CAP TODAY and the Association for Molecular Pathology have teamed up to bring molecular case reports to CAP TODAY readers. AMP members write the reports using clinical cases from their own practices that show molecular testing’s important role in diagnosis, prognosis, and treatment. The following report comes from Massachusetts General Hospital and Harvard Medical School. If you would like to submit a case report, please send an email to the AMP at [email protected]. For more information about the AMP and all previously published case reports, visit www.amp.org.
Ting Zhao, MD
Robert P. Hasserjian, MD, PhD
Ivan A. Chebib, MD
Adam S. Fisch, MD, PhD
July 2025—We report the case of a 75-year-old female who initially presented with a 10-cm left pretibial mass on MRI. A biopsy revealed a high-grade spindle cell sarcoma with myofibroblastic differentiation. PET CT showed intense uptake in osseous lesions at the scapula, vertebral bodies, iliac bones, sacrum, and femoral head. A sacrum biopsy confirmed metastatic spindle cell sarcoma. The patient underwent chemotherapy and radiotherapy. A follow-up CT scan several months later revealed new liver and lung metastases, prompting molecular analysis of the sacrum specimen.
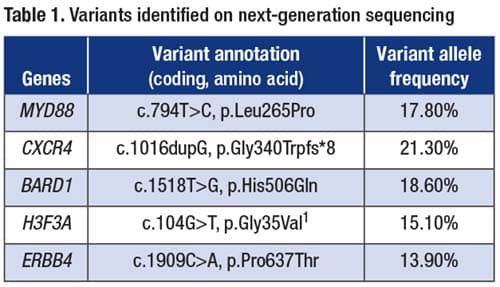
The patient’s sacrum biopsy was analyzed using the PGDx Elio assay (Personal Genome Diagnostics, Baltimore), which targets more than 500 genes using a hybrid-capture-based next-generation sequencing system. NGS showed sequence variants in the following genes (codon, variant allele frequency): MYD88 (p.L265P, 17.8 percent; Fig. 1A), CXCR4 (p.G340Wfs*8, 21.3 percent; Fig. 1B), BARD1 (p.H506Q, 18.6 percent), H3F3A (p.G35V, 15.1 percent), and ERBB4 (p.P637T, 13.9 percent) (Table 1). The tumor mutational burden was low (4.6 mutations/Mb).
The MYD88 p.L265P variant is a recurrent driver mutation in both Waldenström’s macroglobulinemia (WM) and IgM monoclonal gammopathy of undetermined significance (IgM MGUS). WM is defined by an IgM monoclonal component in the blood associated with elevated serum viscosity, in the setting of lymphoplasmacytic lymphoma (LPL). The MYD88 mutation is detected in more than 90 percent of patients with LPL/WM and in up to 80 percent of those with IgM MGUS.1-3 It may also be present in a subset of other hematologic malignancies, such as diffuse large B-cell lymphoma and chronic lymphocytic leukemia.3 Additionally, nonsense and frameshift mutations in the C-terminus of CXCR4 are observed in approximately 40 percent of LPL/WM cases, often occurring alongside MYD88 mutations.4 CXCR4 mutations have also been observed in patients with IgM MGUS.5 The H3F3A p.G35V variant, which is equivalent to p.G34V depending on which initial methionine is used for numbering, is thought to likely be related to the sarcoma given it has been reported in mesenchymal tumors, specifically a subset of giant cell tumors of bone, and other variants at the H3F3A p.G34 locus have been described in osteosarcoma.6-8 In addition, H3F3A p.G35V has not been reported in lymphoproliferative disorders. BARD1 p.H506Q and ERBB4 p.P637T are considered variants of uncertain significance, without previous reports in the literature.

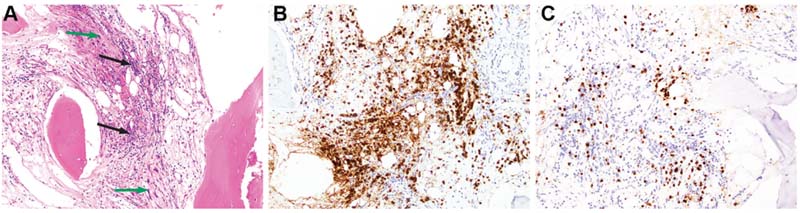
Following the molecular results, re-review of the sacrum biopsy revealed, in addition to the metastatic sarcoma, patchy infiltrates of small lymphocytes with slightly irregular nuclei, mature chromatin, and occasional intermixed plasma cells, present within the bone marrow (Fig. 2A). Subsequent immunohistochemistry showed that most of the lymphocytes were CD20+ PAX5+ B cells (Fig. 2B), which were CD5 negative. CD3 highlighted fewer interspersed small T cells (Fig. 2C). MUM1 was positive in a few intermixed plasma cells, which appeared polytypic for kappa and lambda light chains by chromogenic in situ hybridization; only rare plasma cells were IgM positive. Overall, the CD20+ B-cell infiltrates occupied 10 to 15 percent of the intertrabecular marrow space.
This B-cell-rich infiltrate raised concern for a concurrent small B-cell lymphoma, specifically LPL. A conclusive lymphoma diagnosis on the biopsy was precluded by the limited lymphoid infiltrate and lack of definitive light chain clonality in the plasma cell component. Further evaluation showed a trace band of monoclonal IgM kappa light chain on serum protein electrophoresis (SPEP), and increased serum free kappa light chain at 35.7 mg/dL (ref: 3.3–19.4) as well as elevated serum kappa/lambda free light chain ratio at 2.08 (ref: 0.3–1.7). Peripheral blood flow cytometry did not detect clonal B cells, and the complete blood count was normal aside from a slight increase in neutrophils and a mild decrease in lymphocytes. Taken together, the histological and laboratory findings in the setting of the molecular results strongly suggested a B-cell lymphoproliferative disorder. A bone marrow biopsy was recommended for further evaluation, which the patient elected to defer during the treatment of her metastatic sarcoma. Following additional chemotherapy and radiotherapy for sarcoma, the patient experienced disease progression and was subsequently placed on home hospice care.
IgM MGUS is defined by the presence of a circulating monoclonal IgM at a concentration of less than 3 g/dL and, if assessed, a bone marrow containing less than 10 percent monoclonal plasma cells in asymptomatic patients. Most of the bone marrow B cells and plasma cells are polyclonal in IgM MGUS.9 In this case, a trace band of monoclonal IgM kappa light chain was identified on SPEP without definitive histological evidence of clonality on the sacrum biopsy, despite a patchy B-cell-rich lymphoid infiltrate. These findings, along with the MYD88 p.L265P and CXCR4 truncating alterations, may be indicative of IgM MGUS. Moreover, recent studies have shown that MYD88 p.L265P may be present in B-cell precursors and normal nonclonal B cells in patients with LPL/WM.9,10 Given the limited sampling from the sacrum biopsy, an overt LPL cannot be entirely excluded without a more comprehensive bone marrow biopsy for better characterization.
In conclusion, we report a case of metastatic sarcoma with incidental detection of molecular variants associated with B-cell lymphomas. These findings, cross-referenced with histological and laboratory results, suggested the presence of an underlying B-cell lymphoproliferative disorder, most likely IgM MGUS. Subtle findings of neoplastic lymphoid infiltrates may be difficult to detect based on morphology alone. However, molecular findings, sometimes incidental, may enable early detection and facilitate close monitoring of disease progression, emphasizing the importance of clear and timely communication by pathologists with patient-facing clinical colleagues.
 Landgren O, Staudt L. MYD88 L265P somatic mutation in IgM MGUS. N Engl J Med. 2012;367(23):2255–2257.
Landgren O, Staudt L. MYD88 L265P somatic mutation in IgM MGUS. N Engl J Med. 2012;367(23):2255–2257.- Jiménez C, Sebastián E, Chillón MC, et al. MYD88 L265P is a marker highly characteristic of, but not restricted to, Waldenström’s macroglobulinemia. Leukemia. 2013;27:1722–1728.
- Alcoceba M, García-Álvarez M, Medina A, et al. MYD88 mutations: transforming the landscape of IgM monoclonal gammopathies. Int J Mol Sci. 2022;23(10):5570.
- Kaiser LM, Hunter ZR, Treon SP, Buske C. CXCR4 in Waldenström’s macroglobulinema: chances and challenges. Leukemia. 2021;35:333–345.
- Moreno DF, López-Guerra M, Paz S, et al. Prognostic impact of MYD88 and CXCR4 mutations assessed by droplet digital polymerase chain reaction in IgM monoclonal gammopathy of undetermined significance and smouldering Waldenström macroglobulinaemia. Br J Haematol. 2023;200(2):187–196.
- Leske H, Dalgleish R, Lazar AJ, Reifenberger G, Cree IA. A common classification framework for histone sequence alterations in tumours: an expert consensus proposal. J Pathol. 2021;254(2):109–120.
- Yamamoto H, Iwasaki T, Yamada Y, et al. Diagnostic utility of histone H3.3 G34W, G34R, and G34V mutant-specific antibodies for giant cell tumors of bone. Hum Pathol. 2018;73:41–50.
- Koelsche C, Schrimpf D, Tharun L, et al. Histone 3.3 hotspot mutations in conventional osteosarcomas: a comprehensive clinical and molecular characterization of six H3F3A mutated cases. Clin Sarcoma Res. 2017;7:9.
- Rodriguez S, Celay J, Goicoechea I, et al. Preneoplastic somatic mutations including MYD88L265P in lymphoplasmacytic lymphoma. Sci Adv. 2022;8(3):eabl4644.
- Kaushal A, Nooka AK, Carr AR, et al. Aberrant extrafollicular B cells, immune dysfunction, myeloid inflammation, and MyD88-mutant progenitors precede Waldenstrom macroglobulinemia. Blood Cancer Discov. 2021;2(6):600–615.
Dr. Zhao is a clinical fellow in pathology, Dr. Hasserjian is director of the hematopathology fellowship and professor of pathology, Dr. Chebib is director of the cytopathology laboratory and fine-needle aspiration biopsy service and associate professor of pathology, and Dr. Fisch is assistant pathologist and assistant professor of pathology—all in the Department of Pathology, Massachusetts General Hospital and Harvard Medical School.

