Summary
Two new ADLM documents provide guidance on toxicology testing for emergency departments, replacing a 2003 guideline. The documents address test menus, turnaround times, and the importance of educating ED staff on assay limitations. Recommendations include a standard urine drug screen menu, reporting practices, and considerations for point-of-care testing and oral fluid testing.
Amy Carpenter
November 2025—Guidance on how best to meet the emergency department’s toxicology testing needs fills two ADLM documents that are intended to replace a practice guideline published in 2003.
The guidance document on laboratory testing for drugs of misuse to support the ED will be published soon. The second document, guidance on laboratory support for targeted and comprehensive toxicology testing for the ED, will soon be available for public comment.
Speakers at the ADLM meeting in July provided a preview of both.
“Similar to the 2003 document, we address test menu, turnaround times, cutoffs, reporting for clarity, and the importance of educating ED staff on the assay limitations—what we can and can’t detect,” said Stacy Melanson, MD, PhD, coauthor of the drugs of misuse document and division chief of clinical pathology at Mass General Brigham. The guidance is centered on urine-based testing, with limited discussion of other matrices, and immunoassays, point-of-care testing, and mass spectrometry.

Urine drug testing that should be available to all EDs, ordering indications, immunoassay pros and cons, and the role of mass spectrometry are some of what’s covered.
Out of scope is forensic-related testing, including chain of custody. “While they are toxicologically relevant, they are usually not available in the emergency department,” Dr. Melanson explains. Also out of scope is blood alcohol.
Hospital-based U.S. laboratories are the targeted audience, and all recommendations are based on extensive literature searches and expert opinion.
“Primarily, these tests are being ordered on trauma and behavioral health patients,” Dr. Melanson said, and typically the results have a greater impact on downstream care—where patients will go, how they’ll be managed, what post-procedure outcomes might be—than on immediate ED care.
The authors agreed the term drugs of abuse needs to be replaced, Dr. Melanson said, adding, “It’s pretty standard but also stigmatizing.” For now, the term is drugs of misuse.
“Urine is the preferred matrix,” she said, despite its limitations, among them that results can be positive for several days and thus might not explain the acute situation. The guidance includes recommendations on definitive testing by mass spectrometry, as well as the need for collaboration with medical toxicologists and poison control centers.
In Fig. 1 is the recommended urine drug screen menu. “We are recommending basically six drugs on your urine drug screening panel that should be available in all hospital laboratories,” Dr. Melanson said: amphetamines, benzodiazepines, cocaine metabolite, fentanyl, opiates, and oxycodone. Amphetamines were considered optional in the 2003 guideline. “If it’s relevant to your population and your ED, you can consider 6-acetylmorphine, barbiturates, buprenorphine, cannabinoids, and PCP.” The authors recommend not testing for propoxyphene and tricyclic antidepressants.
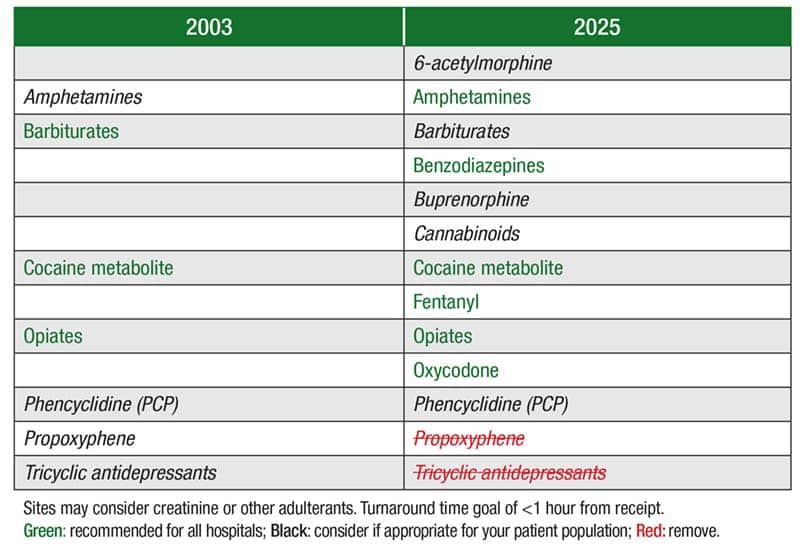
Laboratories may consider using urine creatinine or other adulterant testing, Dr. Melanson said, and a turnaround time goal of less than one hour from receipt in the laboratory is recommended.
Brigham and Women’s Hospital and Massachusetts General Hospital adopted the recommended menu in October 2024 and July 2022, respectively.
At Mass General, barbiturates and cannabinoids were removed from the panel (they can be ordered individually), and fentanyl and oxycodone were added to the panel. “After they removed barbiturates and THC, the volume decreased significantly, with basically no individual orders for barbiturates, suggesting it probably wasn’t clinically useful.” There were individual orders for cannabinoids, some from the ED and some from transplant.
Fentanyl and oxycodone volumes increased to match the urine drug screening panel. Previously, both had been available as part of a substance use disorder panel, not the panel typically ordered in the ED.
Fentanyl positivity decreased compared with when it was on the substance use disorder panel, likely owing to the ED patient population abusing fentanyl or being prescribed fentanyl at lower rates.
THC positivity was variable, likely owing to the low volume of test orders post-implementation.
Before the revised menu was implemented in 2024, Brigham and Women’s Hospital had eight drugs on its panel: amphetamines, benzodiazepines, cocaine, fentanyl, methadone, opiates, oxycodone, and THC. The removal of methadone and THC from the panel (then available individually) was followed by a significant drop in methadone and THC orders, similar to what was seen at Mass General. At Brigham too, the orders for THC testing came from the ED and transplant.
Benzodiazepine positivity increased significantly in both hospitals. “This is because both of us at different times went live with the new Roche benzodiazepine assay, which has better cross-reactivity for lorazepam and clonazepam and metabolites,” Dr. Melanson said.
She and her colleagues discovered that finding in a study they did to verify the clinical performance of the new qualitative Roche Benzodiazepines II (BNZ2) immunoassay, and to compare its performance to the Roche Benzodiazepines Plus (BENZ) assay in two patient populations: urine drug screening in the ED and compliance monitoring (Tolan NV, et al. J Mass Spectrom Adv Clin Lab. 2024;33:14–20). They observed higher clinical sensitivity and a lower false-negative rate with the BNZ2 assay in both populations because of its improved cross-reactivity for lorazepam and clonazepam metabolites. “The new assay [BNZ2] picked up an additional 35 specimens with lorazepam metabolites because there’s beta glucuronidase in the assay,” she said. Clonazepam use was detected in an additional 11 samples for the same reason.
“We as laboratorians need to constantly be looking at what available immunoassays are out there, their analytical performance, and which assays may provide better data for our emergency room,” Dr. Melanson said.
To report urine drug screening results, the recommendation is to report qualitatively, using with a positive result the words presumptive, preliminary, or unconfirmed positive to emphasize the possibility of false-positive results.
The authors also recommend flagging positive results as abnormal, “even though a positive result could be normal in someone who is prescribed that drug,” Dr. Melanson noted. A flag will ensure clinicians will be notified or see it more clearly in the electronic health record.
The recommendation for specimen storage is 48 to 72 hours for negative samples and an extended time period for all positive and pediatric specimens.
The authors also recommend adding either a generic or test-specific comment to make clear the limitations of immunoassay screens in the ED patient population, and they provide this example: “This screening test was performed using immunoassay technology, which may provide false-positive or false-negative results. For questions or to request confirmatory testing, contact the laboratory.”
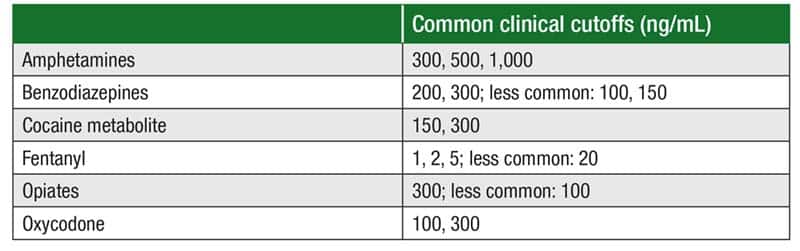
In Fig. 2 are the six recommended metabolites and common clinical cutoffs. “Appropriate cutoffs, comments, and guidance should be discussed with your emergency department,” Dr. Melanson noted.
To ensure accurate interpretation of results, targeted education and stronger collaboration between laboratory experts and health care professionals may be needed, as revealed in a study Dr. Melanson and coauthors did (Snozek CLH, et al. Am J Clin Pathol. 2025;163[1]:69–79; also CAP TODAY, May 2025, page 1, “Misses, hits in urine drug test interpretation”).
Dr. Melanson provided an implementation checklist—“not specific to urine drug screening,” she said, “but things to think about when adding an immunoassay.”
No. 1 is to consult clinicians: “Do you think based on our local needs we would want to implement more than the standard six urine drug screens?”
No. 2 is the budget impact, whether it’s instrument related, staffing, space, plumbing, electrical, construction, or service and maintenance consumables.
Information systems is No. 3: additional licenses to pay for, hardware, middleware, rules based on the urine drug screens, a new server, or remote diagnostics. “And it needs to be built in the EHR and LIS, and they need to communicate.”
No. 4 is validation, including not only sensitivity and specificity but also reporting results, establishing cutoffs, whether to flag, whether to add comments, and determining stability, rejection criteria, and the interferences that should be called out.
Billing is on the checklist, too—“a necessary evil,” as Dr. Melanson put it. “There’s usually a lack of communication and understanding between who is doing the billing and who is providing CPT codes,” she said. “So this is something to consider when bringing on a new test.”
Also on the list: determining the impact to other network sites and reviewing standard operating procedures and training. And among the many clinical requirements to consider are turnaround time, specimen storage, and automated reflex versus requests for confirmation.
For most drugs of misuse, analytical performance at the point of care is comparable to that of laboratory-based testing. “It also decompresses the central laboratory,” Dr. Melanson said, and makes it possible for patients to see there was no specimen mixup.
“But there are disadvantages to point of care, too, many of which are the reasons we do not do it,” she said. For one, the test menu may be lacking. “It’s also subjective interpretation; the absence of a band is positive and you’re requiring usually a nonlaboratorian to interpret that.” Typically, POC testing requires manual result entry in the LIS, and “billing can be tricky.”
“And one of our main drivers for not doing point-of-care testing are the regulatory and compliance challenges of having nonlaboratorians do this, whether waived or nonwaived,” Dr. Melanson said.
Oral fluid for drugs of misuse testing is “pretty good for 6-AM and amphetamine,” Dr. Melanson said.
“It’s not as easy to adulterate oral fluid and resists poppyseed products and overhydration.” It’s noninvasive and nonintrusive, and can detect the parent and metabolite in real time.
Its disadvantages are that the small specimen volume restricts the number of analyses and confirmations that can be performed, and oral contamination (drugs, suspensions, elixirs, food and drink) can affect results. It can also be difficult to collect in patients who abuse stimulants.
Oral fluid is suboptimal for benzodiazepines, buprenorphine, and THC. Dr. Melanson and colleagues compared the clinical utility and analytical performance of oral fluid and urine as matrices for detecting common benzodiazepines and opioids (Petrides AK, et al. Clin Chim Acta. 2018;481:75–82). They found that oral fluid is the preferred matrix for 6-AM while urine is preferred for 7-aminoclonazepam, lorazepam, oxazepam, hydromorphone, and oxymorphone. Oral fluid should be considered, they wrote, if adulteration risk is high and the use and/or misuse of benzodiazepines, hydromorphone, and oxymorphone is low.
“It’s kind of a balance between the different advantages and understanding the performance and your positivity rates for different drugs,” Dr. Melanson said.
Mass spectrometry is a central question in the document, but Dr. Melanson distilled its essence to the following: “We recommend it if the patient’s history is unavailable or unreliable, if the patient has a presumptive positive result for an immunoassay that has broad cross-reactivity—e.g. amphetamines or benzodiazepines—when a drug in question is not detected by immunoassay—e.g. novel or designer drugs—and if the result will change patient management and inform follow-up care.”
As to future directions, the authors cite several, one of which is more robust educational tools. “We need to do better at educating,” she said. In addition, “We need to potentially look at alternative matrices,” such as blood. “And should we define algorithms and not leave it up to clinicians?” Currently, there is no reflexing of immunoassay to mass spectrometry for several reasons, she said, among them: “If the patient is discharged, is it really useful?” But the other question is whether the laboratory should define when mass spectrometry should be used in the ED.
Immunoassays are slow to change and update and make available, especially when FDA approval is required, Dr. Melanson said. “So we’re advocating for available, affordable, accurate, and rapid mass spectrometry so we can have more definitive testing that gives us a turnaround time that’s acceptable.” If that should become available, then adequate reimbursement will be the challenge.
The final future direction: a greater international perspective as the recommendations are updated in the years ahead.
Patrick Kyle, PhD, professor of pathology and and director of the clinical and special chemistry and toxicology laboratories at the University of Mississippi, introduced the tier one and tier two test recommendations in the separate new guidance on laboratory support for targeted and comprehensive toxicology testing for the ED.
The recommendation is that tier one tests be available on a stat basis, with turnaround time goals of 30 to 60 minutes once the laboratory receives the samples. These results have significant impact on the care of a poisoned patient.
Tier two test results are generally used to confirm the specific exposure or for broad-spectrum drug screening, and the results often do not have a significant impact on immediate care. “But I would venture to say that’s likely due to the turnaround times involved in some of the mass spectrometry-based testing,” Dr. Kyle said.
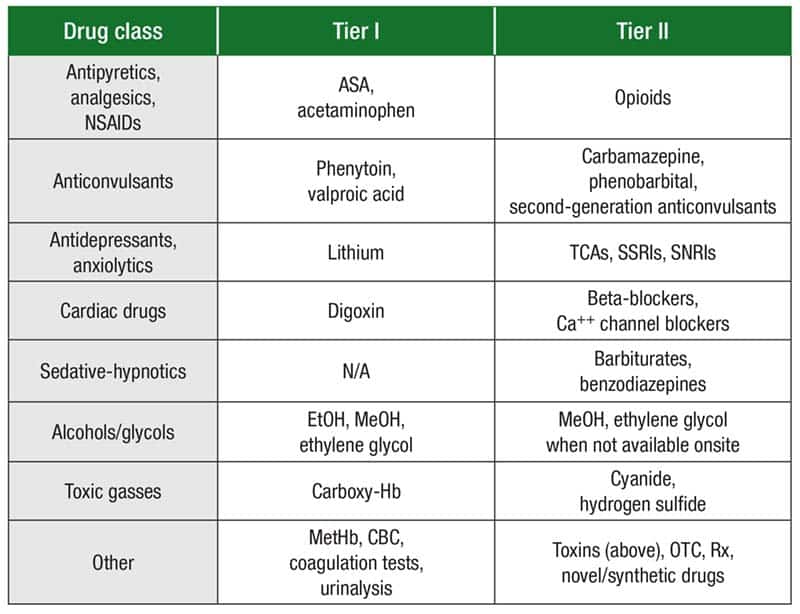
In Fig. 3 are the tier recommendations for eight drug classes. “For antipyretics, analgesics, and nonsteroidal anti-inflammatory drugs, we have to have stat testing for acetylsalicylic acid or aspirin, as well as acetaminophen.” In tier two for the same drug class, patients often present with specific symptoms and are responsive to naloxone, so opioids are tier two.
In the antidepressants and anxiolytics drug class, “we moved tricyclic antidepressants into tier two for this document because they’re not nearly as highly prescribed as they were 25 years ago,” Dr. Kyle explained.
In the sedative-hypnotics class, barbiturates and benzodiazepines are in tier two owing to their low mortality (unless mixed with other substances, ethanol in particular).
Ethanol, methanol, and ethylene glycol are in tier one of the alcohols/glycols drug class. “When those are not available onsite,” Dr. Kyle said, “the laboratory should have protocols for sending samples out, know exactly which reference lab or hospital across town it’s going to, and get that done very quickly.”
In the guidance document Dr. Kyle previewed are recommendations for all laboratories, one of which is that point-of-care testing for toxic substances other than drugs of misuse should not be used. “Point-of-care testing for anything other than your recreational drugs should not be performed,” he said.
Another is that laboratories and clinical experts, including emergency medicine and medical toxicology providers, should discuss the needs for tier one and tier two testing based on the patient populations and lab capabilities, and this is a theme that runs through the document, Dr. Kyle said.
Third is the recommendation that the laboratory provide direct access to the laboratory director or other qualified personnel for consultation on test selection, performance criteria, and result interpretation. “We serve our pain management clinicians in a huge way in this regard,” Dr. Kyle said, “because we have so many immunoassays that are reactive or nonreactive to different drugs.
“If you have ED clinical toxicologists in your emergency department,” he adds, “get to know them well.”
Dr. Kyle presented a sampling of the recommendations for high-complexity testing:
- Potentially interfering substances must be evaluated for each analytical method.
- Rigorous reevaluation of analytical interferences should be performed after any significant change to specimen processing, analytical methodology, assay reagents, hardware, or software.
- For compound identification, laboratories should not rely on mass spectrometry match criteria alone; they should also include retention time, isotope pattern, fragmentation pattern, and other criteria, whenever possible.
- Laboratories using high-resolution mass spectrometry should strive to use 5 ppm as a threshold and should validate their identification criteria using known positive and negative samples.
- Laboratories should seek to use a second and different analytical method when uncertain about analyte identity and should seek to use a different specimen aliquot to reduce the risk of contamination.
- The laboratory and ED should provide continuing education on current test performance, novel compounds/drugs, and recommendations on new assays or target analytes. “There should be a continuous line of communication between ED toxicologists and the laboratory so you can provide this information to your other clinical providers,” Dr. Kyle said.
Illustrating the importance of collaboration, communication, and education was Trevonne M. Thompson, MD, professor of emergency medicine and medical toxicology and director of the Division of Medical Toxicology at the University of Illinois Chicago. He spoke in the ADLM session and shared this case, among others.
A 50-year-old person who attempted self-harm at home was brought to a critical access hospital after an overdose. The patient arrived at the ED profoundly sedated, Dr. Thompson said, and the family believed the patient ingested an entire bottle of prescribed lorazepam.
A urine drug screen was ordered, for which the results were returned in about four hours and were negative for benzodiazepines. The physician pursued a massive workup on why the patient was profoundly sedated: additional laboratory tests, advanced imaging, a lumbar puncture, and an electroencephalogram. The patient was ultimately intubated.
The critical access hospital staff called the Illinois Poison Center, after which the poison center staff called the hospital laboratory. “It turns out that in this critical access hospital, they don’t have a more modern benzodiazepine test in the lab and they don’t detect for lorazepam,” Dr. Thompson said. The physician is thinking the benzodiazepine test is negative, but “all benzodiazepines are not made the same. We know this,” he said. “Everybody does not know this.”
Patients who overdose on a benzodiazepine alone may be profoundly sedated but usually will do well without intubation, he said.
“We need to collaborate and communicate with one another,” Dr. Thompson said. “What are we testing for? What are we not testing for? What’s a false-positive? What’s a false-negative?” The recommendation in the guidance says laboratories should educate ED and other clinicians about the limitations of urine drug testing, particularly false-positives, false-negatives, and the possibility that the presence of a drug may indicate past exposure but not necessarily have an impact on current patient behavior.
“We have to have an understanding of what we are testing for and not testing for,” Dr. Thompson said of immunoassay performance. “Knowing that in this hospital their benzodiazepine screen is not testing for lorazepam is important.” And here the recommendation says immunoassay performance characteristics depend on whether the assay is designed to detect a drug or drug class, and the laboratory should educate ED staff on the performance of each assay and include interpretive comments to reflect performance.
Another recommendation says laboratories should attempt to optimize the detection of relevant compounds such as glucuronidated benzodiazepines and synthetic opioids. “If we’re not testing for it but maybe we should, that’s a part of the dialogue as well,” he said.
On the question of turnaround time, the recommendation is “generally less than 60 minutes.”
“Four hours for a urine drug screen is a really long time in emergency department time,” Dr. Thompson said. “Even in a critical access situation, having an understanding of what we should expect with turnaround times for acute testing becomes very important because, I can tell you as an emergency physician, if I’m not getting answers, I’m going down pathways that might end up including a whole lot of extra work.”
Amy Carpenter is CAP TODAY senior editor.

