Editor: Frederick L. Kiechle, MD, PhD
Submit your pathology-related question for reply by appropriate medical consultants. CAP TODAY will make every effort to answer all relevant questions. However, those questions that are not of general interest may not receive a reply. For your question to be considered, you must include your name and address; this information will be omitted if your question is published in CAP TODAY.
[button color=”blue” size=”small” link=”https://captodayonline.com/q-a-submission/” target=”blank” ]Submit a Question[/button]
Q. Does a histotechnologist need a bachelor’s degree to run in situ hybridization? Is there a minimum educational requirement to run (not interpret) ISH testing in an anatomic pathology laboratory?
A. November 2019—The educational requirements to perform ISH, as well as such high-complexity testing activities as quality control interpretation and grossing, are a minimum of an associate’s or bachelor’s degree in a course of study defined in laboratory general checklist requirement GEN.54750 Nonwaived Testing Personnel Qualifications. More specifically, an associate’s or bachelor’s degree in a chemical or biological science or medical laboratory technology is required, as is specific laboratory training, which is also noted in the checklist requirement. Histotechnology is an acceptable course of study, according to the Centers for Medicare and Medicaid Services, because it is considered a biological science.
Clinical and Laboratory Standards Institute. QMS03: Training and Competence Assessment; Approved Guideline. 3rd ed. 2009.
College of American Pathologists. GEN.54750 Nonwaived testing personnel qualifications. In: Laboratory general checklist. Sept. 17, 2019.
U.S. Department of Health and Human Services, Centers for Medicare and Medicaid Services. Clinical Laboratory Improvement Amendments of 1988; final rule. Fed Regist. 1992:7175 [42CFR493.1423], 7183 [42CFR493.1489].
Cynthia McGurn, MHA, MT(ASCP)
Technical Specialist, Laboratory Accreditation
College of American Pathologists, Northfield, Ill.
Q. Does using an alcohol swab affect the results of an ethanol blood test?
A. There have been case reports in the scientific literature suggesting that use of an alcohol swab can influence blood testing for ethanol (ETOH).1 However, recent studies that examined the issue directly demonstrated that there is not a significant impact from using alcohol swabs prior to blood ethanol testing.
One such study analyzed 56 paired venous blood samples from healthy volunteers for ethanol.2 Prior to blood draw, each volunteer was swabbed with 70 percent isopropyl alcohol on one arm and saline on the other. There was no statistically significant difference between the groups. A more recent study involved swabbing alcoholic antiseptic (0.5 percent chlorhexidine, 70 percent ethanol) on both arms of 23 healthy volunteers and then collecting blood from each arm.3 The alcohol was not allowed to dry prior to phlebotomy on one arm, and it was properly dried using a cotton ball prior to blood draw on the other arm. In both sets of samples, the ethanol concentrations were undetectable by an enzymatic assay and a reference gas chromatography method. Based on these studies, it is unlikely that use of an alcohol swab will affect the results of an ETOH blood test.
- Yigit O, Arslan S. Swabbing skin with alcohol could change the blood alcohol level significantly. J Emerg Med. 2012; 43(4):718–719.
- Tucker A, Trethewy C. Lack of effect on blood alcohol level of swabbing venipuncture sites with 70% isopropyl alcohol. Emerg Med Australas. 2010;22(1):9–12.
- Lippi G, Simundic AM, Musile G, Danese E, Salvagno G, Tagliaro F. The alcohol used for cleansing the venipuncture site does not jeopardize blood and plasma alcohol measurement with head-space gas chromatography and an enzymatic assay. Biochem Med (Zagreb). 2017;27(2):398–403.
William Clarke, PhD, MBA, DABCC
Director, Clinical Toxicology, Johns Hopkins Hospital
Professor of Pathology,
Johns Hopkins University School of Medicine
Baltimore
Member, CAP Toxicology Committee
 In this “Best of Q&A” series, we reprint select coagulation-related questions and answers. All have been chosen for their timeliness and relevance today. The following question and answer were published in November 2015.
In this “Best of Q&A” series, we reprint select coagulation-related questions and answers. All have been chosen for their timeliness and relevance today. The following question and answer were published in November 2015.
Q. Is there a recommended procedure for or reference article about checking APTT reagent sensitivities (for the identification of factors VIII and IX) when changing lot numbers and reference range?
A. The activated partial thromboplastin time (APTT) clot-based assay is a global test used to detect factor deficiencies in patients with a bleeding diathesis or as a preoperative screen to ensure normal coagulation laboratory parameters before an invasive procedure. Of note, in patients without a history of hemostatic or thrombotic disorders, the literature suggests that preoperative screening with the prothrombin time (PT) or APTT is of little clinical utility, as discussed by the Choosing Wisely campaign.1 An additional use for the APTT is therapeutic monitoring for unfractionated heparin, yet this is being replaced by anti-Xa assay in many institutions.
It is desirable to have APTT systems that are sensitive to factor levels in the 30 percent to 40 percent range.2 Having a system in which APTT (or PT) begins to prolong when factor levels are higher than 40 percent is likely to provide little clinical utility and may initiate unnecessary laboratory workups. Conversely, laboratories would like to detect mild factor deficiency (for example, mild hemophilia A) in patients at risk for bleeding during high-risk hemostatic challenges such as neurosurgery. In this scenario, an APTT sensitivity of 20 percent for factor VIII activity would be unacceptably low. Ultimately, there is a balance between being adequately sensitive to factor deficiencies that cause bleeding (factors VIII, IX, and XI) without unnecessarily detecting mild factor deficiencies that do not cause bleeding (factor XII, prekallikrein, high-molecular-weight kininogen).
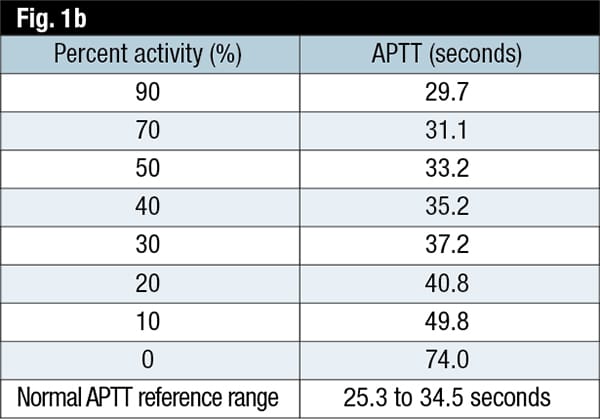
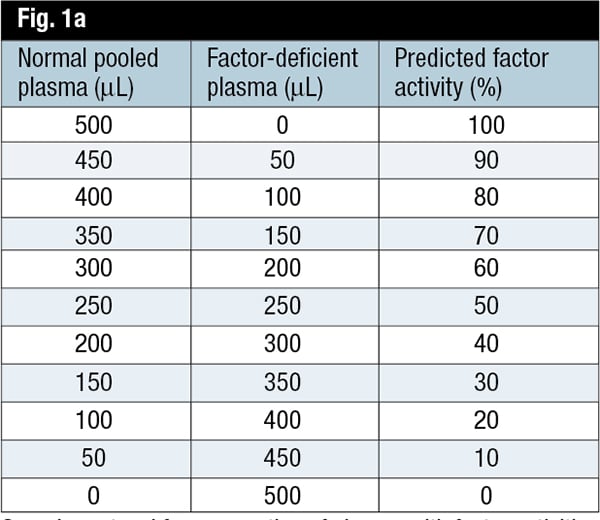
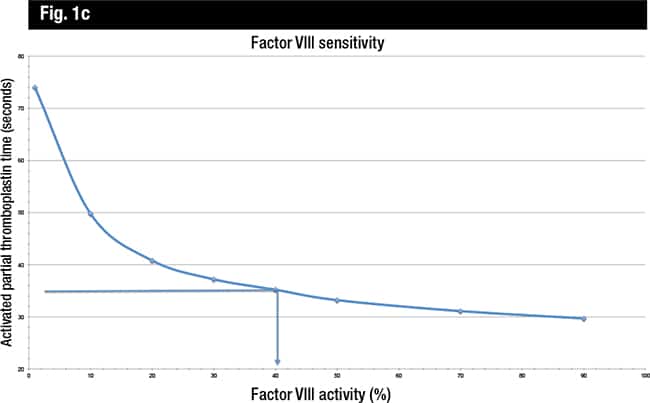
The Clinical and Laboratory Standards Institute provides guidance for determining the APTT sensitivity to clotting factor deficiencies in its H47-A2 publication.3 The CLSI recommends performing APTT (or PT) on samples with a range of single factor activity (e.g. zero to approximately 100 percent). The dilutions are prepared by mixing assayed normal pooled plasma with assayed factor-deficient plasma (Fig. 1a). The factor activity and corresponding APTT results for each dilution are tabulated (Fig. 1b) and then graphically represented with the APTT values (in seconds) on the y-axis and the percent activity for the factor on the x-axis (Fig. 1c), using log-log graph paper. This procedure does not require that the factor activity be measured; rather, the APTT is plotted against the predicted factor activity after mixing with factor-deficient plasma. The upper limit of the laboratory’s APTT reference interval (mean PTT ±2 SD) is drawn on the graph, and this is the estimate of the sensitivity for that assay reagent-instrument system. The CLSI clearly states, “This value should be considered an estimate and should not be considered absolute, because of variables in the materials used.”
It is important to confirm the adequacy of the normal pooled plasma. Plasma should be pooled from at least 20 donors4 to ensure sufficient factor concentrations (near 100 IU/dL or %), and this information is provided by the commercial manufacturer.3 Lawrie, et al., found that the CLSI-recommended procedure is misleading.5 In particular, they found that using different normal pooled plasmas and factor-deficient plasmas resulted in varying factor activities for a certain system. In addition, when performing thrombin-generation tests (TGT), they discovered the potential to generate thrombin was not completely dependent on the level of component clotting factors. From these data, they suggest lyophilized-deficient plasmas may have procoagulant factors exerting an effect on the TGT or the test systems described in the CLSI guidelines. Thus, the authors recommend that factor sensitivity “should either be determined by the reagent manufacturers for specific instrument/reagent systems or by individual laboratories using well-characterized samples from patients with inherited coagulation deficiencies. We would suggest testing a minimum of 20 deficient samples with potencies evenly distributed in the range [of] 10–50% for each of the intrinsic coagulation factors (factors VIII, IX, XI and XII).” This recommended procedure will be difficult for most laboratories due to limited access to such factor-deficient samples.
Whether performing the APTT sensitivity studies per the CLSI recommendations or by using samples from factor-deficient patients, either method can be problematic for smaller laboratories. Many routine coagulation laboratories do not have factor activity assays available in their labs. The CLSI does state that the APTT sensitivity may be obtained from manufacturers or from published studies.3 Many experts in coagulation laboratories find the CLSI procedure helpful to estimate sensitivity, to assess the adequacy of the upper limit of the reference interval,2 and to assist in the interpretation of APTT mixing studies. Notably, the CAP does not require that laboratories assess the sensitivity of their PT or APTT reagents with reagent lot changes, but it is good laboratory practice to understand the assay’s performance or, at least, be aware of information provided by the manufacturer.
- Choosing Wisely, an initiative of the ABIM Foundation. www.choosingwisely.org/societies/american-society-for-clinical-pathology.
- Higgins R, Olson J. [Q&A column.] CAP TODAY. 2015; 29(1):55.
- Clinical and Laboratory Standards Institute. H47-A2: One-Stage Prothrombin Time (PT) Test and Activated Partial Thromboplastin Time (APTT) Test; Approved Guideline. 2nd ed. 2008.
- College of American Pathologists. HEM.37991 Mixing studies procedure. In: Hematology and coagulation checklist. July 28, 2015.
- Lawrie AS, Kitchen S, Efthymiou M, Mackie IJ, Machin SJ. Determination of APTT factor sensitivity—the misguiding guideline. Int J Lab Hematol. 2013;35(6):652–657.
Andrew Jackson Goodwin IV, MD
Currently Associate Professor
Department of Pathology
, University of Vermont
College of Medicine
Burlington
Member of the CAP Coagulation Resource
Committee at time of original publication
Russell A. Higgins, MD
Currently Professor, Clinical Department of
Pathology and Laboratory Medicine
University of Texas Health San Antonio
Medical Director,
University Health
System Pathology Services, San Antonio
Chair of the CAP Coagulation Resource
Committee at time of original publication

