Amy Carpenter
June 2025—At Corewell Health William Beaumont University Hospital in Royal Oak, Mich., the PSA test volume for men ages 40 to 49 is significant—about 150 samples per month, or 10 percent of the laboratory’s total PSA testing volume. But with no PSA assay approved for men under age 50, the laboratory set out to validate its Abbott Architect assay for this age group as a laboratory-developed test.
Qian (Katie) Sun, PhD, D(ABCC), technical director of automated chemistry and urinalysis, explained how it was done in an ADLM session last year on designing validation protocols for off-label use of tumor markers in diverse patient populations. She also spoke about the use of hCG immunoassays as a tumor marker in gestational trophoblastic disease and select germ cell tumors. (Co-speaker Jonathan Genzen, MD, PhD, MBA, of ARUP Laboratories, presented on body fluids. See story here.)
There is no consensus in the clinical recommendations for prostate cancer screening. The U.S. Preventive Services Task Force says screening is the individual’s choice and the starting age is 55. For persons at average risk, the American Cancer Society recommends screening starting at age 50 and the National Comprehensive Cancer Network recommends screening starting at 45. The ACS and NCCN recommend starting even earlier for those at higher risk.
Various cutoffs have been proposed. The commonly cited cutoff is 4.0 ng/mL. A cutoff of 3.0 ng/mL was taken from the European Randomized Study of Screening for Prostate Cancer. Recent studies proposed age-specific cutoffs: 2.5 ng/mL for people in their 40s, 3.5 ng/mL for those in their 50s, 4.5 ng/mL for those in their 60s, and 6.5 ng/mL for those in their 70s.

For the analytical validation at Corewell Health, the laboratory team used commercially available material for linearity and quality control materials for precision, and for accuracy they compared their results with Mayo Clinic’s Roche Cobas PSA method using 24 samples from men ages 40 to 49. Mayo Clinic had verified the reference range for this age group.
“All the comparison results were within the total allowable error of 20 percent or 0.2 nanogram per mL,” said Dr. Sun, who is also an assistant professor, Department of Pathology and Laboratory Medicine, Oakland University William Beaumont School of Medicine.
For analytical specificity validation, Dr. Sun and colleagues decided the analytical specificity study performed by Abbott was sufficient because there’s no evidence, Dr. Sun said, that the matrix of serum samples from men between 40 and 49 differs significantly from that of men 50 and older.
For analytical sensitivity, they performed their own study. The assay package insert lists the assay limit of quantification as 0.1 ng/mL. To evaluate the limit of quantification, they prepared five samples using patient specimens with a target concentration of 0.01, 0.04, 0.07, 0.11, and 0.14 ng/mL.
They ran the five specimens in five replicates over five days and then calculated the standard deviation and coefficient of variation based on their results. All five samples had a CV of less than 20 percent and thus the analytical sensitivity was found to be acceptable.
To evaluate clinical sensitivity and specificity, they did a four-year chart review of patients at Corewell Health East from 2020 to 2023. Patients were divided into two groups: those with a prostate cancer diagnosis based on the diagnosis code or biopsy result and the control group—those with a PSA order from their physicians and no diagnosis code for prostate cancer.
Patients were divided by age: 40 to 49 years and, as a reference population, age 50 and older. “That’s because we need criteria to decide whether sensitivity and specificity parameters for men between 40 and 49 would be acceptable or not,” Dr. Sun said. Of men ages 40 to 49, 33 had a prostate cancer diagnosis; 1,226 patients were in the control group. In the reference population of men 50 and older, 233 patients had a prostate cancer diagnosis; 10,848 patients did not.
Men ages 40 to 49 who were diagnosed with prostate cancer had an average PSA of 4.61 ng/mL (2.41–406.40 ng/mL). In the control group the average PSA in this age group was 0.64 ng/mL (<0.10–8.96 ng/mL). Men 50 and older who were diagnosed with prostate cancer had an average PSA of 6.23 ng/mL (0.89–>1000.0 ng/mL). In the control group, average PSA for this age group was 0.90 ng/mL (<0.10–81.43 ng/mL). PSA results were extracted from within six months prior to biopsy.
“In my health care system, when I did the chart review, I noticed that the youngest patient who was diagnosed with prostate cancer was only 36 years old. And there’s one patient who passed away from metastatic prostate cancer at age 41,” Dr. Sun said. “So in my health care system, there’s truly a clinical need for offering this test to younger men.”
To calculate clinical sensitivity and specificity, Dr. Sun and colleagues used a cutoff of 2.5 ng/mL for men ages 40 to 49 (based on previous publications and ROC analysis of their own data) and 4 ng/mL for men 50 and older (based on package insert, confirmed with ROC analysis).
For men 40 to 49, clinical sensitivity and specificity were 97 percent, which was higher than for men 50 and older (clinical sensitivity, 83 percent; clinical specificity, 94 percent). “That’s why we decided our results are acceptable,” Dr. Sun said.
Can her laboratory’s findings be applied to other platforms?
In a study published in 2024, van Rossum, et al., used external quality assessment materials based on patient serum to evaluate whether PSA results were comparable among the Abbott Alinity, Beckman Coulter DxI, Roche Cobas, and Siemens Atellica (van Rossum HH, et al. Clin Chem. 2024;70[4]:669–679). They compared each method with a consensus mean. “Overall, the results are pretty comparable,” Dr. Sun said. The largest positive bias was observed with the Roche method (+10.0 percent); the largest negative bias was observed with the Siemens Healthineers method (−7.1 percent). For Abbott, the difference with the consensus mean was −0.3 percent. For the Beckman Coulter assay, it was −2.7 percent. All results were within the desirable bias criterion of ±10.6 percent.
Dr. Sun and colleagues performed a clinical validation study in collaboration with three other institutions that were using the Beckman Coulter DxI, Roche Cobas, and Siemens Atellica PSA assays. Each institution pulled its own patient data using the same criteria the Corewell Health laboratory used for its validation study, she said. The Corewell laboratory provided the Abbott Architect data. Cutoffs were 2.5, 3.0, 3.5, and 4.0 ng/mL for ages 40 to 49 and 4.0 ng/mL for the reference population.
They were unable to establish an optimal cutoff for the two institutions using the Beckman Coulter and Roche PSA assays because the clinical sensitivity results for men ages 40 to 49 using various cutoffs were lower compared with the reference group result, Dr. Sun said. For the institution using Siemens Atellica, the optimal cutoff was determined to be 3 ng/mL; for Abbott Architect, it was 2.5 ng/mL.
For Beckman Coulter DxI, the clinical sensitivity ranged from 64.71 (2.5 ng/mL cutoff) to 50.0 (4.0 ng/mL cutoff) percent in men 40 to 49 and was 88.24 percent in men 50 and older. For Roche Cobas, sensitivity was 76.92 percent at all four cutoff levels in men 40 to 49; it was 84.50 percent in men 50 and older. For Abbott Architect, sensitivity was between 97.06 (2.5 ng/mL) and 55.88 (4.0 ng/mL) percent in men 40 to 49 and 83.19 percent in men 50 and older. For Siemens Healthineers Atellica, it was 75.68 (2.5 ng/mL) to 62.16 (4.0 ng/mL) percent in men 40 to 49 and 75.62 percent in men 50 and older.
Clinical specificity for Beckman Coulter DxI ranged from 96.80 (2.5 ng/mL) to 98.70 (4.0 ng/mL) percent in men 40 to 49; it was 91.09 percent in men 50 and older. For Roche Cobas, clinical specificity ranged from 93.35 (2.5 ng/mL) to 98.79 (4.0 ng/mL) percent in men 40 to 49; in men 50 and older it was 85.39 percent. Specificity for Abbott Architect was between 97.46 (2.5 ng/mL) and 99.26 (4.0 ng/mL) percent in men 40 to 49; it was 93.56 percent in men 50 and older. For Siemens Healthineers Atellica, clinical specificity was between 97.24 (2.5 ng/mL) and 99.19 (4.0 ng/mL) percent in men 40 to 49 and 93.40 percent in men 50 and older.
“Overall,” Dr. Sun said, “there’s a lot of variation in the results among different institutions and different methods.” One reason might be that the data are preliminary. “We do not have enough cancer patients between 40 and 49, so to have a more definitive answer, we may need to expand our study.”
The varying clinical practices among institutions, particularly since there is no consensus in the clinical recommendations for PSA screening in younger men, may also explain the variation in results, she said. “There also might be slight bias among different methods.”
Turning next to hCG as a tumor marker, Dr. Sun noted that NCCN clinical practice guidelines recommend its use as a tumor marker in testicular cancer and gestational trophoblastic disease for diagnosis, prognosis, and monitoring.
“And there is potential use of hCG as a tumor marker in select patients with ovarian cancer for the purpose of diagnosis and/or monitoring of residual disease and recurrence,” she said. “However, currently there is no hCG test available that’s FDA approved for oncology. All the commercially available hCG assays are approved only for use in pregnancy.”
One of the concerns about using the same hCG immunoassay in pregnancy and in oncology is that several hCG variants have been documented in cancers, Dr. Sun said. “In pregnancy diagnosis and monitoring, we’re looking for the intact hCG, which is composed of the alpha-subunit and beta-subunit. We’re also looking for total hCG, which is composed of the intact hCG as well as a free beta-subunit.”
In cancers, the hCG variants that have been documented include the β-subunit (hCGβ) and hyperglycosylated hCG (h-hCG), she said. “Most serum hCG immunoassays are designed to pick up as many hCG isoforms as possible. So most commercially available assays can pick up the majority of those variants.” They recognize the intact hCG, nicked hCG (hCGn), hyperglycosylated forms, beta-core fragments, and free beta-subunits.
Pretorius, et al., studied the comparability of total hCG tumor marker assays (Pretorius CJ, et al. Clin Chem Lab Med. 2020;58[3]:438–444). They measured total hCG (hCGt) in 390 samples from 137 females and 253 males with a tumor marker request and in 208 pregnancy controls, using the Beckman Coulter Access Total βhCG, Abbott Architect Total-βhCG, Roche Cobas hCG+β, and Siemens Healthineers Immulite HCG. (The Roche Cobas hCG+β immunoassay received clearance for use as a tumor marker in Australia, where the study was done, by the Australian Therapeutics Goods Administration.)
It has been reported that the molar ratio of the beta-subunit to hCGt increases with malignancy, as Dr. Sun noted. “However, cross-reactivity of the beta-subunit is different among the four methods.” All four immunoassays had similar cross-reactivity with an intact hCG. “However, one of them, the Beckman Coulter Access assay, can pick up twice the amount of free beta-subunit compared with the other three immunoassays,” she said. The Beckman Coulter Access Total βhCG had 220/142 percent hCG fragment recognition of free beta-subunit compared with the other assays: Abbott Architect Total βhCG, 97/87 percent; Roche Cobas hCG+β, 108/102 percent; Siemens Healthineers Immulite HCG, 112/111 percent.
The authors did not expect the four immunoassays to be comparable, Dr. Sun said. They write, “We hypothesised that regression slopes would be altered and that outliers would be more common with tumour marker than with pregnancy samples if the detection of malignancy associated hCG molecular forms differed amongst assays. Further such systematic differences would be obvious and large enough to change clinical management decisions.”
Said Dr. Sun, “The regression slope was largely unaltered.” The authors reported that the outlier rates were similar for male and female tumor marker and the pregnancy groups: 1.6 percent, 2.2 percent, and 2.9 percent. The outliers were randomly distributed among the methods, they write, “and we were confident that they would not adversely influence clinical decisions.”
“So the authors concluded,” Dr. Sun said, “that the hCG results were clinically equivalent with no systematic difference among the four assays.” Among the limitations is that they did not identify the type of malignancy in the vast majority of the tumor marker group, Dr. Sun said. And the other is that one of the assays used in their report is known to overestimate beta-subunit by about twofold. However, this assay was not responsible for any of the outliers identified, which David Grenache, PhD, D(ABCC), described in an accompanying editorial as “curious and warranting of further investigation” (Grenache DG. Clin Chem Lab Med. 2020;58[3]:323–325).
To overcome some of these limitations, she said, the authors of a study published in 2023 assessed the utility of five quantitative hCG immunoassays as tumor markers in trophoblastic and non-trophoblastic disease (Franks CE, et al. Clin Chem. 2023;69[6]:606–614). Remnant specimens were obtained from 150 patients—18 patients had gestational trophoblastic disease, 42 had germ cell tumors, and 90 had other malignancies. Five analyzer platforms were used for split specimen analysis of hCG: Abbott Architect Total, Roche Cobas STAT, Roche Cobas Total, Siemens Dimension Vista Total, and Beckman Access Total.
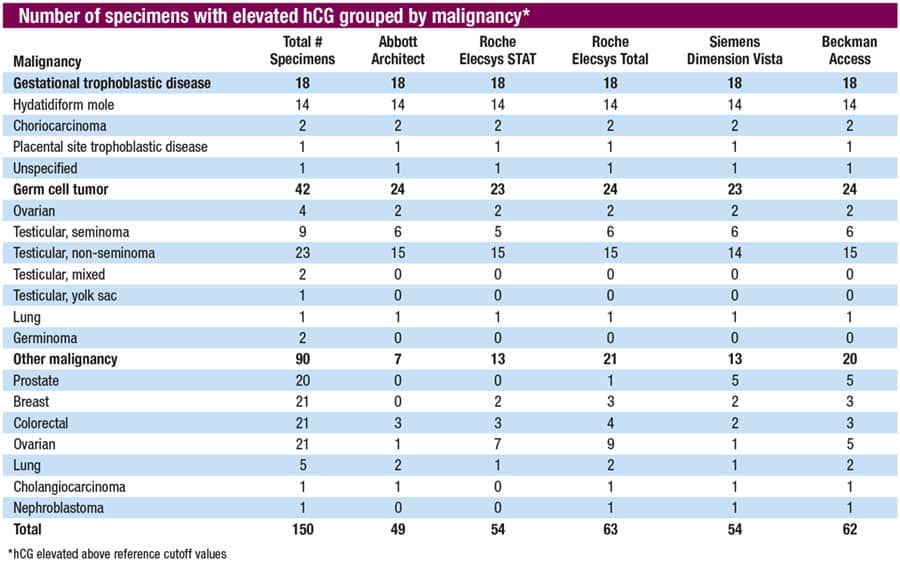
“All five immunoassays can pick up all of the 18 specimens from patients with gestational trophoblastic disease,” Dr. Sun said. “They can pick up a similar number of samples from patients with germ cell tumors”—23 to 24 of the 42. “The variation lies with patients with other malignancies” (prostate, breast, colorectal, ovarian, lung, cholangiocarcinoma, and nephroblastoma). The five assays picked up between seven and 21 of the 90 specimens. (See “Number of specimens with elevated hCG grouped by malignancy.”)
The authors reported that the frequency of elevated hCG concentrations (above reference cutoffs) was highest in gestational trophoblastic disease (100 percent), followed by germ cell tumors (55 to 57 percent) and other malignancies (eight to 23 percent). “Overall,” they write, “the Roche cobas Total detected elevated hCG in the greatest number of specimens (63/150),” followed closely by the Beckman Access (62/150). Detection of elevated hCG in trophoblastic disease was nearly equivalent among all immunoassays (range: 41 to 42 of 60).
Dr. Sun summed up: “All evaluated hCG immunoassays are adequate for use as a tumor marker in gestational trophoblastic disease and select germ cell tumors. And when used for tumor monitoring, it’s important that providers use the same assay or adhere to the same platform to ensure the results are consistent.”
Amy Carpenter is CAP TODAY senior editor.

