Chatbot market revving up: COVID-19 a driving force
June 2020—The global market for health care chatbots has been growing at a fairly rapid pace in recent years, but “COVID-19 is the thing that’s going to make chatbots mainstream,” says Greg Kefer, chief marketing officer at the chatbot company LifeLink Health.
A 2019 Allied Market Research report, released just months prior to the COVID-19 pandemic, projected the health care chatbot industry would reach $345.3 million by 2026, a steep rise from 2018, when it garnered $116.9 million (www.alliedmarketresearch.com/healthcare-chatbots-market). But now, the COVID-19 pandemic has put into sharp relief one of the key value propositions of chatbots—unlimited scale, which means the timeline for adoption “just got massively compressed,” says Kefer, whose software-as-a-service company develops enterprise-level chatbots for large health care organizations.
[dropcap]A[/dropcap]mong the institutions that have adopted LifeLink’s health care chatbots on a large scale is Arizona-based Banner Health, which implemented the company’s software across the 28 emergency departments in its system to improve the check-in procedure and convey information related to the status of lab results and other types of testing. Organizations like Banner, says Kefer, are pioneers because they have “a more aggressive risk-taking strategy when it comes to innovation and are willing to work with companies to try things. They’re willing to fail, and when they succeed, they start talking about it in public forums. Then, gradually, the rest of the industry begins to follow their lead.”
It’s this can-do attitude that led Banner to install in its emergency departments a bot from LifeLink that acts as a virtual concierge, answering questions that range from “Where’s the bathroom?” to “When can I expect my test results?” To get the conversation started, says Kefer, visitors to the ED are prompted with an SMS message upon check-in that includes a link inviting them to chat with the bot. The conversation looks and behaves like a regular cell phone text message, “but it’s in Web browsers, such as Safari, and that’s because it’s a HIPAA-compliant platform, whereas SMS isn’t,” he adds.
It’s because the bot is integrated into Banner’s Cerner EHR that it can present patients with information about lab testing, imaging, and other procedures that are ordered during their stay in the emergency department. “The chatbot will remind patients what labs have been ordered and present a series of ETAs that project when they will be done,” Kefer explains. In other words, it gives patients an estimate of when their emergency department visit will be complete.
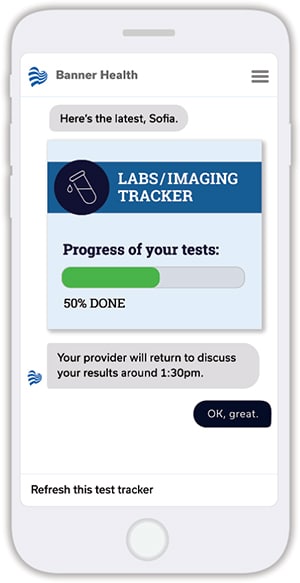 LifeLink is able to do this using algorithms that analyze “a number of different data sources from Cerner—which labs, how many, historical processing time, and the condition of the specific patient,” Kefer says. The information typically is delivered to patients as a graphic that shows the percentage of labs completed. “The technology is configurable,” he continues, “so providers have the option of using exact time estimates, but most prefer the percentage so there’s some wiggle room if something unexpected happens.”
LifeLink is able to do this using algorithms that analyze “a number of different data sources from Cerner—which labs, how many, historical processing time, and the condition of the specific patient,” Kefer says. The information typically is delivered to patients as a graphic that shows the percentage of labs completed. “The technology is configurable,” he continues, “so providers have the option of using exact time estimates, but most prefer the percentage so there’s some wiggle room if something unexpected happens.”
LifeLink’s chatbot technology is also being used to conduct clinical trials at scale, Kefer says. The company provided a digital platform for the BRCA Founder Outreach Study, which connected high-risk individuals with genetic testing for the BRCA mutation. The bot screened potential participants and sent participants’ orders for genetic testing directly to Quest Diagnostics through a backend lab information system integration, he explains. “The whole aspect of [managing] the clinical trial participant was done by the bot.”
With regard to more widespread use, LifeLink is talking to some “very large” lab companies about how to increase the percentage of patients who adhere to their physician’s request to get a blood draw for their physical. “There’s a chatbot dimension there to make sure people do all the right things,” says Kefer. “So if you ask patients, for example, to fast before you draw their blood, the bot can remind them that at 8 PM [the night before], they need to stop eating. That kind of thing is perfect for this kind of technology.”

But despite some outward similarities, Kefer is quick to note the differences between LifeLink’s chatbot technology and a digital assistant like Siri or Alexa: “Our bot is not a natural language processing bot,” he says. In other words, patients can’t ask the bot open-ended queries. Rather, the bot’s conversations with users flow based on dynamically generated decision trees that have been designed for different workflows. Kefer cites an appointment request bot, which LifeLink developed for Philadelphia-based Jefferson Health, as an example. “The next company that wants an appointment bot can use the same pattern,” he explains. “The general flow of that conversation has been designed and optimized to deliver the desired outcome—a scheduled appointment. But it’s also scalable.” This allows health care institutions to potentially go live with the company’s bots “in a matter of weeks,” he adds.
[dropcap]W[/dropcap]hile chatbots are being deployed throughout various areas of health care systems, the symptoms check segment of the health care chatbot industry holds the largest share of the global market, as compared to medical and drug information assistance, appointment scheduling and monitoring, and other applications, according to the Allied Market Research study. A company catering to this market segment is Buoy Health, a SaaS vendor that markets the Buoy Assistant comprehensive symptom checker and patient-facing care triage tool.
The company developed Buoy Assistant, says Eliza Wilson, director of product, by creating what she calls a “map of medicine,” or a catalog of diagnoses and associated symptoms. The map, a Bayesian network, drives the chat experience. Each time an end user provides information about their symptoms, the network uses that information to re-evaluate the question it will ask next, with the intent of ruling out the largest number of potential explanations for the symptoms the user is experiencing, Wilson explains. The bot pulls from a pool of approximately 30,000 questions.
The company, too, is developing technology that allows users to ask the bot open-ended queries. “We’re starting to create more flexible conversation paths, including starting with an open-ended ‘tell us what brings you here today,’ and letting people describe, in their own words, exactly what they’re worried about,” Wilson says.

Like LifeLink, Buoy Health is a vendor of choice at Banner Health. While LifeLink assists patients in the emergency department, “Buoy helps Banner patients decide if they need to seek care in the first place—and what type of care,” Wilson says. People can access Buoy Assistant from Banner’s website to check their symptoms before visiting the hospital.
By February, the company had added COVID-related symptom diagnosis functionality to Buoy Assistant based on CDC guidelines, making the vendor one of the first among a burgeoning number of health care chatbot companies to respond to the COVID-19 pandemic. “When coronavirus started appearing in the U.S., we were able to identify spikes in coronavirus-like symptoms in 17 states before they had been detected by public health agencies,” Wilson says.
In addition to inquiring about symptoms, the chatbot takes into account risk factors such as where people work and their living arrangements to determine their level of exposure to the SARS-CoV-2 virus. Using this information, the Buoy platform directs people to testing locations based on zip code and symptom severity.
In March, Buoy Health contracted with the state of Massachusetts to provide this functionality to the commonwealth’s nearly 7 million residents via the state government website. Under the company’s arrangement with the commonwealth, the platform dynamically updates hospital and test center capacity, says Wilson, “so we can guide patients to areas where there are resources to support them right now.” At CAP TODAY press time, the state of Virginia was planning to go live with the technology as well, using it as a symptom checker and care triage tool. “The major difference is we’re not providing hospital and test center capacity numbers for Virginia, like we are in Massachusetts,” she says.
[dropcap]L[/dropcap]ifeLink, too, is tackling the COVID-19 crisis. When the pandemic struck, the company was working with Memorial Health System, headquartered in Springfield, Ill., to implement a bot that would handle aspects of referral management in lieu of hospital personnel. But when the magnitude of the crisis became clear, the health care system pivoted, implementing LifeLink’s COVID-19 solution instead and putting its original plans for a referral-management bot on the back burner. The COVID-19 bot takes users through a CDC guidelines-based risk assessment and triages them to one of three levels of care. Working at breakneck speed, LifeLink and Memorial Health completed the implementation in just over three days in mid-March.
“What was really important for us was that our end state [what happens after a patient completes the risk assessment] be embedded within the structures and processes,” says Jay Roszhart, president of Memorial Health System Ambulatory Group.

Roszhart noted that the bot has eased the burden on in-house staff by about 20 to 25 percent, in part by addressing “the vast majority of the worried well who are just looking for information.” By mid-May, the tool had handled about 2,200 of the approximately 9,000 encounters directed to the health system’s dedicated COVID-19 respiratory clinics. Staff are following up on the outcomes of symptomatic patients with risk factors, Roszhart says, but the digital assistant could address follow-up care very easily with the appropriate EHR integration.
Bots, in general, “are a dime a dozen,” Roszhart continues. “But finding a vendor that’s willing to partner with you to integrate the bot as part of your workflow is really important.” Building a relationship with LifeLink is helping Memorial Health keep its bot updated as new information about COVID-19 becomes available. “Any time we identify an update or the need for a change in the language or behavior of the bot, since we understand how the bot was built and the decision trees that it was built off of, we’re able to share with LifeLink how we want it to be updated,” Roszhart explains. And because LifeLink hosts the bot, updates don’t need to be pushed to the hospital.
Health care chatbots “are shining in a moment of crisis,” concludes Kefer. “I think the cat’s out of the bag now, and the question is not if, it’s when and how they will be used.” —Charna Albert
FDA gives Leica nod to expand remote diagnosis during pandemic
The FDA has expanded Leica Biosystems’ enforcement discretion relative to using the Aperio WebViewer Web-based viewing software in Aperio eSlide Manager with images acquired on the Aperio AT2 or Aperio AT2 DX digital pathology scanners for remote diagnosis during the COVID-19 pandemic.
This allows pathologists to view and diagnose pathology cases from remote locations, including home offices, using a consumer PC and monitor.
“This application will provide safe and flexible options for pathologists as the Aperio WebViewer operates seamlessly with any of the Aperio AT2 digital pathology systems already installed in hundreds of pathology laboratories across the U.S.,” said Leica Biosystems president Melissa Aquino, in a company press release.
Leica Biosystems, 800-248-0123
Data Innovations debuts latest version of Instrument Manager
Data Innovations has announced the release of its Instrument Manager 2020, version 8.17.00, laboratory management software.
With this latest version, Data Innovations has added the following to Instrument Manager:
-
- time zone awareness capability, which displays time stamps on orders and results that reflect the time zone in which the user’s Instrument Manager workstation is located if it differs from the time zone in which the software’s database server is located.
- Specimen-Management Workspace upgrades, which allow users to launch third-party applications, such as Web links or PDF downloads, from within SM Workspace.
- Caché database upgrade, which allows labs to deploy Instrument Manager on Microsoft Windows Server 2019 and benefit from the associated security updates from Microsoft and InterSystems.
- auto-provisioning using the Lightweight Directory Access Protocol, which integrates user access into Instrument Manager from the LDAP profiles of laboratory networks or health care systems.
Data Innovations, 802-658-2850
Cerner offering select entities de-identified data on COVID-19 patients
Cerner is offering select U.S. health care systems and academic research centers complimentary access to de-identified COVID-19 patient data to support medical treatments, epidemiological studies, and clinical trials related to the disease.
The data include COVID-19-related laboratory results, clinical complications and outcomes that could help drive medical decisions, demographics to help track spread and surge, and information about underlying illnesses, chronic conditions, and treatments. The data are stored on Cerner’s cloud-based HealtheDataLab platform, powered by Amazon Web Services. HealtheDataLab was designed to provide researchers with de-identified patient data that they could manipulate to build complex models and algorithms to inform decision-making.
An application review committee is determining which entities or researchers will benefit from the complimentary offering.
Cerner, 866-221-8877
Dr. Aller practices clinical informatics in Southern California. He can be reached at raller@usc.edu.

