Editor: Frederick L. Kiechle, MD, PhD
Submit your pathology-related question for reply by appropriate medical consultants. CAP TODAY will make every effort to answer all relevant questions. However, those questions that are not of general interest may not receive a reply. For your question to be considered, you must include your name and address; this information will be omitted if your question is published in CAP TODAY.
[button color=”blue” size=”small” link=”https://captodayonline.com/q-a-submission/” target=”blank” ]Submit a Question[/button]
Q. For the population with diabetes, what can the clinical laboratory do to promote evidence-based testing and monitoring for chronic kidney disease?
A. April 2020—The principal laboratory tests for diagnosing and monitoring the severity of chronic kidney disease for adults with type 1 and type 2 diabetes are the estimated glomerular filtration rate (eGFR) by the CKD-EPI equation, calculated from serum creatinine, and the spot or random urine albumin-creatinine ratio (uACR). According to the Kidney Disease: Improving Global Outcomes (KDIGO) 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease, CKD is identified by eGFR < 60 mL/min/1.73 m2 and/or uACR > 30 mg/g (3 mg/mmol), if present for three or more months.
In recent years, CAP Surveys data demonstrate 80 to 90 percent of laboratories in the U.S. reported eGFR with serum creatinine, using a variety of eGFR equations. These include the recommended CKD-EPI equation, as well as the isotope dilution mass spectrometry (IDMS) traceable Modification of Diet in Renal Disease (MDRD) study equation and the original MDRD study equation. Although not assessed in recent CAP Surveys, uACR laboratory reporting is inconstant in that it uses a variety of units. Moreover, some laboratories only report urinary concentrations of albumin alone or with creatinine without calculating the ratio. These nuances in laboratory reporting may seem trivial, but they contribute to the underuse of uACR testing for diabetes, reported at 40 percent and 49 percent in the Centers for Medicare and Medicaid Services Medicare five percent sample and the Optum Clinformatics data sets in 2016, respectively.
According to the Centers for Disease Control and Prevention, 17 percent of American adults are estimated to have CKD. Diabetes is the major risk condition for the disease and is associated with about 40 percent of cases (www.cdc.gov/ckd). Adverse CKD outcomes are CKD anemia, CKD metabolic acidosis, CKD mineral and bone disorder (calcium, phosphorus, intact parathyroid hormone, and metabolic bone disease), cardiovascular events, hospitalization, chronic kidney failure that requires treatment with dialysis or kidney transplant to sustain life, and death.
Kidney failure is a life-threatening illness that, even with dialysis treatment, confers a 50 percent mortality rate after five years—a shorter survival than that of most cancers. On Dec. 31, 2016, there were 726,331 prevalent cases of kidney failure; the crude prevalence was 2,160.7 per million in the U.S. population. Chronic kidney failure significantly reduces quality of life and is linked to a high prevalence of pain and depression, contributing to low employment rates among this group. Total Medicare spending for CKD, including dialysis and kidney transplant, was more than $114 billion in 2016, of which $35.4 billion was spent on chronic kidney failure. Kidney disease is a cost multiplier, with one percent of Medicare beneficiaries with chronic kidney failure accounting for seven percent of Medicare fee-for-service expenditures in 2016 (www.usrds.org).
To address this public health challenge, a growing number of clinical laboratory professional societies and clinical laboratories are collaborating with the National Kidney Foundation to implement the laboratory engagement initiative priorities (www.kidney.org/CKDintercept/laboratoryengagement). The following priorities focus on laboratory implementation:
- Harmonize the descriptive language and reporting of the random or spot uACR as “albumin-creatinine ratio, urine” as an alias to the “microalbumin” test, reported in milligrams per gram (mg/g).
- Standardize use of the CKD-EPI eGFR creatinine equation.
- Implement the “kidney profile” with the clinical laboratory community, which offers eGFR and uACR with a single order. (See “Catching CKD sooner with kidney profile,” CAP TODAY, October 2019.)
The kidney profile is applicable only to targeted testing of risk groups rather than general population screening. In addition to diabetes, other important risk conditions for CKD include hypertension, cardiovascular disease, and family history of CKD. An exhaustive list of risk conditions would include many others, such as cancer and selected chemotherapeutic agents.
The laboratory engagement initiative also aims to assess the impact of the aforementioned three priorities on population health. American adults living with diabetes should be tested for CKD at least annually, according to the clinical practice guidelines of the American Diabetes Association, KDIGO, and Kidney Disease Outcomes Quality Initiative (KDOQI) of the NKF. In addition, the Choosing Wisely recommendations of the American Society for Clinical Pathology recently selected the kidney profile (eGFR and uACR), in addition to serum creatinine, as components of CKD testing for the population with diabetes, hypertension, or both (http://j.mp/ascp-kidneyprofile).
Evidence-based interventions that can prevent the development or slow the progression of CKD in diabetes include glucose and blood pressure control, use of kidney-protective blood pressure medications (angiotensin-converting enzyme inhibitors and angiotensin receptor blockers), and diabetes medications that are sodium-glucose co-transporter-2 inhibitors and glucagon-like peptide-1 receptor agonists. Cardiovascular risk reduction with statin-based therapy is recommended for everyone with CKD over the age of 50. Implementation of the kidney profile as one of four elements of a population health management strategy for diabetes in the Indian Health Service resulted in a 54 percent reduction in chronic kidney failure requiring dialysis initiation between 1996 and 2013. Diagnosis and monitoring of CKD depends entirely on clinical laboratory professionals and thus affords them an opportunity to become engaged in population health and quality improvement.
The laboratorians involved in Project Santa Fe’s Clinical Lab 2.0, who represent major regional U.S. health systems, developed a plan to implement laboratory-driven innovations and analyze their impact on outcomes and expenditures. Laboratory professionals can extend their influence beyond the lab by engaging with patients, interacting with ordering clinicians, and collaborating with administrative and clinical leadership. They can provide value to the administration by promoting enhanced clinical documentation of CKD, CKD severity, and comorbidities that can be leveraged for enhanced reimbursement and population health innovations. Epidemiologic data show that CKD is the most powerful predictor of acute kidney injury, defined by KDIGO as an acute rise in serum creatinine of 0.3 mg/dL over 48 hours. Therefore, AKI detection and intervention programs should also focus on at-risk populations with CKD.
Joseph A. Vassalotti, MD
Chief Medical Officer
National Kidney Foundation
Clinical Professor of Medicine
Icahn School of Medicine
Mount Sinai Medical Center, New York, NY
Q. What are the proficiency testing enrollment requirements if an analyte is tested in multiple locations within the laboratory?
A. One proficiency testing subscription per analyte is required for each CAP/CLIA number for accredited laboratories. If a laboratory routinely uses more than one primary method/instrument for reporting the same analyte, PT can be rotated among the primary instruments/methods, or PT samples can be used after the due date for self-evaluation. For CAP customers, Quality Cross Check programs are a convenient solution, if available, to monitor instrument performance across multiple instruments/departments.
The laboratory would not be out of compliance if it were to order products from another PT provider for the secondary method. However, it should designate only one PT provider to report results of regulated analytes to the Centers for Medicare and Medicaid Services. It should notify the alternative PT provider not to report regulated analyte results to CMS.
Carol Hajkaluk, MT(ASCP)
Senior Technical Specialist
CAP Accreditation Programs
College of American Pathologists, Northfield, Ill.
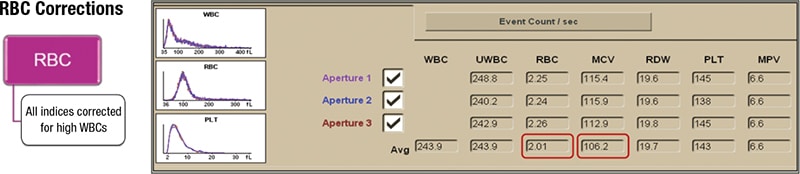
Q. A laboratory in my organization frequently receives specimens from oncology patients exhibiting hyperleukocytosis. The Beckman Coulter DxH 800 analyzer autocorrects the red blood cells, but this lab is still seeing errors on the indices (hemoglobin, mean corpuscular volume). What is the best way to correct for potential white blood cell interference on the RBC indices?
A. To ensure the accuracy and reliability of red blood cell indices, Beckman Coulter has embedded specific quality assurance programs in all its hematology analyzers to help users detect spurious results due to preanalytical phase deficiencies or due to samples presenting with significant hyperlipidemia or hyperleukocytosis.
 To detect erroneous Hgb measurements, the hematology analyzers systematically check the hemoglobin-to-hematocrit relationship using the rule of three. The analyzers then generate a flag—H & H check failed—to alert the user about the risk of providing an erroneous hemoglobin measurement. Beckman Coulter’s website offers a webinar, “The Rule of Three: Quality Assurance Procedures to Minimize the Lab’s Spurious Results,” that explains in more detail this quality assurance program. Moreover, in cases of hyperleukocytosis, the Beckman Coulter DxH 600, 800, and 900 series hematology analyzers use the DataFusion algorithm to automatically correct the hemoglobin measurement and, therefore, ensure the accuracy of this vital parameter.
To detect erroneous Hgb measurements, the hematology analyzers systematically check the hemoglobin-to-hematocrit relationship using the rule of three. The analyzers then generate a flag—H & H check failed—to alert the user about the risk of providing an erroneous hemoglobin measurement. Beckman Coulter’s website offers a webinar, “The Rule of Three: Quality Assurance Procedures to Minimize the Lab’s Spurious Results,” that explains in more detail this quality assurance program. Moreover, in cases of hyperleukocytosis, the Beckman Coulter DxH 600, 800, and 900 series hematology analyzers use the DataFusion algorithm to automatically correct the hemoglobin measurement and, therefore, ensure the accuracy of this vital parameter.
The Hgb measurement is corrected for interference by a high WBC count. The magnitude of interference not only depends on the amount of WBCs but also on the size of the white cells. Size factor is derived from the mean channel of the WBC histogram.
In cases of significant hyperleukocytosis, the DxH 600, 800, and 900 series analyzers not only correct the hemoglobin measurement but also the RBC count and MCV (figure) to ensure maximum quality, reliability, and accuracy of all the RBC parameters and indices.
Rachel D. Burnside, PhD, MBA
Senior Manager
Hematology Product Management
Beckman Coulter, Brea, Calif.

