Charna Albert
July 2025—It was one of those infamous winter days in Chicago. The phlebotomy staff were calling off work, and Gregory S. Retzinger, MD, PhD, had reached his breaking point.
Phlebotomists tend to be in short supply, and Northwestern Memorial Hospital, where Dr. Retzinger is medical director of pathology clinical services, is no exception. The weather made a bad problem worse. “We were backed up in our diagnostic testing center,” he recalls. “We couldn’t collect samples from our outpatients. I said, ‘We can’t keep going through this. We have to have a solution.’”
The Vitestro Aletta Autonomous Robotic Phlebotomy Device (ARPD for short) might be that solution. The device earned a CE mark one year ago, and Vitestro chief commercial officer for North America Bob Gerberich says it has been used in more than 6,000 patient blood draws and is in routine clinic use in several European countries. (Vitestro, a medical robotics firm founded in 2017 in the Netherlands, named the device after Dr. Aletta Jacobs, the first female Dutch physician, who died in 1929.)
Now Vitestro is taking its technology stateside. Northwestern Medicine, Mayo Clinic in Rochester, and Baylor Scott & White Health will serve as premarket clinical study sites in advance of U.S. regulatory authorization and commercialization.

The Aletta ARPD came on Dr. Retzinger’s radar not long after that challenging day in Northwestern Memorial’s diagnostic testing center. Vitestro was already looking for hospital sites for its U.S. clinical studies when he got in touch. “I just happened to call at the right time,” he says.
The most recent data from Vitestro’s ADOPT trial in Europe, presented at this year’s Executive War College conference, demonstrated a first-time venipuncture success rate of 95 percent, a hemolysis rate of less than 0.1 percent, zero device-related adverse events, and a patient acceptance rate of 98 percent (89 percent rated it as very acceptable or acceptable, while eight percent were neutral; 85 percent indicated it was comparable to or less or far less painful than manual phlebotomy). The median time of procedure from application of tourniquet to bandaging was two and a half minutes.
At Northwestern Memorial, the first-stick success rate is about 92 percent, Dr. Retzinger says, meaning the device would be an improvement over “already pretty good numbers.” But his motivation for partnering with Vitestro is the staff shortage.
“The biggest thing for us is we need personnel,” he says. “We want accuracy and precision and all of those things, but we also need to get the samples collected.”
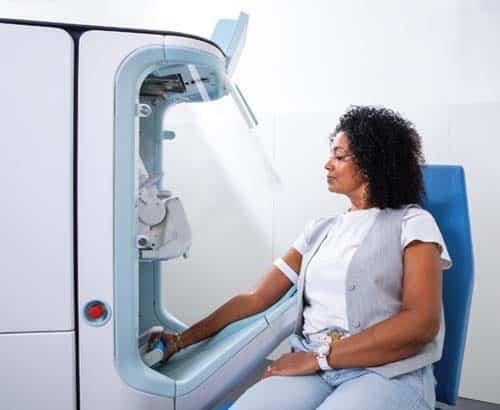
Gerberich says the device is designed to perform every aspect of the blood draw procedure autonomously. This includes positioning and preparing the arm, drawing the blood, changing and inverting (mixing) the tubes, putting pressure on the venipuncture site, and applying the bandage. It uses a multimodal imaging approach, combining near-infrared and ultrasound imaging to identify the optimal vein, with a “complement of Doppler ultrasound” as an extra guardrail and final confirmation that the device has found a vein rather than an artery. “It then is designed to use a combination of AI and robotics to insert the needle precisely in the vein for optimal accuracy,” Gerberich says.
The device is engineered so that the patient isn’t able to see the needle as it is inserted. “The patient never sees the blood leaving their vein or collecting into the tubes, and they never see or feel the tubes being changed,” Gerberich says. And when the needle is inserted, the device is designed to eliminate the micro movements or inconsistencies in force that might be experienced with manual phlebotomy. “That’s what contributes to making it more comfortable for the patient.”


As it scans, the device sprays alcohol to enable acoustic imaging. “Once it identifies the optimal vein, the vein is stabilized to help prevent it from rolling,” Gerberich says. After the robotic arm inserts the needle, the device detects blood flow and initiates the draw. As each tube fills, it is automatically replaced with a new one. “And the device continues to fill and invert the tubes internally,” he says.
Among the institutions that have contracted for or are using the ARPD are Amsterdam University Medical Center, St. Antonius Hospital, OLVG Lab, Result Laboratorium, and Deventer Hospital in the Netherlands and Odense University Hospital in Denmark.
Vitestro hopes to initiate in early 2027 a U.S. multicenter study that will run for about five months. “Our priority will be to provide the highest level of technical and clinical support to our clinical sites throughout the trial,” Gerberich says. “The additional clinical studies performed in the U.S. are designed to support commercialization upon FDA authorization.” Vitestro hopes to submit its FDA application in the latter half of this year.
Brooke Katzman, PhD, co-director of the hospital clinical laboratory, point of care, and the central clinical laboratory and director of laboratory services at Mayo Clinic in Rochester, will lead the multicenter clinical trial coordinating data collection and overseeing manuscript publication. Mayo Clinic will receive two ARPDs, she says. Both will be placed in the hospital’s major outpatient blood collection center. “We see about 1,000 patients a day there, so we think that’s an ideal location for it.”
The study will take place in two phases, Dr. Katzman says, with the first “akin to a typical instrument validation,” evaluating paired collections in healthy participants. “It’s important to demonstrate that if we were to use this device in our patient population, that we trust the results,” she says.

Most of the tests they assess will be what she calls “bread and butter tests,” or “tests that the majority of people who go through our outpatient collection areas may have ordered. We want to know those look comparable.” One variable they’ll look at is fill volume. “We know fill volume is important for coagulation testing, so that’s something we want to see—if there’s a difference in how those tubes get filled.” They’ll assess specimen integrity parameters in addition to laboratory values, she notes, starting with first-stick success rate. “Is it as good as what our phlebotomists can do, or comparable, or close? That’s a question we plan to answer,” she says.
Some of the tests they chose for the study were selected because they are more susceptible to issues of specimen integrity, such as lactate dehydrogenase, which is particularly prone to hemolysis, she notes. They also plan to assess different tube types, “probing the different tests based on some of the preanalytic components that could confound results.”
The second phase of the study will involve a single collection via the ARPD. “It will be for patients who have clinical orders already placed,” she says, “and they will be consented and collected via the robot, instead of manual phlebotomy.” They’ll evaluate the same specimen integrity parameters, including first-stick success rate and rate of hemolysis, and take note of any adverse events. “We’re going to monitor that closely,” she says.
Vitestro envisions one supervisor overseeing up to three ARPDs at a time, she says, though for the Mayo Clinic study, the ratio will likely be one supervisor to one device. “It may not be a true reflection [of the workflow] because we’re having to consent,” she says. “But ideally you would have one person who can manage multiple devices at the same time.”
At Northwestern Memorial, the wait for a blood draw can be lengthy.
Dr. Retzinger hopes avoiding that wait will incentivize patients to participate in the study. “One of the perks of automated phlebotomy is, if the machine is open now, you can wait for a human to become available, or you can have your blood drawn by the instrument,” he says. He expects Northwestern Memorial to receive two devices for the study.
One use case for the device would be emergency department triage, where staffing is short. Yet staffing isn’t the only phlebotomy-related concern in the ED. Hemolysis rates in the ED, Gerberich says, can be as high as 30 percent. “And when you’re doing hundreds of thousands of blood draws annually, that turns into thousands of patients impacted,” he says. “We envision moving from centralized outpatient draw centers to the emergency department, where the need is great, not only because of lack of availability of phlebotomists but because of the quality of the sample.” The current version of the device, he notes, may be well suited to ED triage.
True inpatient use, on the other hand, would require a mobile version of the ARPD, Dr. Katzman says. “It’s large as it is, and it would be difficult to go between rooms.”
Says Gerberich, “We see the potential for a modified version for inpatients in the pipeline, with potential applications beyond diagnostic blood draws. You can envision anything from IV placements, CVC placements, blood donation—there’s all sorts of opportunities for this core technology.” From a development perspective, he says, the difficult part was the vascular access aspect of the technology. “Identifying the optimal vein and inserting the needle with such precision is what opens the door to future applications.”
A more immediate need, and the company’s first order of business, is laboratory information system connectivity. Without it, Dr. Katzman says, “you’re doing a lot of manual, error-prone activities.”
Draw order error is the type of human mistake they hope to avoid, Gerberich says. As of now, “it’s the responsibility of the trained health care professional who labels the tubes in the cartridge to put them in the correct draw order,” he says. “With LIS connectivity, we want to integrate the ability to confirm the correct draw order.” The LIS interface would allow for the added layer of patient identity confirmation. And they plan to integrate fill-level confirmation, too, with built-in fill-level sensors. “All of these things are necessary to keep it a 100 percent autonomous operation,” he says, noting that Vitestro has “connectivity plans with all the major LIS providers.”
On Dr. Retzinger’s wish list for the future is automated sample transport via a pneumatic tube integrated directly into the device. The tubes now collect within the device in bins after the samples are drawn and must be transported to the laboratory periodically. “Our vision is to continue to innovate and work with our hospital and health system partners to have an integrated tube delivery system within the device,” Gerberich says. “But that’s very much the future.”
Dr. Retzinger would also like to eliminate the step in which the physician or health care professional gives the patient the tubes. Rather, the tubes would be integrated into the machine and tube selection automated. Eliminating the need for human intervention, he says, means fewer opportunities to mislabel or misplace samples. He grants that errors don’t occur all that often now. “But when they do, it can be really problematic.”
LIS connectivity was the question on everyone’s mind at the 2025 Executive War College, where Dr. Katzman co-presented a session on the device to a packed room. “For us, and probably for most health systems, it will need to be integrated into the LIS,” she says. “That was the biggest question—how does it interface? How do orders get across?”
Ultimately, the unanswered questions couldn’t dampen the enthusiasm in the room, and it’s easy for her to see why. “It’s the newest thing to happen in phlebotomy in the last 70, 100 years,” she explains. In a field where little seems to change, “this is a huge change.”
For patients difficult to draw, the ARPD offers an experience different from manual phlebotomy, Gerberich says.
In the most recent data from the European trial presented at the Executive War College, the ARPD demonstrated a 99 percent first-time venipuncture success rate in the subgroup that self-reported as difficult to draw. In contrast, the first-time success rate for manual phlebotomists is “between 75 and 80 percent in that patient population,” Gerberich says, adding, “Difficult intravenous vascular access patients can be a challenge for the most experienced phlebotomists.”
Then there are patients with high body mass index and deep-set veins. With this population, he says, “it’s the ultrasound that’s the differentiator. Number one, it’s colorblind, so skin tone and color doesn’t impact it. And it is designed to identify a vein deep into the tissue easily and accurately.”
The European experience has also found, perhaps counterintuitively, that the older patient population was quicker to accept the device. “They’ve had years of experience having their blood drawn, so they’ve lived the challenges,” Gerberich says.
In a recent unpublished U.S. patient acceptance study involving 86 outpatient participants, he says, when asked about having their blood drawn with the ARPD, “93 percent of patients were very willing, willing, or neutral after seeing the device. Additionally, 99 percent of patients responded very positively, positively, or were neutral toward the device, and 93 percent expressed complete trust, trust, or neutral trust in it.” Vitestro plans to prepare a comprehensive overview of this study for publication.
Dr. Retzinger anticipates phlebotomists moving into the role of overseeing ARPD devices.
“There will be a more intellectual component to the phlebotomist in the future,” he says. “There will still be individuals involved in phlebotomy—just not the way they are now.”
There’s one thing that won’t change, in his view. “Phlebotomy is the face of the laboratory, as far as patients are concerned,” he says. “Nobody comments on the goodness or badness of your sodium, potassium, BUN, and creatinine. They do comment about your phlebotomy services.”
For those in the laboratory, automated phlebotomy might mean greater accuracy and precision, or even doing away with one of the last manual processes in laboratory medicine.
For patients? It’s shorter wait times on a winter day.
Charna Albert is CAP TODAY associate contributing editor.

