Moreover, the consumer and market demand for noninvasive diagnostic testing that previously required invasive procedures will continue to catalyze the commercialization of technology that supports comprehensive diagnostic workups in convenient ambulatory settings. This movement to using technology to “diagnose patients where they are” will continue to highlight the lab’s strategic role at the forefront of early detection and care optimization. Under the value-based care paradigm, the metrics of such optimization are both better clinical outcomes (including under the quality measures of pay-for-performance systems) and more cost-effective delivery of health care.
Longitudinal data: Clinical laboratories daily produce thousands to millions of unique data points that can demonstrate changes in a disease or health condition over time. This data set is of value only when those data are connected one to another, and to associated clinical metadata, so as to facilitate proactive prediction and early detection of clinical conditions. Trends in the structured data of the clinical laboratory can provide information on disease progression and comorbidities that alter the risk profile of that disease condition. The architecture of that data can establish whether a patient is being monitored in accordance with national treatment guidelines, the converse being gaps in care that will need to be closed. The assembled data informs health care entities on the health status of the regional community they serve, so as to guide strategic design and implementation of overall health care programming for that region.
Health equity: The clinical laboratory sits at the crossroads of patient access to health care and patient health outcomes. Laboratories have an untapped opportunity to influence key features of health equity: access, wellness, continuity in care, gaps in care, structural inequity, disease burden, and population outcomes.11 The laboratory cannot remain on the sidelines; it must contribute to strategic programming in health equity. This includes having knowledge about inequitable practices in medicine, data intelligence to identify and prioritize opportunities for intervention, expertise in and a commitment to broad partnerships in program design and execution, and clearly defined outcome objectives with measures for assessing the value of the laboratory contribution to such programs.
Pathologists as caregivers: While not traditionally thought of as caregivers (aka, “clinicians”), pathologists are increasingly embracing roles as caregivers, enhancing patient-centered care through innovative approaches. The establishment of Pathology Explanation Clinics (PECs) allows pathologists to engage directly with patients, providing personalized explanations of laboratory results and addressing individual concerns.12 Patient-pathologist consultations offer patients the opportunity to view their biopsy slides and discuss their diagnoses directly with pathologists, fostering a deeper understanding of their conditions and empowering them in their health care journey.13 Additionally, the development of patient-centered pathology reports, which present complex medical information in accessible language,14 has been demonstrated to enhance patient comprehension and reduce anxiety.15,16 With ever-increasing patient consumerism, these initiatives underscore the evolving role of pathologists as integral members of the frontline patient care team, committed to improving patient outcomes through direct communication and education.17
Can the clinical laboratory trigger coordinated care pathways for the patient’s health care journey?
In 2017, the Project Santa Fe Foundation established the Clinical Lab 2.0 movement18 for the express purpose of creating a disruptive value paradigm and exploring alternative business models that expand the role of diagnostic services in the future health care ecosystem. The starting point for doing so is to convert the transactional activities of resulting laboratory testing (what can be called Clinical Lab 1.0) to the integrated and actionable activities of Clinical Lab 2.0 (Fig. 3). Doing so requires the laboratory to be part of both the health system data analytic teams and the program design teams that are building the requisite clinical programming for value-based care. These programs must achieve target population outcomes and the necessary quality metrics for the following: 1) wellness and preventive care, 2) proactive management of patients with chronic conditions, and 3) closure of the gaps in care, which can place patients at increased risk for catastrophic health events.
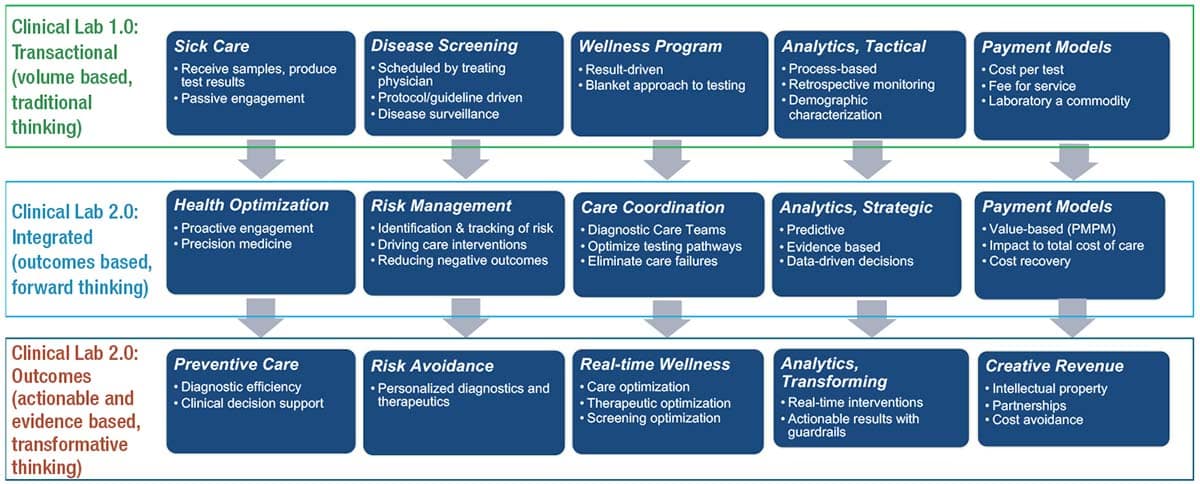
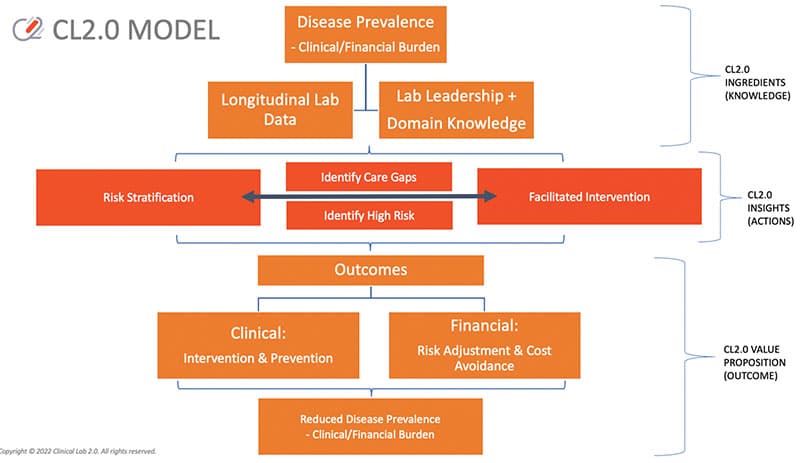
What makes the local laboratory so well placed for coordinated care pathway initiation is that structured and statistically combinable laboratory data informs the activities of value-based care. Published examples from Clinical Lab 2.0 demonstration projects include proactive identification of acute kidney injury during hospitalization,19 proactive identification and management of prenatal care20 and persons with diabetes,21,22 and proactive identification of evolving chronic kidney disease in regional populations.7 While these peer-review examples may seem limited, the unifying themes are generalizable: 1) there is an unmet need on the part of a key stakeholder (e.g. hospital leadership, the health system risk-based population health group, the state Medicaid program); 2) the clinical laboratory provided either or both longitudinal and population-based laboratory data to inform clinical care programming; and 3) the laboratory worked directly with the stakeholders to ensure that these laboratory-based data streams translated into effective clinical programming. The sequence of steps is mapped out in Fig. 4, conceptualized as Clinical Lab 2.0 Laboratory-Initiated Care Pathways.23
Will pathologists provide leadership for programmatic delivery of value-based care?
Pathologists and clinical PhDs already carry enormous and largely unappreciated responsibility as directors of clinical laboratories, over and above their immediate obligations to patient care. While director-level responsibilities can be delegated, the legal and regulatory responsibility for laboratory performance is nontransferable and must be fulfilled at the highest level of commitment. The question can be asked: Can and should pathologists and clinical PhDs add programmatic leadership outside the laboratory to their scope of practice? For the contributors to this article, the answer is more than yes—we consider leadership outside the laboratory to be essential to the practice of medicine as laboratorians. We must be “present” and serve as members of the strategic planning teams for design of health care delivery. Absent such involvement, we are confined anonymously to our clinical laboratories, without ability to bring our expertise and domain knowledge to bear on actual health care decisions and delivery models. Collectively, it is the multidisciplinary and interprofessional health care team that delivers value-based care.
Which leads us to the stakeholders of value-based care. The first stakeholder is the individual patient. Whether the patient is well or experiencing an acute illness and/or coexisting with a chronic disease condition, design of health care delivery to promote the optimal patient experience is very much a laboratory concern. Phlebotomy is an immediate example: The skill and demeanor of the professional performing the venipuncture is a key part of the patient experience. The second stakeholder is the health care provider for that patient. The performance of the laboratory is either a source of satisfaction or substantial dissatisfaction to providers. Regardless of the business model for health care delivery, these two stakeholders are the reason clinical laboratories exist.
But value-based care requires laboratory leadership to work with a broader array of stakeholders:
- Health care organizations, independent provider practices, and payers who are at financial risk under value-based payment agreements, which involve quality metrics, patient experience, patient outcomes, and cost outcomes.
- Clients of the laboratory that: a) have influence and authority to implement policies and clinical protocols and to enact workflows that support evidence-based pathways for care and support the Quadruple Aim in health care; b) use tools and data to develop clinical insights to identify risk, stratify patient populations, and change provider and patient behaviors in order to focus on improved clinical outcomes; and/or c) focus on cost avoidance in the total cost of care as a key component of meeting value-based payment metrics.
- Employers and government entities that carry the overall financial risk for health care coverage of a population.
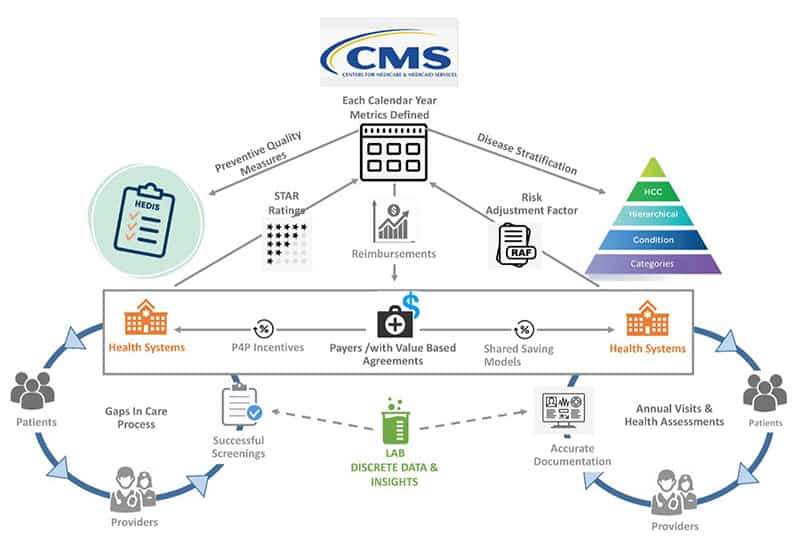
To work with these stakeholders, laboratory leadership needs to understand the mechanics of the value-based payment system. Fig. 5 illustrates the critical role of the clinical laboratory in informing both arms of this paradigm. For a substantial portion of the chronic conditions falling under the value-based payment system, longitudinal data are key. The data must be analyzed both for where there are gaps in care that need to be closed (the transactional pay-for-performance left side of Fig. 5), and where a patient is at risk of going (the predictive risk-stratification/shared savings right side of Fig. 5). These data, and the laboratory professional expertise that can be brought to bear on leveraging these data, are what clinical laboratory leadership brings to the table for stakeholders operating under a value-based payment system. Although the clinical laboratory currently receives no financial incentives through either the pay-for-performance or shared savings set of programs, the clinical information generated by the clinical laboratory is a crucial component of either system being able to operate. Laboratory leadership will need to articulate the statement-of-work they will carry responsibility for, in order to justify access to the funds flow of value-based care. Eligible deliverables can include the steady stream of actionable laboratory data intelligence, proactive programming to enhance access of a regional population to laboratory testing and thence coordinated care, and specific interventions to improve the ability of an integrated delivery network to meet the metrics of value-based care.
Role of pathology informatics in guiding laboratory engagement in value-based care
The aspirational role of the clinical laboratory at the center of health care delivery has long been advocated through the literature of our own specialty,24 with strong arguments made for deploying clinical laboratory analytics25 and computational pathology26 within the context of the subspecialty of pathology informatics.27-29 With the continued maturation of clinical decision support30 and the entry of artificial intelligence into the “big data” realm of clinical laboratory practice,31-34 the potential for these four subdomains of pathology informatics to inform the practice of laboratory medicine and digital anatomic pathology looms large. AI-driven tools can guide digital pathology and clinical laboratory reporting, but remain confined to our own duty cycles. It is essential that pathologists are engaged in the broader AI-driven strategies to streamline the clinical diagnostic pipeline and assessment and management of patient outcomes, boosting the efficacy of health care delivery and hopefully its cost efficiency.
The opportunities to apply the power of laboratory-based data analytics to the human condition are immense. Examples range from the importance of data-driven leadership in diagnostic stewardship35,36 and the opportunity to reduce care variation in pursuit of clinical quality37 to the highly integrative Kidney Precision Medicine Project38 and digital maturity models that improve quality and safety.39 The patient experience also is a critical component of value-based care, and patient access to laboratory test results is an opportune domain for pathologists to guide AI-assisted patient portals.40 However, examples of the clinical laboratory extending its data analytical reach into the realm of value-based health care remain limited (for example, see reference 41), with calls for greater effort in doing so from around the globe, and not just within the U.S.42-45 It is hoped that the strategic principles laid out herein will empower leaders in pathology informatics worldwide to embrace this challenge.
Threats and risk in taking on a role in value-based care
Siloed laboratory data and leadership: Many laboratories lack the requisite data infrastructure and the time or expertise to step beyond their traditional role and responsibilities as providers of diagnostic results. While laboratory results are already transferred from the laboratory information system to the electronic health record, we are advocating that laboratory leadership should travel with the data. However, providing leadership is not done in isolation; the practice of medicine is a team effort. The professional expertise embodied in laboratory leadership must be part of the broad teams of health system strategic planners. Then all teams may fulfill their assigned roles in program deployment and execution. Our experience is that, without such proactive engagement, the laboratory will remain consigned to being a commodity and will not be viewed as an asset. This admonition applies regardless of whether the clinical laboratory is an in-system or independent laboratory.
Encroachment: Scope of practice is jealously guarded by each medical specialty. Several contributors to this article have experienced blunt declarations from medical colleagues that pathologists do not have the clinical expertise to understand the meaning of laboratory results emanating from their laboratories. We have found that proactively engaging with medical colleagues as part of strategic planning, and proactively establishing the respective responsibilities of each medical practitioner in the clinical workflow, can help ensure that such accusations are not forthcoming.
Patient-physician relationship: However complex a multidisciplinary and interprofessional health care team might be, the treating physician (however defined) is the medical professional carrying ultimate responsibility for the successful provision of health care to any given patient. The clinical laboratory supports that singular relationship. Our advocacy is that, in bringing their expertise forward, pathologists can proactively map out their respective responsibilities for data integration, interpretation, and leveraging, as part of ensuring that the chain-of-custody for laboratory-generated diagnostic information can culminate in optimized patient care.
Consumerism: While there is opportunity, there also is threat in regard to patient consumerism. An underperforming clinical laboratory can be a singular source of dissatisfaction, to patients and their providers. The willingness of payers, health systems, and clinical strategists to work with laboratory leadership in the value-based space will depend heavily on the overarching performance of the respective clinical laboratory.
Regulatory requirements: All medical professionals are subject to the requirements of patient privacy, data security, and confidentiality. The activities described herein fall within the paradigm of patient quality and safety, and as part of health system programming are permissible. That being said, the regulatory requirements of data curation and transfer must be respected. The specific challenges of adopting artificial intelligence as part of the practice of medicine are subject to the regulations and statutes of specific countries and jurisdictions. Our advocacy is that clinical laboratory professionals, and the professional societies of which they are members, have a particularly important role to play in navigating the regulatory and statutory environment of this artificial intelligence-enhanced future.
‘New’ business of the laboratory in value-based care
While the fee-for-service system of payment continues to dominate the U.S. advocacy landscape for pathology and laboratory medicine, every major health care stakeholder is increasingly subject to and participating in the value-based payment system. As the market opportunity of value-based care becomes visible for the clinical laboratory, embracing the Clinical Lab 2.0 practice model will be essential. Following are the important actions required of clinical laboratories:
- Be knowledgeable and engaged in your hospital, health plan, or health system’s plans for value-based reforms. Allow and encourage pathologists, clinical PhDs, and managers who want to be engaged to attend committees, workgroups, or system-level planning events, with time away from the bench or microscope to enable them to be present at these crucial endeavors.
- Implement innovative information technology and diagnostic integration solutions. Empower laboratories to be key partners in delivering integrated, real-time health care data to patients and providers across the health system service area. Developing standards for data aggregation of longitudinal data and interoperability will identify data infrastructure gaps and help guide implementation of AI-enhanced algorithms for supporting early diagnosis and intervention, identifying gaps in care, and identifying patients at higher risk for disease onset or worsening.
- Educate the laboratory workforce to participate in population health management. Create educational opportunities and develop programming recommendations to equip professional and technical/managerial personnel with the necessary skills to design future care models, use advanced risk stratification techniques, and leverage longitudinal data for early detection. Be part of health system implementation of population-level coordinated care management programs such as for diabetes, cardiovascular and renal disease, and colorectal cancer screening.
- Demonstrate the laboratory’s role in patient outcomes. Proactively identify the successful programmatic outcomes that can be attributed to laboratory involvement. This enables clinical laboratories to showcase their value across the health care ecosystem through reproducible data-driven initiatives. This is a battle won in detail: Every example builds the case for the clinical laboratory as an asset, not just a commodity.
- Define alternative revenue strategies. Participate in redefining the landscape of laboratory services by creating alternative payment and business models within the space of value-based care. Advocate for policies that integrate pay-for-performance and risk stratification payment methods with laboratory data, with funds flow directed toward the clinical laboratory performing such services.
The Project Santa Fe Foundation has formally commissioned working groups to strategize in the latter four domains. Our goal is to then partner with the broader laboratory industry community to socialize these concepts, and then to represent clinical laboratories in the national health care community of value-based care. By embracing these strategies, pathologists and clinical laboratorians can position themselves as essential contributors to the evolution of value-based care.
Conclusion
This position paper argues that reengineering the current laboratory infrastructure from “reactive confirmation” of a physician’s diagnosis to a “predictive and proactive medicine” approach supporting early detection of chronic conditions is essential and aligned with the definition of value-based care. This realignment requires that clinical laboratory leadership educate themselves in the strategies and mechanics of value-based care, and then proactively engage in population health programmatic efforts in their medical communities. Laboratory leadership must also help develop a new business model for these professional activities, including the establishment of alternative payment streams for the clinical laboratory over and above the existing fee-for-service system. Let us continually strive toward improving the human condition by helping to create a sustainable health care system that focuses on proactive prediction, personalized care, and improved population health. The potential value of pathology and the clinical laboratory does not end when we release a result. Rather, that moment is where our true value begins.
The authors’ institutional affiliations follow: Khosrow Shotorbani, MBA, MLS (ASCP), Project Santa Fe Foundation, LLC; Kathleen Swanson, MS, RPh, Project Santa Fe Foundation, LLC; Bruce A. Friedman, MD, Department of Pathology, University of Michigan, Ann Arbor; Eric F. Glassy, MD, Affiliated Pathologists Medical Group, Rancho Dominguez, Calif.; Michael J. Crossey, MD, Syncronys HIE, Albuquerque, NM; Mark K. Fung, MD, PhD, Department of Pathology and Laboratory Medicine, University of Vermont College of Medicine, Burlington; Karen L. Kaul, MD, PhD, Department of Pathology and Laboratory Medicine, NorthShore/Endeavor Health, Evanston, Ill.; Octavia M. Peck Palmer, PhD, Department of Pathology, University of Pittsburgh; Nancy Stratton, MBA, COLA, Inc., Columbia, Md.; Myra L. Wilkerson, MD, Department of Pathology and Laboratory Medicine, Geisinger Medical Center, Danville, Pa.; Richard J. Zarbo, MD, DMD, Department of Pathology, Henry Ford Health System, Detroit, Mich.; Gaurav Sharma, MD, Department of Pathology, Henry Ford Health System, Detroit, Mich.; Ronald Jackups Jr., MD, PhD, Department of Pathology and Immunology, Washington University School of Medicine, St Louis, Mo.; Ulysses G.J. Balis, MD, Department of Pathology, University of Michigan, Ann Arbor; Junya Fukuoka, MD, PhD, Department of Pathology, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki, Japan; John V. Groth, MD, Department of Pathology and Laboratory Medicine, NorthShore/Endeavor Health, Evanston, Ill.; Lena Chaihorsky, Alva10 Inc., Cambridge, Mass.; Merce Jorda, MD, PhD, MBA, Department of Pathology and Laboratory Medicine, University of Miami (Fla.) Miller School of Medicine; Chen Liu, MD, PhD, Department of Pathology, Yale School of Medicine, New Haven, Conn.; Mario Plebani, MD, Department of Medicine—DIMED, University of Padova, Italy, and Department of Pathology, University of Texas Medical Branch at Galveston; R. Keith Laughman, DBA, CareTinuum Advisors, Brentwood, Tenn.; James M. Crawford, MD, PhD, Department of Pathology and Laboratory Medicine, Northwell Health, New Hyde Park, NY.

