Focus on cytology
Nicholas Stanzione, MD
Christopher O’Conor, MD, PhD
Varsha Manucha, MD, MBBS
August 2025—Human papillomavirus testing has become the standard of care in head and neck squamous cell carcinoma (HNSCC) because of the unique clinical features, staging, and treatment options for HPV-associated HNSCC. HPV-associated oropharyngeal squamous cell carcinoma (OPSCC) exhibits a favorable prognosis and improved response to chemoradiation compared with conventional HNSCC and non-HPV-associated forms,1 and reporting the HPV status is frequently part of clinical trial enrollments. Many patients with OPSCC present with enlarged level II or III cervical lymph nodes and, as a result, cervical lymph node fine-needle aspiration is often the first, and sometimes only, tissue obtained for diagnostic testing. With a growing menu of options available to test for HPV status, including polymerase chain reaction, DNA in situ hybridization, mRNA ISH, liquid-based HPV assays, and p16 immunohistochemistry, it is not always clear when and which HPV test to use, especially when the diagnosis is made on a cytology specimen.
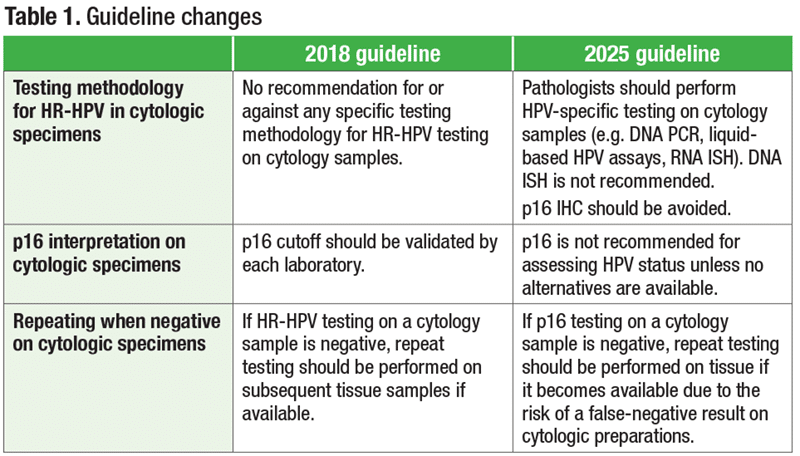
The CAP published in 2018 its first clinical practice guideline for the testing of HPV in HNSCC and its application, interpretation, and reporting, based on a systematic literature review.2 The updated guideline, published in March in Archives of Pathology & Laboratory Medicine, integrates the most recent advancements and understanding of HPV testing. Summarized in Table 1 are the changes made to the guideline on HPV testing, specifically on cytologic specimens.3
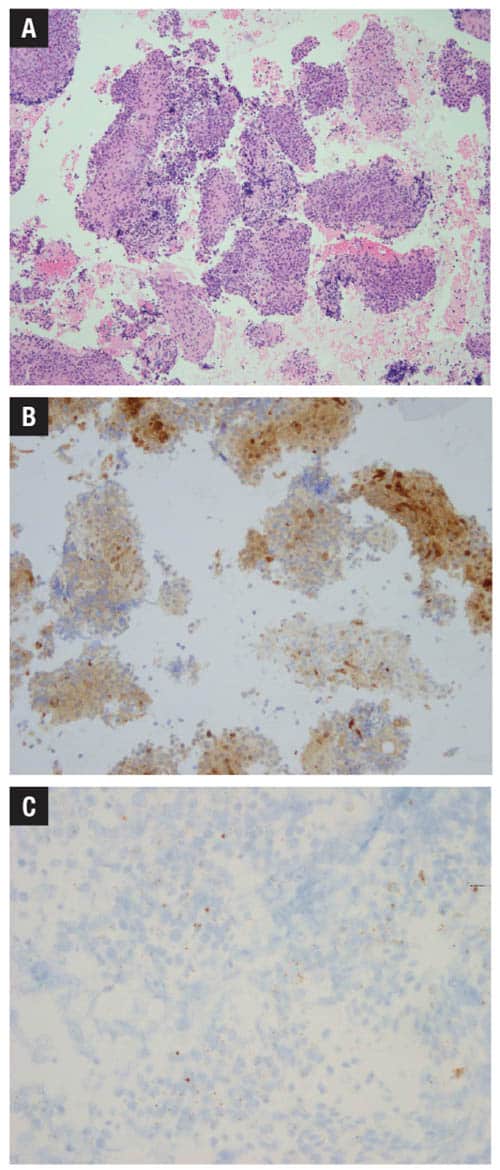
The 2025 guideline strongly recommends performance of HR-HPV testing on head and neck FNAs of nodal SCC specimens from all patients with either unknown primary or oropharyngeal or sinonasal primary. However, in contrast to the 2018 guideline, the current guideline recommends against use of p16 IHC as a surrogate marker for assessing HPV status in cytology specimens. It has long been known that the performance of p16 on cytologic samples can vary widely (Fig. 1B), likely due to differences in cold ischemic time, fixation techniques, cell block tumor cellularity, and cell preservation, resulting in increased risk of false-negative results and lack of a well-defined cutoff for positivity.4 While the 2018 guideline encouraged each laboratory to validate its performance of p16, the authors updated the recommendation to advise avoiding altogether p16 IHC on cytology specimens of neck FNAs, except in select circumstances. Notably, a recommendation from the 2018 guideline on repeating HR-HPV testing has been updated to say specifically that only for p16 should testing be repeated on subsequent tissue samples when available because of this assay’s well-documented risk of false-negative results.
There are a plethora of HPV-specific testing methodologies, particularly when it comes to cytology. HPV RNA ISH is a great option that has been gaining adoption due to its outstanding test performance, even on cell block material, and its compatibility with automated stainers. DNA PCR is adequate only in conjunction with p16 IHC-positive tumors and is not recommended as a confirmatory test. One of the most exciting developments in this area may be the emerging use of liquid-based assays to determine HPV status, such as validating cervical liquid-based HPV assays on cytologic supernatants, and the detection of circulating tumor HPV DNA from serum.5
In summary, the updated guideline for HPV testing in HNSCC in the context of cytologic samples is to perform HPV-specific testing in lieu of p16 IHC due to the inconsistencies in p16 performance on cell block materials. Which methodology your laboratory seeks to adopt will depend on its individual needs, workflow, and logistics. HR-HPV ISH testing on cell block material is one of the most common testing approaches, although interest in liquid-based assays is growing.
- Marur S, D’Souza G, Westra WH, Forastiere AA. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 2010;11(8):781–789.
- Lewis JS Jr, Beadle B, Bishop JA, et al. Human papillomavirus testing in head and neck carcinomas: guideline from the College of American Pathologists. Arch Pathol Lab Med. 2018;142(5):559–597.
- Lewis JS Jr, Beadle B, Bishop JA, et al. Human papillomavirus testing in head and neck carcinomas: guideline update. Arch Pathol Lab Med. 2025;149(6):e115–e150.
- Wong KS, Krane JF, Jo VY. Heterogeneity of p16 immunohistochemistry and increased sensitivity of RNA in situ hybridization in cytology specimens of HPV-related head and neck squamous cell carcinoma. Cancer Cytopathol. 2019;127(10):632–642.
- Siravegna G, O’Boyle CJ, Varmeh S, et al. Cell-free HPV DNA provides an accurate and rapid diagnosis of HPV-associated head and neck cancer. Clin Cancer Res. 2022;28(4):719–727.
Dr. Stanzione is assistant professor, Department of Pathology and Laboratory Medicine, University of California, Los Angeles. Dr. O’Conor is assistant professor, Department of Pathology, Microbiology, and Immunology, Vanderbilt University Medical Center. Dr. Manucha is professor, Department of Pathology, University of Mississippi Medical Center, Jackson. Drs. Stanzione and O’Conor are members and Dr. Manucha is vice chair of the CAP Cytopathology Committee.

