embolism. While air may be introduced primarily into the arterial system, all venous gas emboli have the potential to secondarily involve the arterial system (i.e., paradoxical emboli). Paradoxical emboli may arise via intracardiac shunts (e.g., probe patent foramen ovale, atrial septal defect), intrapulmonary shunts (e.g., arteriovenous malformation or fistula), or by simply overwhelming the normal physiologic mechanisms preventing arterial embolism (e.g., filtration by pulmonary capillaries). Arterial gas emboli may obstruct end arteries; while gas emboli to viscera or skeletal muscles are generally well tolerated, the heart and brain are particularly vulnerable to hypoxia. Gas embolization to coronary arteries may cause ischemia/infarction, dysrhythmias, cardiac failure, or arrest. Cerebral gas embolization may result in stroke, seizures, unconsciousness, and impairment of the respiratory/circulatory centers (Figure 24-6).
Autopsy
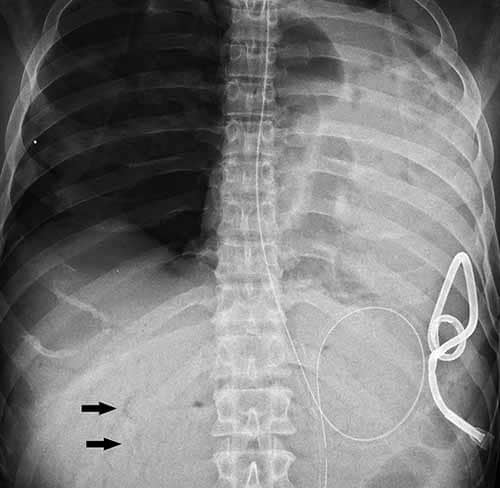
If gas embolism is suspected at autopsy, radiography or CT prior to prosection is strongly encouraged. Scenarios that may suggest gas embolism are recent traumatic injury to the thorax, head, or neck, sudden death following a therapeutic or surgical procedure, and recent delivery of child or abortion. External examination findings supportive of fatal air embolism include subcutaneous emphysema and crepitance. There may additionally be marbling, congestion, and/or petechiae that is particularly prominent superiorly (e.g., in the skin of the face, neck, or chest). Chest radiography is not as sensitive as CT and can appear normal in cases of smaller venous gas embolism; with larger volumes, radiolucency (gas) may be appreciated in the main pulmonary artery, right heart (the so-called “C” sign), and hepatic veins (Figure 24-7). CT, by contrast, affords for detection of even small amounts of gas in soft tissues or anatomic cavities. As decomposition can lead to postmortem gas production soon after death, familiarity with typical postmortem gas distribution patterns and/or gas chromatographic analysis may prove necessary in order to distinguish antemortem gas embolism.
In instances in which postmortem radiology is unavailable, “Richter’s Technique” provides an alternate means of assessing for gas embolism. In this method, prior to opening the calvarium or cutting any arteriovenous neck or subclavian vessels, intracardiac air is assessed. The sternum is cut at the level of the 2nd ribs and the thoracic plate removed exposing the pericardial sac. The sac is opened, filled with water, and the right ventricle or pulmonary artery is punctured with leaking bubbles indicating intracardiac gas. An aspirometer can be used for gas volume measurement or subsequent gas chromatographic analysis in medicolegal cases as necessary. In addition to intracardiac air, internal examination in cases of gas embolism may be notable for grossly apparent gas bubbles in the superficial vessels of the mesentery, brain, and other tissues. On opening larger vessels such as femoral veins, pulmonary arteries, and vena cava, the blood may have a “carbonated” appearance with a brighter red coloration than usual. Histologic sections may reveal round “vacuoles” obstructing vessels.
Fat Embolism
Etiology
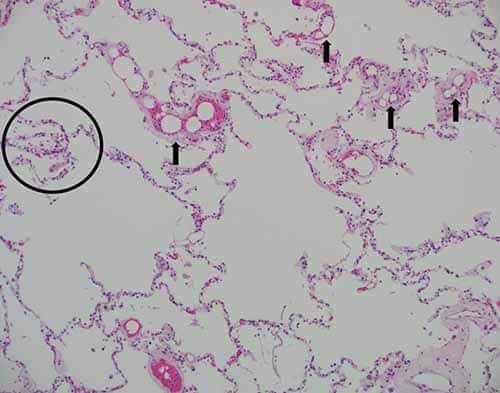
Fat embolism refers to fat globules in the bloodstream. The release of fat and bone marrow fragments into the pulmonary circulation is an exceedingly common finding at autopsy following cardiopulmonary resuscitative efforts (i.e., histologically detected in 88% of cases). It is suspected that such fat likely embolizes into the vasculature from resuscitative rib/sternal fractures or from contused subcutaneous thoracic adipose tissue. While most commonly associated with traumatic bone injury or injury/disease of adipose tissue (e.g., liposuction, lipoinjection, panniculitis, acute fatty liver), conditions resulting in osteonecrosis (e.g., osteomyelitis, bone tumor lysis, sickle cell hemoglobinopathies) may also cause fat embolism. Although the majority of fat emboli are subclinical, rarely globules may obstruct pulmonary capillaries causing respiratory symptoms, or enter the systemic arterial circulation embolizing to the brain and other sites. Embolized fat may be degraded into glycerol, free fatty acids, and other toxic intermediaries causing a proinflammatory state and end-organ dysfunction. The potentially lethal clinical manifestations that may develop are known as fat embolism syndrome.
Fat Embolism Syndrome
Fat embolism syndrome (FES) develops following an insult resulting in the release of fat into the circulation. It is classically characterized by hypoxemia, neurologic manifestations, and petechiae arising within 12–72 hours of the insult and has an up to 10% mortality rate. Patients may not develop the triad of classical symptoms, however; in one series, hypoxemia was most common (96%) followed by neurologic abnormalities (59%) and petechial rash (33%). Neurologic abnormalities typically arise following hypoxemia and can range widely from confusion and lethargy to seizures and coma. Other minor diagnostic criteria that may suggest the diagnosis of FES are fever, tachycardia, retinopathy, and unexplained anemia or thrombocytopenia. As the disease course can be fulminant, radiologic/laboratory findings are non-specific, and patients may not fully manifest the classic triad of symptoms, the diagnosis of FES may not be made until the postmortem examination.
Autopsy
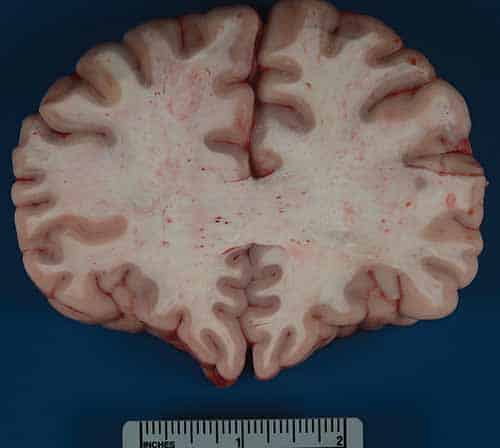
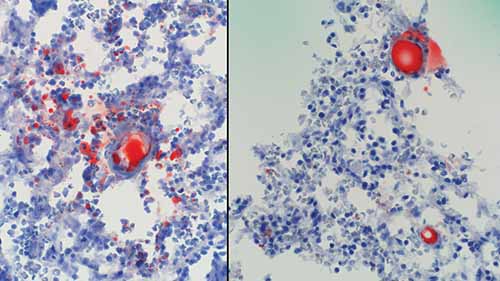
In cases of FES, external examination may be notable for a petechial red-brown rash involving the head, neck, anterior thorax, axillae, and/or conjunctivae. On internal examination, pulmonary and cerebral petechiae may be apparent. The central nervous system petechiae are typically most pronounced in the white matter (Figure 24-8) and spinal cord. A thorough histologic sampling is advised. While subclinical resuscitative fat emboli are frequently seen in the pulmonary vasculature, widespread systemic involvement (e.g., brain, skin, kidney) may suggest FES. Histologic sections of the rash may show round to ovoid vacuoles occluding small dermal capillaries with resultant erythrocyte extravasation, and similar intravascular vacuoles can typically be seen in multiple tissue types (Figures 24-9 and 24-10). To distinguish whether vacuoles represent fat versus air emboli, confirmatory fat stains can be performed. Lipids are dissolved by standard tissue processing solvents; however, stains including oil red O (stains fat red), Sudan black (stains fat black), or Sudan III (stains fat orange) can be performed on frozen section tissue (Figures 24-11 and 24-12). Alternatively, post-formalin tissue fixation methods involving osmium tetroxide or linoleic acid/chromic acid can be employed.
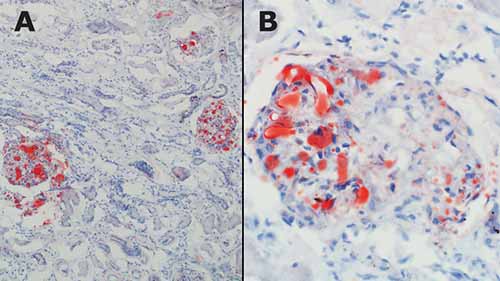
Amniotic Fluid Embolism
Etiology
Amniotic fluid emboli are thought to enter the maternal circulation via a defect in the maternal/fetal interface, likely at the level of the endocervical veins, placental attachment site, or site of uterine trauma. Traditionally, it has been proposed that a pressure gradient favoring the entry of amniotic fluid should exist such as strong uterine contractions during labor. Components of embolized amniotic fluid may include epithelial squamous cells sloughed from fetal skin, mucin or bile pigment excreted from fetal intestine as meconium, adipose from the vernix caseosa, and lanugo hairs (Figures 24-13 and 24-14). Amniotic fluid emboli (AFE) are largely innocuous; the passage of fetal tissue into maternal vasculature at delivery appears almost ubiquitous, and arterial catheterization studies have shown that 20%–100% of pregnant women without syndromic manifestations have squamous cells in their pulmonary arterial circulation.
Amniotic Fluid Embolism Syndrome
Rarely—i.e., in approximately 1 in 40,000 births—AFE syndrome may develop. The presentation is typically abrupt and rapidly progressive with an up to 60% maternal mortality rate. Perinatal mortality estimates range from 9–44%. Diagnostic criteria include the sudden onset of hypotension with respiratory compromise or cardiorespiratory arrest followed by DIC in the absence of fever. The syndrome typically arises during labor or within 30 minutes of placenta delivery; individual cases have also been reported following medical/surgical abortion, amniocentesis, amnioinfusion, and blunt abdominopelvic trauma. Unlike fat, air, or thrombotic emboli which cause morbidity/mortality primarily via mechanical obstruction of circulation, the pathogenesis of AFE syndrome is thought to be attributable to a pathologic inflammatory response; the maternal response to antigenic fetal material results in endogenous inflammatory mediator release which secondarily affects cardiopulmonary function and activates the coagulation cascade. As the diagnosis is based on the patient’s clinical presentation and is essentially one of exclusion, autopsy should be encouraged.
Autopsy
The gross findings with AFE syndrome are usually non-specific but may include pulmonary edema or congestion, atelectasis, or hyperinflation. Stigmata of DIC can be seen, although thrombi in the main pulmonary arteries or heart is atypical. Careful examination of the uterus and placenta should be performed. While not diagnostic of AFE syndrome, histologic detection of AFE can certainly provide helpful supportive evidence. Submission of multiple pulmonary histologic sections may increase likelihood of detection as AFE tend not to be uniformly distributed. Other organs/tissues in which AFE have been detected include uterus/cervix, heart, liver, spleen, kidneys, adrenal glands, pancreas, and brain. Special stains and immunohistochemical markers that may prove helpful in detecting AFE include cytokeratin AE1/AE3 (squamous epithelial cells), oil red O on frozen tissue (adipose), Alcian blue PAS or TKH-2 (mucin). Some studies have suggested that AFE syndrome may represent an anaphylactoid reaction with elevated postmortem serum tryptase levels being supportive evidence; however, other studies have found that serum tryptase has poor sensitivity for AFE syndrome diagnosis.
Septic Embolism
Etiology
Septic emboli are typically bacteria (or rarely fungi or other organisms) admixed with thrombus that have detached from their source and traveled through blood vessels to a different location. Septic emboli may result in two types of pathology: ischemic injury and/or infectious spread, which may include abscess formation.
The most common sources for septic emboli are infected cardiac valves (infective endocarditis), implanted medical devices (e.g., stents, catheters, ports, prosthetic valves), and septic pelvic thrombophlebitis (e.g., post-puerperal uterine infection). A bacteremia typically initiates the process. The bacteremia may result from intravenous injection, dental or surgical procedures, local infections, and malignancies.
Bacterial Endocarditis
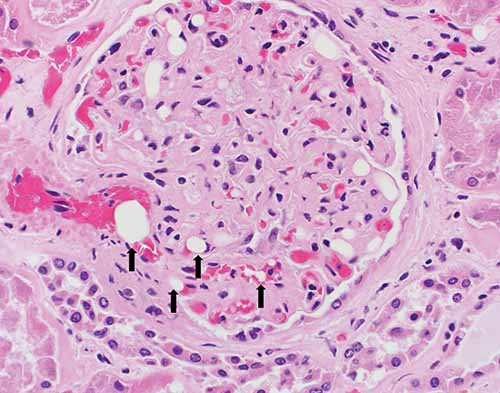
Patients with underlying cardiac valvular disease (e.g., myxomatous mitral valve, degenerative calcific aortic stenosis, rheumatic disease) may be predisposed for this complication but it also occurs with normal heart valves. Staphylococcus aureus is a highly virulent organism and may infect a normal heart valve producing necrotizing lesions. Less virulent bacteria (such as Streptococcus viridans) more commonly infect abnormal/diseased valves and follow a more prolonged and insidious course. Endocarditis may result from bacteremia due to an invasive colonic adenocarcinoma. Detection of intestinal bacteria, such as Streptococcus bovis, may be a clue to the site of the originating infection (Figure 24-15).
Differential Diagnosis
The differential diagnosis of endocarditis and septic emboli includes non-infectious vegetations such as nonbacterial thrombotic endocarditis (i.e., NBTE or marantic endocarditis), which may be seen in hypercoagulable states including malignancy, or Libman-Sacks endocarditis seen with antiphospholipid syndromes (e.g., systemic lupus erythematosus). It also includes metastatic disease with tumor emboli (clusters of malignant cells lodged in the vessel), disseminated fungal or mycobacterial (miliary) infections which may involve multiple organs, talc/microcrystalline cellulose granulomatosis in the lung vasculature (e.g., from the injection of crushed pills), and fat embolism (see above section on fat embolism for further details).
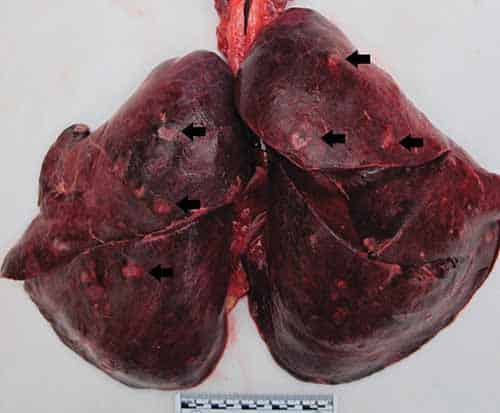
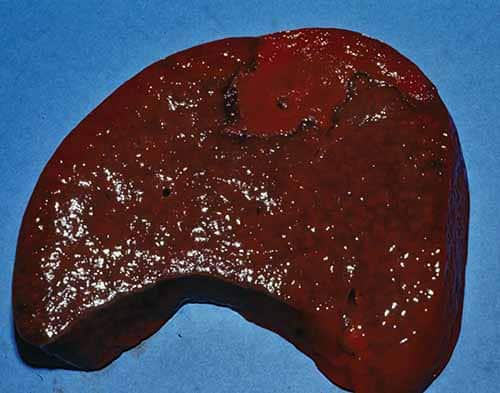
Autopsy
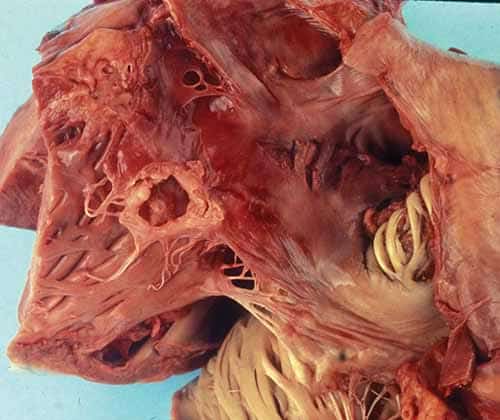
At autopsy, one may see a wide distribution of lesions involving multiple organs. The prosector should look for both the origin (heart valve, medical device) as well as the end organ damage from the embolism (Figure 24-16). Endocardial vegetations are friable tan-blue–red lesions that may erode or ulcerate the cardiac valve. Microscopically, they are a mixture of fibrin, inflammatory cells, and bacteria. The infection may involve the adjacent myocardium causing a “ring” abscess. Embolization from right-sided cardiac vegetations (i.e., tricuspid and pulmonary valves) typically involve the lungs causing a multifocal inflammatory process. Rarely, a patent foramen ovale (PFO) or atrial septal defect may allow the embolism to gain access to the systemic circulation. Left-sided endocarditis vegetations embolize to the arterial circulation which may cause end-organ damage (e.g., brain, spleen, kidney, colon) including infarcts, such as cerebral strokes (Figure 24-17). In the embolized tissue, histologically, there may be fibrinoid necrosis of the vessel with neutrophilic infiltrates in the vessel wall and perivascular tissues, infarction, and/or hemorrhage. Common locations include:
- Brain: Septic cerebral emboli may be silent clinically or may result in ischemic strokes and/or brain abscesses both causing neurological deficits based on the location and extent of the damage.
- Spleen and kidneys: Septic emboli may result in infarcts, hemorrhage, and/or abscess. The spleen is a common abdominal site for systemic septic emboli.
- Coronary artery: Septic coronary artery embolization may occur with aortic valve endocarditis and result in a myocardial infarct.
- Mesenteric vessels: Septic embolic occlusion of the major mesenteric arteries may result in segmental colonic infarcts.
- Pylephlebitis: Portal vein thrombosis due to intra-abdominal infection (e.g., cholangitis or diverticulitis) and liver abscesses also may be seen.
- Periphery and skin: Septic emboli may cause acute extremity ischemia that may lead to infarction with the need for limb amputation. Septic microemboli of the skin result in subtle skin lesions (Janeway lesions of the palms and soles, or Osler nodes in the fingers).
- Neck: Lemierre syndrome is an acute oropharyngeal infection (most frequently caused by Fusobacterium necrophorum) with secondary septic thrombophlebitis of the internal jugular vein that initially arises from a peritonsillar infection leading to a local septic thrombophlebitis and then septic emboli (Figure 24-18).
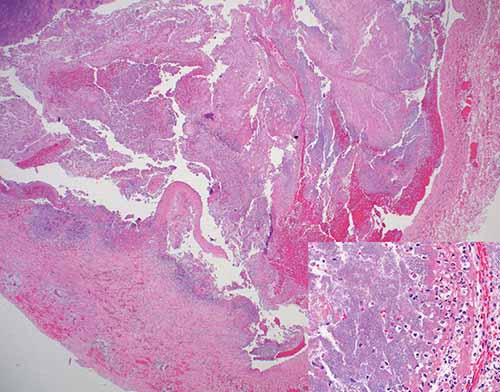
Figure 24-18 Lemierre’s Syndrome: septic thrombus within right internal jugular vein (H&E, 20×). Inset: Alternating bacterial colonies, fibrin lines, and fresh blood within thrombus (H&E, 400×).
Certification
These end-organ injuries should not be misconstrued as the underlying cause of death. These are immediate causes while the underlying cause is the disease or injury that started the infectious process. For example, a cerebral infarct due to S. aureus endocarditis of the aortic valve in a chronic intravenous drug user should list the chronic intravenous drug use as the underlying cause of death.
Conclusion
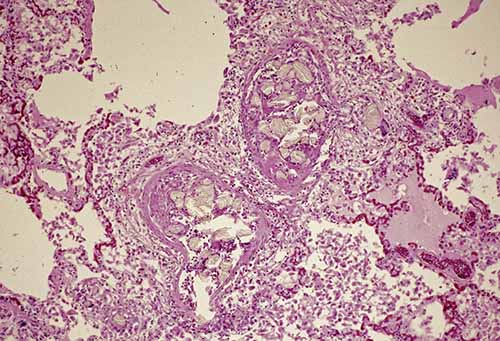
While the topics covered above represent the major thrombotic and non-thrombotic emboli of significance to pathologists at autopsy, any endogenous or exogenous substance gaining access to the vasculature has the potential to embolize. Additional examples of endogenous emboli include tumor emboli and atheroemboli, while exogenous emboli also include silicone embolism, bullet/shotgun pellet emboli, food embolism, and talc/microcrystalline cellulose granulomatosis (Figure 24-19). Many emboli represent differential diagnoses for each other, and certain embolization syndromes are diagnoses of exclusion. Careful assessment of vasculature grossly and microscopically is advised, with utilization of ancillary testing as indicated.
Pearls and Pitfalls
- Dissection of deep veins of the pelvis and both legs is advised in all instances of PTE. Ancillary testing for hereditary thrombophilias can be important in young decedents, decedents without clear risk factors, or those with a family history of thrombosis who die of PTE.
- Gas emboli are most commonly iatrogenic in nature. In cases with a suggestive clinical history, radiography or CT is strongly encouraged prior to prosection.
- FES is characterized by the clinical triad of hypoxemia, neurologic manifestations, and petechiae. While lipids are dissolved by standard tissue processing, fat stains can be performed on frozen or formalin-fixed tissue.
- AFE are neither pathognomonic nor required for the diagnosis of AFE syndrome. AFE syndrome typically occurs during labor or within 30 minutes of placenta delivery.
- With arterial septic emboli, end-organ injuries represent immediate causes of death while the underlying cause is the disease or injury that started the infectious process.
- Any endogenous (e.g., tumor emboli) or exogenous (e.g., bullet emboli) substance gaining access to the vasculature has the potential to embolize.

