Summary
The 2024 revision of the 2017 McDonald criteria for diagnosing multiple sclerosis allows the kappa-free light chain index to replace oligoclonal band detection in cerebrospinal fluid analysis. This change addresses the limitations of oligoclonal banding and the inadequate sensitivity of the CSF IgG index. The revised criteria aim to improve MS diagnosis and facilitate earlier access to therapy.
Amy Carpenter
February 2026—The 2017 McDonald criteria for diagnosing multiple sclerosis were revised in 2024, and the new criteria were published last fall. For cerebrospinal fluid analysis in the context of an MS workup, the kappa-free light chain index can now replace oligoclonal band detection, which is the gold standard but labor-intensive and rater-dependent (Montalban X, et al. Lancet Neurol. 2025;24[10]:850–865).
With oligoclonal banding’s drawbacks and the inadequate sensitivity of the CSF IgG index (another test for MS), “The field is ripe for new biomarkers of MS to address some of these challenges,” Maria Alice V. Willrich, PhD, D(ABCC), said in an ADLM session last summer. She is co-director of the protein and antibody immunology laboratories and the clinical mass spectrometry laboratory at Mayo Clinic in Rochester, Minn.
Dr. Willrich reported on the traditional biomarkers in the ADLM session on lab biomarkers for MS. Co-presenters Harald Hegen, MD, PhD, and Florian Deisenhammer, MD, both of the University of Innsbruck, Austria, delved into the data on κ-free light chains and how to offer testing in alignment with the new criteria (to be reported in an upcoming issue).
MS is the most common immune-mediated inflammatory demyelinating disease of the central nervous system, and its cause remains unknown. “We still are to find the antigen that causes multiple sclerosis, if there is only one that causes it,” Dr. Willrich said. Variable clinical and pathologic features are present: inflammation, demyelination, and axonal degeneration. Its suggestive features include presentations of relapses and remissions and onset between ages 15 and 50.
Among the environmental risk factors are Epstein-Barr virus infections, geographic latitude and birthplace, sunlight exposure, and vitamin D concentration.
“There is genetic susceptibility, with over 200 variants identified,” Dr. Willrich said, “but they have only modest effects on triggering the disease.” The female to male ratio is about four to one, and the incidence of MS in women has increased over the years, she said, with the ratio approximately 10 years ago at 2.7 to one.
The blood-brain barrier limits the exchange of many compounds. In laboratory testing for demyelinating diseases, “We can run analyses of total proteins and specific proteins in CSF.
When the blood-CSF barrier is damaged or impaired, there could be a flow of blood proteins to the CSF, causing false elevations of CSF results in some cases.” By looking at indexes that put in context both CSF and serum analyses, she continued, “we can correct any elevations coming from blood and evaluate if the CSF protein content is abnormal, offering evidence of intrathecal synthesis of proteins in the CSF.”
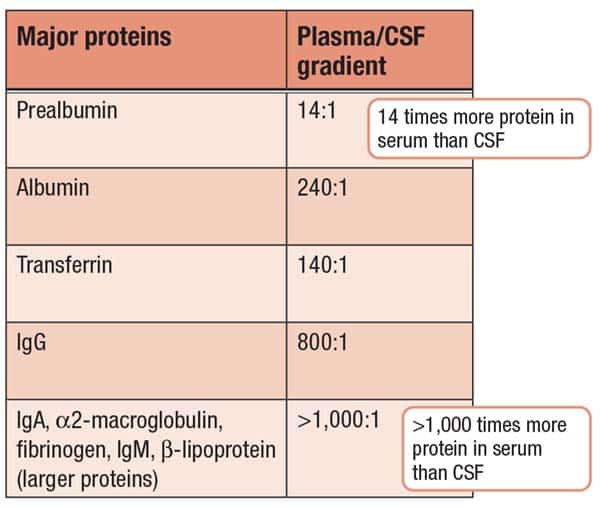
Protein concentrations are much lower in CSF than in serum (Fig. 1). “The smaller proteins are more abundant in CSF than in serum, and the relative distribution of CSF proteins is not identical to blood’s, especially when compared with larger-molecular-weight proteins like α2-macroglobulin, fibrinogen, IgA, IgM, and β-lipoprotein,” Dr. Willrich said, “where we have more than 1,000 times more of those larger proteins in serum than CSF.”
Several assays use paired CSF and serum samples for demyelinating disease, and the majority require the samples to be collected at the same time. The length of the period allowed between collection times has to be validated, Dr. Willrich said, whether it is within 24 or 48 hours or longer. “Sometimes it’s a week or 10 days, depending on the local validation the labs have used.”

