Amy Carpenter
March 2025—The Ventana CLDN18 (43-14A) RxDx Assay, the companion diagnostic for zolbetuximab, detects the claudin 18 protein in formalin-fixed, paraffin-embedded gastric adenocarcinoma including gastroesophageal junction tissue specimens.
Gastric intestinal metaplasia can be used as a positive tissue control and system level control and should display weak to moderate membrane staining.
And cases are considered positive if 75 percent or greater of viable tumor cells demonstrate moderate or strong membrane staining or apical or microluminal staining.
Those are the takeaways of the USCAP webinar presented in December by Angela N. Baldwin, MD, MPH, pathology liaison, Division of Medical and Scientific Affairs, Roche Diagnostics. She covered staining, scoring, challenges, artifacts, and a few illustrative cases.
Two global phase three studies, SPOTLIGHT and GLOW, demonstrated that 38 percent of patients with advanced gastric-GEJ cancers are CLDN18.2 positive and may be candidates for zolbetuximab (Vyloy) with chemotherapy.
Biomarker testing has revealed four distinct molecular gastric and GEJ cancer patterns: chromosomal instability (CIN), Epstein-Barr virus (EBV), microsatellite instability (MSI), and genomically stable. “About 20 percent of all gastric and GEJ tumors are genomically stable and commonly contain a claudin 18 aberration,” Dr. Baldwin said.
“Biomarker prevalence can vary among studies owing to tumor heterogeneity, differences in patient population, clinical trial methodology, and the diagnostic assays used,” she said. PD-L1 positivity in gastric and GEJ cancer ranges from 16 to 73 percent depending on the combined positive score cutoff used. HER2 is positive in 22 percent of cases, and four percent of cases are MSI-high. Two novel biomarkers—fibroblast growth factor receptor 2b (FGFR2b) and CLDN18.2—are positive in 30 and 36 percent of cases, respectively.
PD-L1 inhibitors and chemotherapy are the only first-line options for HER2-negative, advanced gastric and GEJ cancers. Treatment options for second line and later are even more limited with minimal overall survival benefit.
CLDN18.2 is the dominant isoform in healthy gastric tissue and may be retained in malignant transformation and overexpressed in malignant pancreatic and esophageal tissue. CLDN18.1 is expressed in normal and malignant lung tissue.
“Studies have shown that claudin 18.2 may become more exposed and accessible to antibodies as gastric tumors develop,” Dr. Baldwin said. In gastric epithelial cells, CLDN18.2 is typically buried in the tight junction supramolecular complex, regulating selective barrier properties and contributing to cell-to-cell epithelial adhesion. Malignant transformation leads to polarity disruptions and structural loss. As a result, CLDN18.2 may be more exposed and accessible to antibodies. Its presence is retained throughout malignant transformation.
Several studies of gastric and GEJ cancers have used the Ventana CLDN18 assay to demonstrate CLDN18.2 positivity ranging from 24 to 38.5 percent. “No clear differences have been observed in the prevalence of common biomarkers between claudin 18.2-positive and -negative patients,” Dr. Baldwin said, “with both groups demonstrating similar levels of HER2, PD-L1, MSI, and EBV positivity.”
Zolbetuximab, approved by the Food and Drug Administration last fall, targets CLDN18.2, tagging gastric and GEJ cancer cells for destruction by activating cytotoxic immune effector functions.
Zolbetuximab does not bind closely to claudin 18.1, reducing the risk of off-target effects outside of gastric carcinoma tissue. Patients with greater than or equal to 75 percent of gastric adenocarcinoma and/or GEJ tumor cells demonstrating moderate to strong membrane staining above background are considered eligible for treatment with zolbetuximab.
The clone 43-14A is a murine monoclonal antibody. Nonneoplastic gastric epithelium presents with strong membrane staining with CLDN18, and there will be an absence of CLDN18 staining in lamina propria, lymphocytes, smooth muscle, blood vessels, and peripheral nerve tissue. Gastric tumor cells may show an absence of staining or weak to strong staining.
Human gastric tissue with intestinal metaplasia can be used as a positive tissue control as well as system level control with these criteria: The system level control should be stained, fixed, and processed in the same manner as patient specimens; be run for each set of test conditions to monitor proper functioning of the reagents and instrument within the staining run; and consist of fresh specimen tissue prepared and fixed as soon as possible in a manner identical to the test sections. It may contain both positive and negative staining elements and serve as both the positive and negative control tissue.
“Various elements of the gastric tissue with intestinal metaplasia must be evaluated to determine if it’s an acceptable system level control,” Dr. Baldwin said. For positive staining, the control is acceptable if there is strong membrane CLDN18 staining in the normal gastric epithelial cells and the presence of weak to moderate membranous CLDN18 staining of epithelial cells in areas of metaplasia. For negative staining, there must be an absence of CLDN18 staining in the lamina propria, lymphocytes, smooth muscle, blood vessels, and peripheral nerve tissue for the system level control to be deemed acceptable. Any deviation makes the control unacceptable.
A negative reagent control must be run for every specimen. “A tissue section will be stained with the negative monoclonal control as a negative reagent control to evaluate any nonspecific staining,” Dr. Baldwin said, to ensure that the observed staining is specific to the target antigen and not a result of nonspecific interactions or artifacts.
Before evaluating a slide stained with the Ventana CLDN18 antibody, the case-matched negative reagent control slide must be assessed first and exhibit acceptable staining—that is, negative for specific staining—for the corresponding CLDN18 (43-14A) slide to be considered evaluable.
The scoring algorithm for the Ventana CLDN18 assay is based on the interpretation of two parameters from stained tissue slides: the percentage of viable tumor cells stained and staining intensity.
The recommended scoring approach begins with a review of the H&E-stained slide to ensure a minimum of 50 viable tumor cells and notation of the location of glands, signet ring cells, diffuse tumor cells, or areas of necrosis.
“Next review the claudin 18-stained slide at a low power to determine the overall extent of staining,” Dr. Baldwin said. “Then go down to a higher power of 10× or 20× to examine the tumor for claudin 18 staining.” Confirm areas of moderate to strong membrane staining and exclude areas of weak staining, necrotic debris, or mucin staining. “Finally, go back up to 4× to visually estimate the percentage of claudin 18-positive viable tumor cell membrane staining, taking heterogeneity into account,” she said.
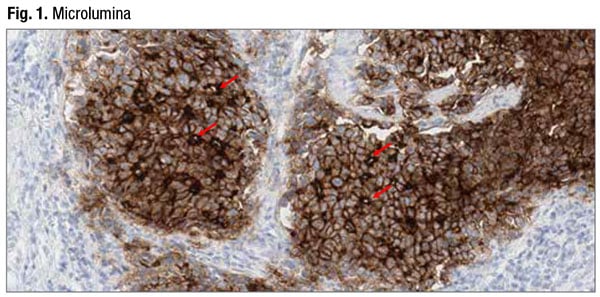
The staining may be heterogeneous, varying from tumor area to tumor area, field to field, and even within the same gland, Dr. Baldwin noted. The full spectrum of staining intensity may be present—absent, weak, moderate, and strong. A staining pattern may be apical, circumferential (partial or complete), basolateral/lateral, or microluminal. All these patterns are included in scoring, but only moderate to strong staining is counted toward total percent tumor cell membrane staining. In Fig. 1 is an example of microluminal pattern staining. “Microlumen pick up the stain and form brown dots,” Dr. Baldwin said. “Any tumor cell in contact with the brown-staining lumen is considered positive.”
Claudin 18 can stain nonneoplastic epithelium, but the presence of nonneoplastic epithelium is not required on the specimen or case slide, Dr. Baldwin said. If it is present, nonneoplastic epithelium should show strong 3+ staining; if not, the staining procedure must be repeated.
Gastric intestinal metaplasia, if present on the case slide, may show from zero or no staining to 3+ strong staining, she said. “Squamous mucosa of the gastroesophageal junction should have no staining.”
Dr. Baldwin shared two case examples to highlight the differences between low and high tumor-cell-density positive cases.
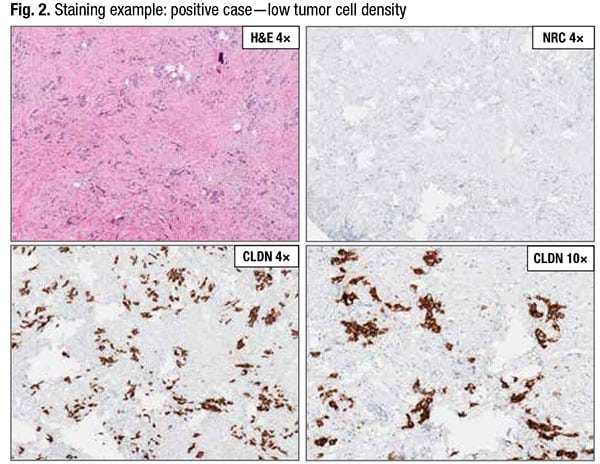
In Fig. 2, the H&E-stained slide at low power (top left) has relatively low tumor density. The negative reagent control slide (top right) is acceptable with absence of any specific staining. The CLDN18 slide at low power (bottom left) “is where we will visually estimate the percent of positive-staining tumor cells,” Dr. Baldwin said, and the CLDN18 slide at higher power (bottom right) “is where we will be able to detect any necrotic debris or mucin picking up the stain and exclude it from our overall tumor cell percentage.” Tumor cells with weak membrane staining would be excluded. This case exhibits moderate to strong membrane staining in 95 percent of tumor cells and is considered a positive case.
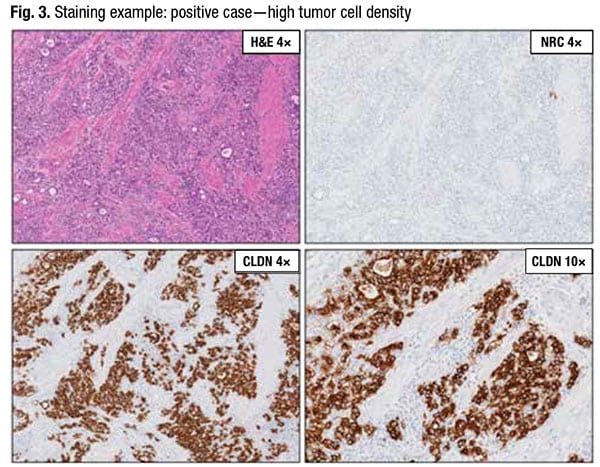
In contrast, Fig. 3 is a positive case of high tumor cell density (top left). The negative reagent control slide (top right) is acceptable with absence of specific staining. In the CLDN18 slide at low power (bottom left), the majority of tumor cells appear to be positive. In the CLDN18 slide at higher power (bottom right), “there is no necrotic debris, mucin, or weak-staining tumor cells for us to exclude from the score,” Dr. Baldwin said. This case, too, exhibits moderate to strong membrane staining in 95 percent of tumor cells and is positive.
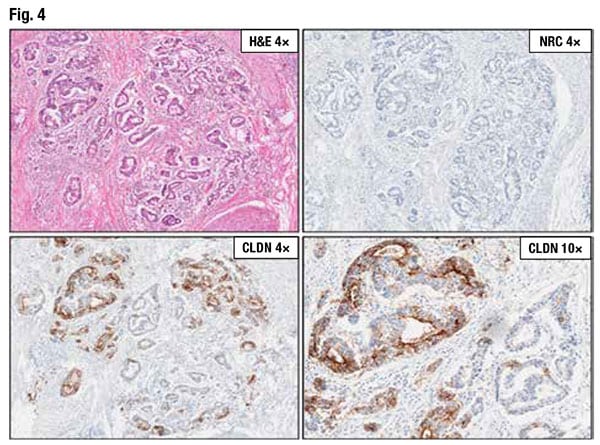
In Fig. 4 numerous malignant glands are visible on the H&E-stained slide (top left). The negative reagent control (top right) is acceptable with absence of specific staining. The CLDN18 slide at low power (bottom left) reveals that more than half of the tumor cells are staining. At higher power (bottom right), there is necrotic debris staining in the gland (top left of bottom right image), which will be excluded from the final score.
“This case presents a bit of a challenge because it is close to the cutoff of 75 percent,” Dr. Baldwin said, and thus should be evaluated by a second pathologist. It’s a case of moderate to strong membrane staining in 60 percent of tumor cells and is considered negative.
In Fig. 5 are nonneoplastic glands staining “as expected with strong 3+ membranous staining,” Dr. Baldwin said.
Also seen are lymphocytes with absence of staining as expected. There are diffuse tumor cells (bottom panel) with predominantly weak and occasionally moderate membrane staining.
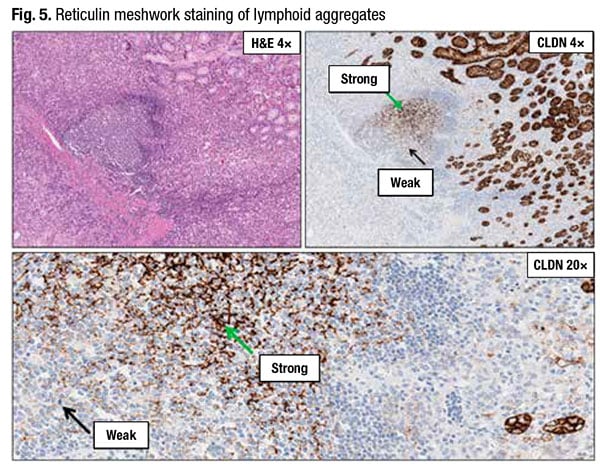
The potential pitfall is the strong staining in the reticulin meshwork of the lymphoid aggregates. “When you see strong linear or chicken-wire-type staining near a lymphoid aggregate, investigate further to see if it is the reticulin meshwork picking up the claudin 18 stain,” Dr. Baldwin advises.
Another challenge: Capillaries with prominent endothelium can mimic tumor cells (Fig. 6). (Black arrows in the H&E at 20×, top right, point to capillaries, some of which have red blood cells inside.)
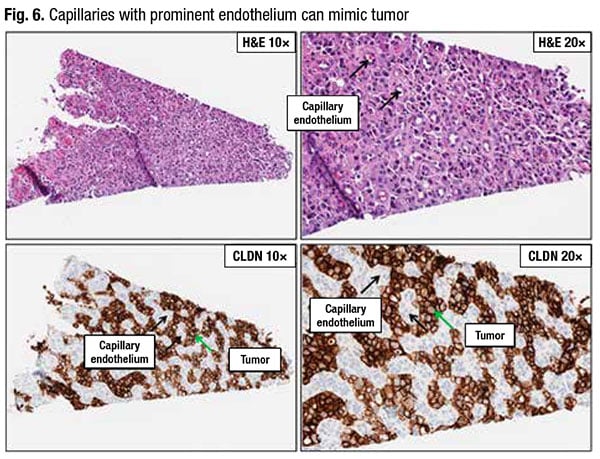
“On both the H&E and claudin slides, the capillary endothelium easily blends in with the tumor cells,” Dr. Baldwin said. “However, on the higher power claudin 18 slide on the bottom right we see the moderate to strong membrane staining of the tumor cells while the capillary endothelial cells do not stain.”
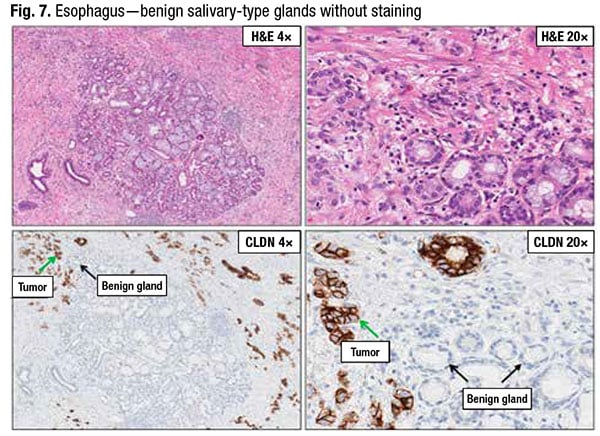
In Fig. 7 Dr. Baldwin noted that benign salivary-type glands of the esophagus may be confused with malignant glands. “But note that these benign glands do not stain with claudin 18,” she said.
Squamous mucosa of the esophagus does not stain with claudin 18. However, gastric tumor cells in the gastroesophageal junction may infiltrate into the squamous epithelium. In Fig. 8, positive staining claudin 18 tumor cells may be seen within the squamous mucosa.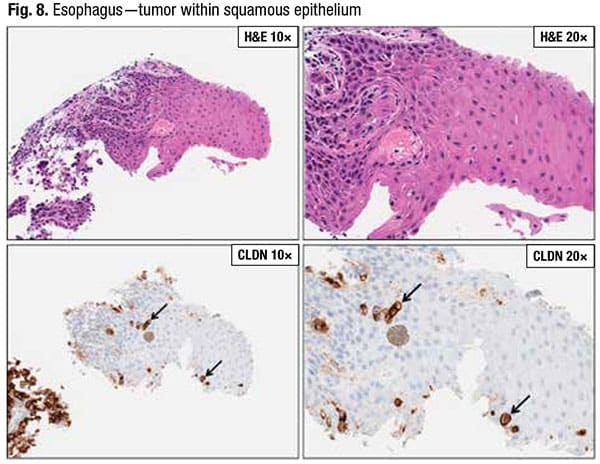
Dr. Baldwin closed with a few words about assay artifacts:
- Inflamed stromal staining can be seen in association with malignant glands. “This is usually minimal and does not interfere with the interpretation of the CLDN18 assay.”
- Focal DAB trapping and blush-like staining of smooth muscle can occur but should not interfere with the interpretation of the CLDN18 assay.
- Parietal cells may have a cytoplasmic blush. This is considered acceptable.
Amy Carpenter is CAP TODAY senior editor.

