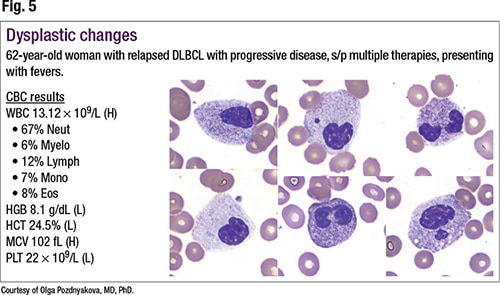
Dr. Pozdnyakova noted in Fig. 5 (two left images in top row and one image on bottom left) monolobated nuclei, usually called Stodtmeister cells. She pointed also to bilobed nuclei (lower middle and upper far right), referred to as pseudo Pelger-Huët cells. “Some cells also show cytoplasmic hypergranulation. These morphologic findings are concerning for neoplastic rather than reactive changes,” she said. A molecular analysis identified ASXL1 and DNMT3A mutations consistent with treatment-related MDS. “And in this case, it is possible that hypergranulation and vacuolization are a sign of dysplasia rather than infection.”
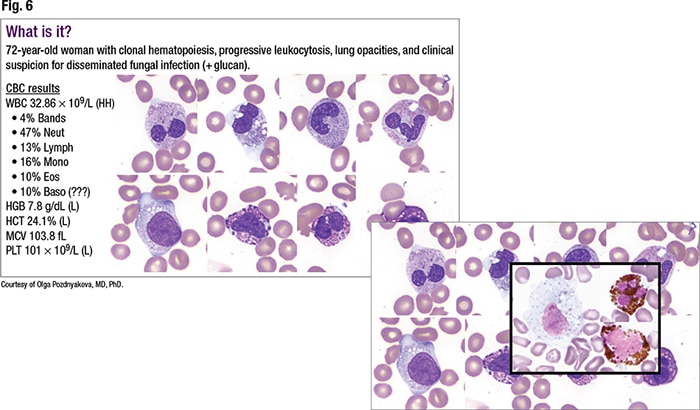
 Dysplastic changes in neutrophils can present in many forms, Dr. Pozdnyakova cautioned, presenting what she later described to CAP TODAY as the most recent difficult case (Fig. 6). The 72-year-old female patient had a known history of clonal hematopoiesis and presented with progressive leukocytosis of greater than 30,000. The WBC count showed neutrophilia and monocytosis. Ten percent of the cells were called basophils.
Dysplastic changes in neutrophils can present in many forms, Dr. Pozdnyakova cautioned, presenting what she later described to CAP TODAY as the most recent difficult case (Fig. 6). The 72-year-old female patient had a known history of clonal hematopoiesis and presented with progressive leukocytosis of greater than 30,000. The WBC count showed neutrophilia and monocytosis. Ten percent of the cells were called basophils.
“Upon review it was clear that the granules in those cells that were called basophils are much larger than in typical basophils and are reminiscent of encapsulated intracellular yeast, particularly Histoplasma, which corroborated the clinical impression of disseminated fungal infection,” Dr. Pozdnyakova said. “However, Histoplasma urine antigen came back negative.”
Dr. Pozdnyakova then requested a myeloperoxidase stain, which accentuated forms that turned out to be dysplastic primary granules in the setting of progression of the MDS/MPN overlap syndrome. And once a peripheral blood smear was reviewed, it demonstrated other more typical dysplastic changes, she said, noting a pseudo-Pelger-Huët nucleus.
 A session attendee noted that few laboratories offer myeloperoxidase. Dr. Pozdnyakova acknowledged that the Dana-Farber Cancer Institute hematology laboratory is likely “the last man standing” in offering the myeloperoxidase cytochemical stain. They use nonspecific esterase, MPO, PAS, and specific esterase for all new cases of acute leukemia. “It’s a very rapid test, and it allows us to put a case into an acute myeloid leukemia with monocytic differentiation category, or just typical AML if it’s MPO positive, very fast,” she said—“or into acute lymphoblastic leukemia if it’s PAS positive.”
A session attendee noted that few laboratories offer myeloperoxidase. Dr. Pozdnyakova acknowledged that the Dana-Farber Cancer Institute hematology laboratory is likely “the last man standing” in offering the myeloperoxidase cytochemical stain. They use nonspecific esterase, MPO, PAS, and specific esterase for all new cases of acute leukemia. “It’s a very rapid test, and it allows us to put a case into an acute myeloid leukemia with monocytic differentiation category, or just typical AML if it’s MPO positive, very fast,” she said—“or into acute lymphoblastic leukemia if it’s PAS positive.”
Administration of growth factors is another important cause of neutrophilia, presenting with the morphologic finding of a significant degree of left shift that frequently includes blasts, Dr. Pozdnyakova noted. However, occasionally this clinical information isn’t readily accessible. In the case of a 56-year-old male patient with Burkitt lymphoma who was taking Granix for neutropenia, the blast count was two percent (Fig. 7). She said she has come across cases with blast counts reaching 10 percent. The additional morphologic features to look for in such cases are prominent toxic changes and nuclear-cytoplasmic dyssynchrony. “For example, you can see retention of primary granules with very mature nucleus,” she said. In looking at the promyelocytes, “sometimes you can see prominent perinuclear hoffs. Very often neutrophilia is accompanied by thrombocytosis,” but it wasn’t seen in this case.

