Karen Titus
July 2025—For all the words that have been devoted to the topic of how to meditate, the path to enlightenment can be disarmingly simple: start by noticing. Likewise, there is a fairly simple set of instructions for untangling one of the vexing problems associated with immune checkpoint inhibitors. Though the drugs can create impressive antitumor response, they can also lead to immune-related adverse events (known as irAEs), including colitis and gastritis, which manifest as histologic changes that can be seen in both the upper and lower gastrointestinal tract. Left unaddressed, severe reactions can disrupt treatment.
It’s complicated. And it’s not. The first step, says Raul S. Gonzalez, MD, professor of pathology and laboratory medicine and director of the gastrointestinal pathology service, Emory University School of Medicine, is cultivating a certain mindfulness. “We need to have knowledge that checkpoint inhibitors can cause immune-related adverse events such as gastritis and colitis, to be aware that these things happen,” says Dr. Gonzalez, who is also co-director of anatomic pathology research at Emory and chair of the CAP Surgical Pathology Committee. “It’s just one more diagnosis to add to our list of considerations when we are looking at gastrointestinal tract mucosa biopsies.”
And beyond that? “Broadly speaking,” he says, “I would not say we do anything unique or special for these cases.”
And Thich Nhat Hanh was just a peaceful dude.
Nevertheless, even as the stakes of using immune checkpoint inhibitors, or ICIs, remain high, the way forward doesn’t have to be convoluted, say experts in the field.
Immune checkpoint inhibitors increase T-cell antitumor activity but lead to systemic toxic effects.

Though immune-mediated colitis is a relatively new phenomenon, it can also seem familiar, says Yinghong Wang, MD, PhD, director of the Oncology-GI Toxicity Program and of fecal microbiota transplantation at MD Anderson Cancer Center and chair of its immunotherapy toxicity working group. “The endoscopic and histological features are more or less overlapping with inflammatory bowel disease, but not exactly the same,” says Dr. Wang, who is also professor on tenure track, Department of Gastroenterology, Hepatology, and Nutrition.
Until now, clinicians have been using the IBD scoring system (the Mayo endoscopic subscore, or MES) to assess immune-mediated colitis, though it’s an imperfect tool at best. Immune-mediated colitis includes a wider range of endoscopic and pathologic features than IBD, she says, which has prompted her and other specialists in the field to develop a more targeted scoring system (Wang Y, et al. Gastrointest Endosc. 2024;100[2]:273–282.e4). Dr. Wang is currently using it in research settings, as well as in a follow-up validation study at MD Anderson.
From Dr. Wang’s perspective, the main question for pathologists, oncologists, and internal medicine specialists boils down to: Are these pathological findings consistent with immunotherapy toxicity? Because the spectrum is so broad, she says, it’s not surprising that there may not be widespread familiarity with immune-mediated colitis.
Histologic inflammatory features can include apoptosis, neutrophilic inflammation of crypts and crypt abscesses, crypt architectural distortion, granulomas, and increased intraepithelial lymphocytes. Any of these findings could raise the question of whether they’re part of an immunotherapy toxicity presentation. Eosinophilic colitis can have the same presentation, for example, as can graft-versus-host disease, with its many apoptotic bodies. “It’s a confusing picture,” says Dr. Wang.
All of these can also indicate immunotherapy colitis, she says, “if the clinical context is very suspicious and the temporal relationship is very close. So you have to put all the other factors together to increase your success of looking at your differential diagnosis.”
Only within the overall clinical picture does it become easier to sort this out, she says. If the patient is receiving immunotherapy, did they have a preexisting inflammatory bowel condition? And what is the time onset of the flareup?
“Those are the things that give me some good correlations,” Dr. Wang says.
Dr. Gonzalez concurs that patient history is important when considering the possibility that a colitis or gastritis is caused by an immune checkpoint inhibitor.
“Usually that information is available, but obviously if we don’t know a patient is on one of these medications, it becomes more challenging to suggest in the differential diagnosis that an injury is being caused by them.”
Even as the shore of certainty recedes, however, there is a typical scenario in which he’ll strongly consider an irAE in his differential. He pays particular attention in the case of older patients, who are more likely to be receiving cancer treatment. (“Though, as we sadly know, it’s becoming more common in younger patients,” Dr. Gonzalez says.) And with a new-onset colitis or other gastrointestinal tract injury, Dr. Gonzalez will take note when “the biopsy shows findings that I have informally described to my trainees as, ‘Reminiscent of inflammatory bowel disease, but a little funny.’”
He says pathologists should first ask if a colitis biopsy is IBD or something else. (Though gastritis and esophagitis are also possibilities, he says colitis is the most common type of immune checkpoint inhibitor injury pathologists are likely to encounter.) Ulcerative colitis, Crohn’s disease, and IBD tend to first manifest in younger patients (teens, 20s), he says. But in older patients, if he sees a new colitis, with features that would prompt him to think of an IBD diagnosis—cryptitis, architectural distortion, etc.—he also considers that aforementioned “funny.”
“Checkpoint inhibitors can cause certain patterns of injury,” Dr. Gonzalez explains, such as granulomas, increased apoptotic bodies, and increased intraepithelial lymphocytes. “If I see any of those in something that otherwise might make me think of IBD, then I have to start thinking of other possibilities.” Given the prevalence of immune checkpoint inhibitors being used clinically, “That’s where my mind goes first.”
All of these findings can be variable, Dr. Gonzalez continues. Some pathologists might struggle with identifying increased apoptosis if it’s subtle, for example. “Sometimes I’ll see a cluster of macrophages and debate whether I think it’s truly a granuloma—that sort of thing,” he says. “But I think most pathologists can at least identify that this colon biopsy is injured; something is going on—let me see if I can figure it out.”
Generally speaking, the differential of colitis can be challenging. “Saying ‘colitis’ is not hard,” he says. But because ICI colitis usually mimics IBD, it’s not an open-and-shut case. And while older patients are more likely to have immune checkpoint inhibitor colitis, they’re also more likely to develop segmental colitis associated with diverticulosis, which can also mimic IBD and other forms of colitis. It’s usually not quite the same pattern as that seen in ICI colitis, but there is overlap, with symptoms as well as age.
“It’s rare for me to confidently be able to say, ‘Yes, this is definitely immune checkpoint inhibitor colitis—we’re done here,’” says Dr. Gonzalez. “I usually have to give some degree of a differential, unless clinically they’re saying this has to be from a patient’s medication, and then I get a perfect histologic correlation. That would be the only time I can be close to 100 percent.”
The endoscopic scoring system is less relevant to pathologists, since it’s generally more of a clinical tool than a histopathologic reporting tool. Dr. Gonzalez says he always reviews the score and the endoscopy note when reading the biopsy, “though it generally does not factor too heavily in my report, unless there’s a major discrepancy.” If the score is high and the colon looks normal, for example, or vice versa, “then I may have to figure out if something unusual has happened.”
Dr. Gonzalez does appreciate a detailed endoscopy report, however. Ideally, it will describe the clinical concern and the possible clinical differential and mention all the biopsy sites. “As with humans in any setting,” he says, “some gastroenterologists are going to be more detailed in their reports than others.”
At Emory, Dr. Gonzalez and his pathologist colleagues see a high volume of colon biopsies. In this daily workflow, gastroenterologists are used to putting their reports into electronic medical records quickly, and pathologists are used to reading them. “Generally we know what’s going on—we know how to parse the reports; we know how to incorporate the findings into our final pathologic diagnosis.
“Nothing is coming out of left field,” he says.
Pathologists who see fewer of these biopsies will face a steeper learning curve, he acknowledges. But it’s navigable.
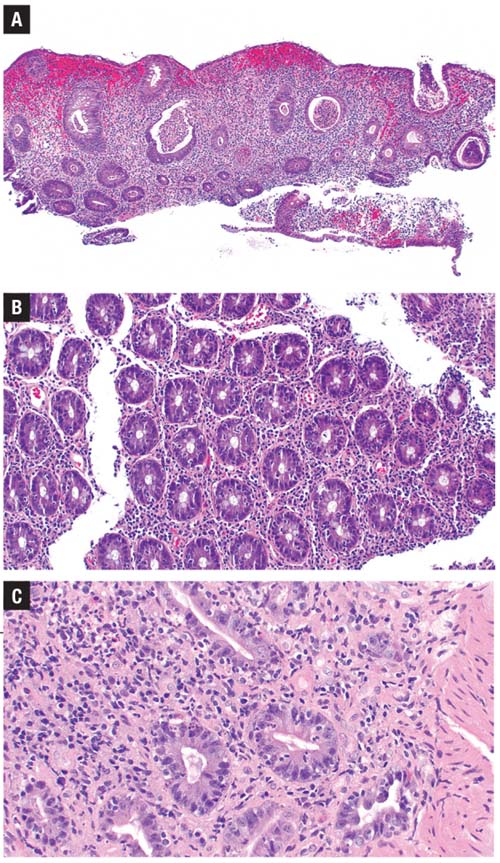
cases of PD-1 inhibitor colitis.
A) There is significant histologic overlap with inflammatory bowel disease. B) This example shows increased intraepithelial lymphocytes in addition to neutrophilic cryptitis.
C) Here, increased apoptotic
bodies are present.
“I think that any pathologist who sees a decent volume of colonoscopy biopsies is perfectly capable of recognizing these injury patterns once they become familiar with them,” Dr. Gonzalez says, which will allow them to include the possibility of ICI colitis in the differential.
He continues: “I recognize that depending on the practice setting, not all pathologists will be able to obtain a robust clinical history—and again, if you don’t know what medications the patient is taking, it becomes harder to offer a firm diagnosis.”
But he points to a handful of published papers that provide descriptions and photomicrographs of what ICI injury looks like under the microscope in these settings (Zhang ML, et al. Am J Surg Pathol. 2022;46[1]:e15–e26; Tomm NK, et al. Hum Pathol. 2024;148:60–65; Ma C, et al. J Immunother Cancer. 2022;10[3]:e004560; Gonzalez RS, et al. Histopathology. 2017;70[4]:558–567; Oble DA, et al. Am J Surg Pathol. 2008;32[8]:1130–1137). “So awareness of the disease and awareness that it should be incorporated mentally when evaluating a colitis biopsy—that is all any gastrointestinal pathologist needs to do to properly evaluate these cases.”
Dr. Wang shares Dr. Gonzalez’s commitment to thorough reports.
Details matter. When she looks at the pathology report, “I like to see all the pathological features,” she says—neutrophils, eosinophils, lymphocytes, architecture distortion—“so I know, Oh, this is a chronic type of feature, this is acute, this is microscopic.” Less helpful to her are reports that simply say, for example, “There is a chronic colitis.” Specific details may also be useful for clinicians who aren’t necessarily GI specialists. Don’t hold back, in other words. “I really appreciate [when] pathologists can share their thoughts and detailed descriptions on the pathological findings when any pathological samples can be read in more detail. That is a huge help for me, rather than a one-sentence, general description.”
(She offers an additional point of clarification. In the past, physicians used the term inflammatory bowel disease to refer specifically to autoimmune GI inflammation—by default, that included conditions such as ulcerative colitis and Crohn’s disease. But with immune-mediated colitis becoming widely recognized, she says, she’s noticed some physicians are labeling it as an IBD. While that may be correct from a general perspective, “We don’t want people to get misled” about the cause. “This is not an autoimmune etiology; it’s a drug-induced etiology.”)
Dr. Wang says she uses the pathology report primarily to help guide treatment. Given the overlapping symptoms with IBD, “My diagnosis most of the time is made based on the clinical presentation, the temporal relationship to the immunotherapy, the positive stool inflammatory markers,” and, when it’s available, imaging.
The tissue sample can confirm the presence of histological inflammation. Dr. Wang notes, however, “There is a small cohort of patients who present with very aggressive diarrhea symptoms, but they do not have histological inflammation.” Nevertheless, these patients may still have immunotherapy-induced toxicity, even though the pathology report indicates no inflammatory cell infiltration. She and others are at a loss regarding the pathophysiology of these cases, but she’s hopeful that translational research analysis might eventually provide more information.
When a patient is identified as having moderate to severe colitis, she says, “most of the time we like to be on the aggressive side with treatment.” Otherwise, the chance of recurrence is high, which often delays cancer treatment.
When Dr. Wang first came to MD Anderson, about eight and a half years ago, she says the standard practice was often to treat patients’ colitis with steroids, which would then be tapered off once the patients started to improve. But that led to high rates of recurrence. “And once a patient’s CTCAE [Common Terminology Criteria for Adverse Events] is grade two or three, the immunotherapy often is not resumed.”
The goal, she says, is to modify and optimize immunotherapy once the colitis is resolved. “If that patient has a good response to immunotherapy, we want to get patients back on it because they may benefit from it long term, especially if it’s a long-term maintenance treatment,” Dr. Wang says.
“So that’s now the goal of management,” she continues. “First, to decrease the recurrence; second, to be able to let the patient get back on the immunotherapy successfully after the colitis event.”
“Those are two very ambitious goals,” Dr. Wang concedes. But eight-plus years into her tenure at MD Anderson, “I think we are able to achieve them. We are able to introduce the biologic agent early so the patients can usually get into remission much faster and with a lower chance of recurrence.”
The other strategy has to do with rechallenging patients. “Because we are now able to manage these patients more effectively,” Dr. Wang says, “we are able to push for ICI rechallenge early on in the disease course if the patient has that need from a cancer perspective. And we will either provide concurrent biologic treatments to allow the rechallenge to be a little less bothersome or offer a fecal transplant.” The latter can be highly effective, Dr. Wang says, with patients often achieving clinical and endoscopic remission.
Endoscopy evaluation within the first week to 30 days, therefore, “can be very, very beneficial to guide treatment, rather than waiting until the patient has had several refractory disease courses,” she says.
The histology is not the end-all and be-all, but it’s a critical first step. “Pathology has a major role in identifying the toxicity per se, not only in GI but all sites,” Dr. Wang says. Indeed, part of her daily communication is with other clinical specialists, given that patients can have multiorgan toxicities. The treatments involved in colitis could have an impact on other organ sites and can affect her choice of whether to use more steroids, for example. There’s also interest in exploring whether fecal transplant, and its effect on the microbiome, can help with other organ toxicities, she says.
But none of these advances will unfold without that first, “nothing special” pathology testing.
Dr. Gonzalez notes that ipilimumab, a CTLA-4 inhibitor, was the first ICI to be implicated in noticeably higher rates of colitis, some 15 to 20 years ago. More recently—roughly in the past 10 years, he says—PD-1 and PD-L1 inhibitors, starting with nivolumab and pembrolizumab, expanded the picture.
And now? “There are so many coming out that it’s not as easy to keep track,” Dr. Gonzalez says. “Every now and then I have to check: Oh, there’s a new inhibitor.” Generally speaking, they cause similar possible patterns of colitis, he says, although it can differ from patient to patient, and even differ in the same patient over time.
This is no longer a niche topic. Because the drugs are so effective, many patients are on them. This, in turn, “means a larger number of people are going to be developing immune-related adverse events,” says Dr. Gonzalez.
“This isn’t going away,” he says. “This is an exciting area of cancer therapy. These patients are on these medications for a reason. It would be good if they could be kept on the medication until the regimen is completed.”
Karen Titus is CAP TODAY contributing editor and co-managing editor.

