Editor: Frederick L. Kiechle, MD, PhD
Submit your pathology-related question for reply by appropriate medical consultants. CAP TODAY will make every effort to answer all relevant questions. However, those questions that are not of general interest may not receive a reply. For your question to be considered, you must include your name and address; this information will be omitted if your question is published in CAP TODAY.
[button color=”blue” size=”small” link=”https://captodayonline.com/q-a-submission/” target=”blank” ]Submit a Question[/button]
Q. Are there high-specificity immunohistochemistry stains for diagnosing mesothelioma that differ from those recommended in the “Guidelines for pathologic diagnosis of malignant mesothelioma: 2012 update of the consensus statement from the International Mesothelioma Interest Group”?
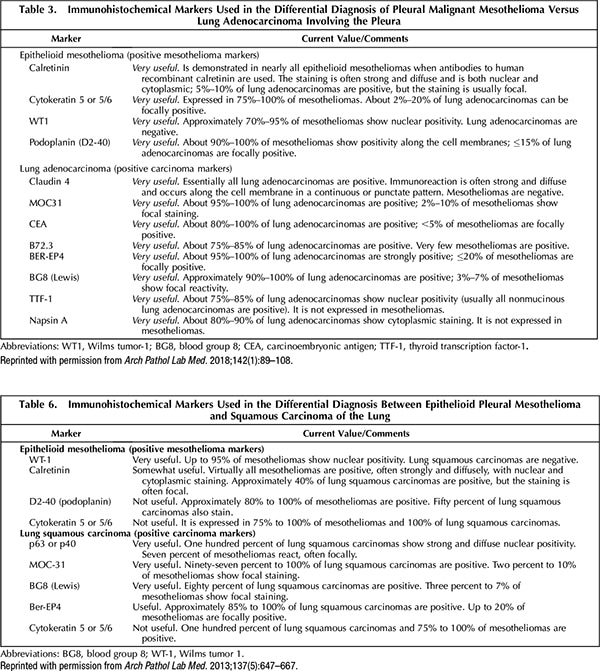
A. June 2020—An updated guideline from the International Mesothelioma Interest Group, published in 2017, did not recommend any new or other markers for mesothelioma. This is seen by comparing table 3 (2017) and table 6 (2012) of the guidelines, as they appeared in the Archives of Pathology & Laboratory Medicine.1,2 However, it is worth noting the addition of claudin 4 as a positive lung carcinoma marker in the 2017 document. Claudin 4 is positive in many carcinoma types but virtually always negative in mesothelioma.
While not included in the 2017 update, GATA3 is another potential marker for mesothelioma, and it is expressed in a variety of other tumors as well.3 In addition, BRCA1-associated protein 1 (BAP1) and methylthioadenosine phosphorylase (MTAP) may be useful in distinguishing reactive mesothelial proliferation from malignant mesothelioma.4
- Husain AN, Colby TV, Ordóñez NG, et al. Guidelines for pathologic diagnosis of malignant mesothelioma: 2017 update of the consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med. 2018;142(1):89–108.
- Husain AN, Colby T, Ordóñez NG, et al. Guidelines for pathologic diagnosis of malignant mesothelioma: 2012 update of the consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med. 2013;137(5):647–667.
- Berg KB, Churg A. GATA3 immunohistochemistry for distinguishing sarcomatoid and desmoplastic mesothelioma from sarcomatoid carcinoma of the lung. Am J Surg Pathol. 2017;41(9):1221–1225.
- Kinoshita Y, Hida T, Hamasaki M, et al. A combination of MTAP and BAP1 immunohistochemistry in pleural effusion cytology for the diagnosis of mesothelioma. Cancer Cytopathol. 2018;126(1):54–63.
Dylan V. Miller, MD
Director, Immunostains and Electron
Microscopy Laboratory
Intermountain Laboratory Services
Murray, Utah
Vice Chair, CAP Immunohistochemistry Committee
Anja C. Roden, MD
Medical Director, Immunostains Laboratory
Mayo Clinic
Rochester, Minn.
Member, CAP Immunohistochemistry Committee
Q. For our hospital tissue and blood committee, we would like to expand the tissue component to look at more preoperative versus postoperative diagnoses (acute appendicitis, for example) and anything else that should be evaluated, such as adequacy of tissue and blood samples. Is there a resource that provides a list of tissue-related quality metrics that we can evaluate?
A. Although the hospital surgical committee usually approves the types of tissues that are submitted to pathology, there are certain surgical specimens that may be exempt from routine or mandatory submission to the Department of Pathology. Local guidance may supersede for foreign bodies, such as bullets or other medicolegal evidence, which are usually given directly to law enforcement authorities.
The best resource for quality metrics is the CAP’s Quality Management in Anatomic Pathology.1 Among the best resources for practical guidance is the Clinical and Laboratory Standards Institute’s “QMS01: A Quality Management System Model for Laboratory Services” (formerly “GP26-A3: Application of a Quality Management System for Laboratory Services; Approved Guideline, 3rd ed.”).2 It is our experience that tissue-related quality metrics can also be gleaned from a health care institution’s occurrence management program.
We typically look at the total number of errors or occurrences reported over a specific time period (quarterly, for example) to determine the most commonly occurring error and to identify errors that resulted in a HARM (hospital stay, readmission, and mortality rates) score greater than our institution’s internally set threshold of acceptability. For example, we would investigate and perform root-cause analysis for any error that was scored at a high level, which would indicate serious harm to a patient. Specimen misidentification would fall under this category, especially if that misidentification led to an incorrect diagnosis or treatment.
Another example would be if we noticed a significant number of errors, such as tissue floaters on stained slides. This type of problem would trigger an investigation, data collection, and evaluation of all the work processes leading to the stained slide and may ultimately result in potential changes to a written procedure, which would include documented staff training on the new procedure.
The bottom line is that taking advice from a published expert is good practice, but it can be equally beneficial to monitor in-house errors or occurrences to find out where the problems lie and how to make impactful decisions about systematically evaluating and improving quality.
-
- Zhai Q, Siegal GP. Quality Management in Anatomic Pathology. Northfield, Ill.: CAP Press; 2017.
- Clinical and Laboratory Standards Institute. QMS01: A Quality Management System Model for Laboratory Services; 5th ed.; 2019.
Elizabeth Sheppard, MBA, HT(ASCP)
Head, Global Market Access
Roche Tissue Diagnostics
Tucson, Ariz.
Former Advisor
CAP/NSH Histotechnology Committee
Sue E. Lewis, BS, HTL(ASCP), QIHC
Member, CAP/NSH
Histotechnology Committee

