In the ICC, because there is 16 percent blasts—more than 10 percent—the case would qualify as MDS/AML with mutated TP53. Patients with more than five percent blasts have a poor prognosis and should be considered for allogeneic stem cell transplant if possible, she said.
The WHO classification does not recognize myeloid neoplasms with TP53 mutation as a distinct category. “Most AML cases are classified as AML with myelodysplasia-related cytogenetic abnormalities by virtue of the complex karyotype they are typically associated with,” Dr. Loghavi said. ICC recognizes MDS/AML with mutated TP53, “and you don’t need a biallelic or multi-hit status. Any TP53 mutation with a VAF of more than 10 percent will qualify for this category.” AML with mutated TP53 is identified by more than 20 percent blasts and any TP53 mutation with a VAF of more than 10 percent.
“These patients are going to have a very short window between when you can get them into remission and into transplant,” she said.
She cited one of many publications that showed “when you have a myeloid neoplasm with a TP53 mutation with an increase in blasts—more than five, 10 percent—regardless of the allelic status and the blast count, the patients are going to do equally as poorly” (Grob T, et al. Blood. 2022;139[15]:2347–2354).
Dr. Loghavi presented a second case, that of a 71-year-old man who underwent a workup for pancytopenia (Loghavi S, et al. Mod Pathol. 2024;37[2]:100397).
The patient was neutropenic (WBC: 1.5 × 109/L) and had a low leukocyte count and low hemoglobin (8.9 g/dL). He was severely thrombocytopenic (Plt: 6 × 109/L). His MCV was 96 fL.
In the core biopsy, distinct, large cells with crisp, nuclear outlines and carrot-like nucleoli stuck to the membrane were a typical picture of erythroid blasts, Dr. Loghavi said. “All of the hematopoiesis is replaced with erythroid blasts, confirmed on the aspirate smear,” which showed intensely blue cells with round, punched-out nuclei and vacuolated cytoplasm. The CD71 stain confirmed erythroid differentiation. The p53 stain revealed an abnormal mutant pattern. “We had 82 percent erythroid cells, 42 percent of which were erythroid blasts.” (More on this case later.)
The majority of AML cases are classified by their genetic alteration, both in WHO and ICC. “When I’m on an acute leukemia service, if I see 10 AMLs, I’m able to genetically classify nine of them,” Dr. Loghavi said. “It’s rare to come across a case that’s not genetically classified and [have to] go by morphology.” But those rare cases exist, she added, and she suspects the genetic alteration is not being detected or is nondescript. In WHO-HEM5, several alterations are AML-defining, among them the NPM1 mutation.
What’s important, she said, is that the 20 percent minimum blast count requirement for AML has been removed in the WHO classification. An increase in blasts is required, “which is essentially five percent or higher.” The case qualifies as AML with one of several genetic abnormalities listed.
But the 20 percent blast count requirement remains for AML with CEBPA mutation (in WHO) or with BCR::ABL1 (in WHO and ICC). “You don’t want to overdiagnose CML in accelerated or blast phase as a de novo AML, so AML with BCR::ABL1 is restricted to de novo AML cases,” Dr. Loghavi said. “And for CEBPA, the WHO thought there was not sufficient evidence to say that cases with less than 20 percent blasts behave similarly to those with a higher blast count. But in routine practice when you have a pathogenic CEBPA mutation, the blast count is typically high, so that dilemma is not much of a dilemma.” The 20 percent requirement also remains for myelodysplasia-related AML (AML-MR).
If there is no defining genetic abnormality, differentiation is used.
It’s Dr. Loghavi’s view that it was a mistake to completely replace phenotype with genotype in the newer classifications. “Phenotype still speaks to the biology of disease,” she said, “and there’s evidence to support that.” Monocytic, erythroid, and megakaryocytic leukemias don’t respond as well to venetoclax, in part because they’re enriched in TP53 or RAS mutations that make these leukemias resistant to BCL-2 inhibitors. “The differentiation is important, and we should put that in our top diagnostic line. You can have an NPM1-mutated AML and call it that, but if it’s purely monoblastic you want to indicate that and say, ‘This is an NPM1-mutated AML with monoblastic differentiation.’”
The ICC is similar, she said, with the difference being that the AML genetic qualifiers still require 10 percent blasts, including for acute promyelocytic leukemia and core-binding factor AMLs. “In routine practice, it’s not much of a dilemma if you have an APL or a core-binding factor because the blast count is usually more than 10 percent,” Dr. Loghavi said, but a few cases may not meet the 10 percent blast count threshold. In addition, the ICC doesn’t use differentiation. “They call everything AML NOS.”
Back to the case of the 71-year-old patient with pure erythroid leukemia. In the 2016 WHO classification, there was such a category, which is an extreme erythroid differentiation in the blasts. Many publications support the association of pure erythroid leukemia with multi-hit or biallelic TP53 alterations, Dr. Loghavi said. This leukemia is usually seen with a TP53 mutation, a complex karyotype, and a high VAF. “It’s essentially defined by the presence of TP53 mutations,” she said.
The 71-year-old patient had extensive erythroid differentiation and a TP53 mutation. “In the WHO fifth category, even though this has TP53 mutation, the presence of erythroid differentiation supersedes AML with myelodysplasia-related changes,” Dr. Loghavi said. “You would call this an acute erythroid leukemia.”
In the ICC, by virtue of the presence of the TP53 mutation and alterations, all these cases are lumped into AML with mutated TP53. “Even if you want to call it AML with mutated TP53, it’s helpful to put the erythroid differentiation in the diagnostic top line,” she said.
Are TP53 mutations universally bad? The context in which they occur matters, Dr. Loghavi said (Stengel A, et al. Blood Adv. 2023;7[13]:2952–2956). In the majority of TP53-mutated cases, mutation frequency confers a bad prognosis. But when the frequency of TP53 mutations is not high or when the TP53 mutation co-occurs with a mutation like an NPM1 mutation, or in a core-binding factor or an APL, “it usually doesn’t have the same awful prognosis that the other cases do,” though the number of cases is low so the studies are not extensive, she said. When such a case is seen, she advises conveying the difficulty of the classification, noting the presence of co-occurring good and bad mutations, and explaining that the significance of the prognosis is unknown.
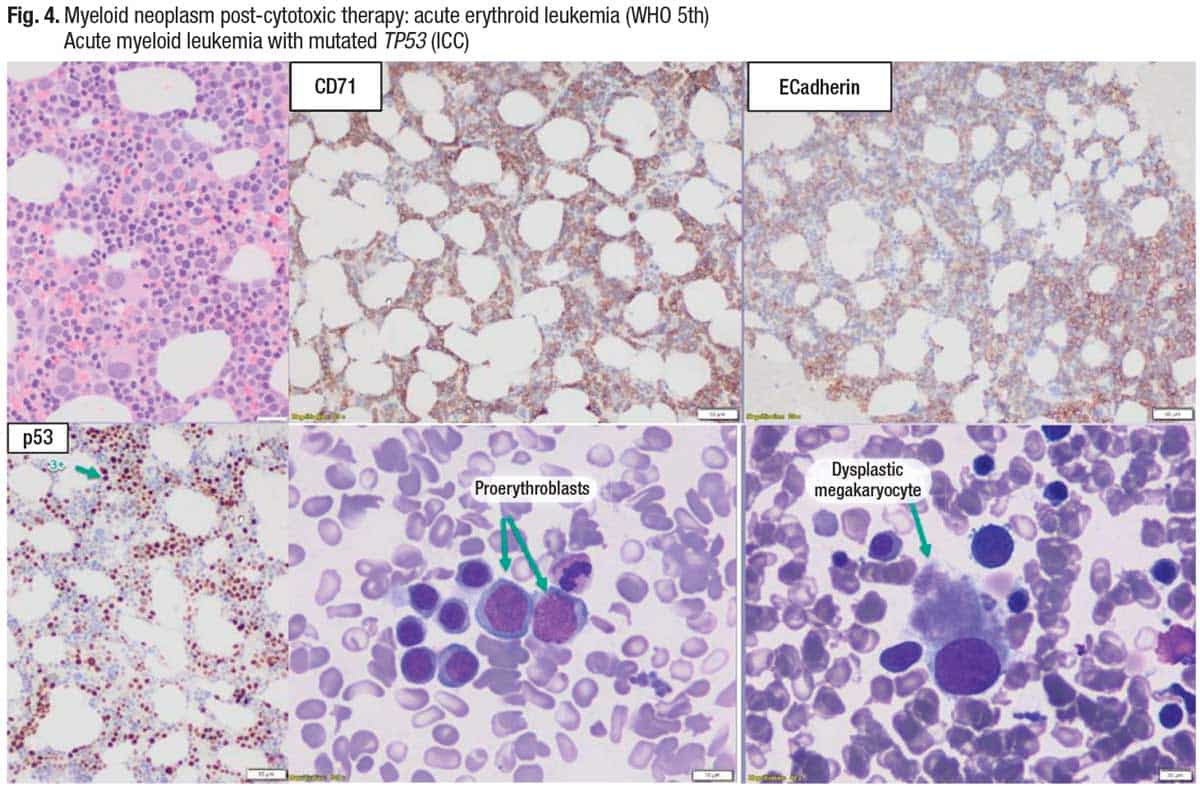
In the third case Dr. Loghavi presented, a 61-year-old female with a past medical history of breast cancer treated with chemotherapy undergoes a workup for cytopenia that identified a TP53 mutation (p. D281G) with a VAF of 76 percent.
(The TP53 mutation is multi-hit because the 76 VAF is more than 50 percent, she noted, adding, “Even if there’s no deletion, this has copy neutral loss of heterozygosity.”)
The CBC reveals pancytopenia: WBC: 2.3 × 109/L absolute neutropenia, Hgb: 6.9 g/dL, Plt: 25 × 109/L, and MCV: 90 fL.
The bone marrow biopsy (Fig. 4, top left) shows that the megakaryocytes are dysplastic, and there is erythroid differentiation (CD71). Immature erythroid cells are highlighted (E-cadherin), “so you can see these are erythroid blasts.” P53 shows an abnormal pattern of staining, and the two aspirate smear images (bottom middle and right) show increased erythroblasts and a dysplastic megakaryocyte.
“One of the subtle differences in the classification of myeloid neoplasms in the ICC and WHO is that in the WHO, therapy-relatedness trumps the classification,” Dr. Loghavi said. “In the ICC, classification trumps therapy-relatedness.” She prefers the ICC approach “because even if you have a patient who has been exposed to chemotherapy, if they have an AML with NPM1 mutation or an APL, that’s probably not going to behave like a therapy-related myeloid neoplasm.” But most cases are post-therapy and have bad genetics, such as a TP53 mutation or KMT2A rearrangement, and will behave poorly, Dr. Loghavi said. In her practice, she first qualifies the case genetically and then will say the patient has a history of exposure to cytotoxic therapy. “That does both classifications justice and leaves the physician with treatment options.”
In the case of the 61-year-old female patient, there is erythroid differentiation and thus the case would be classified as acute erythroid leukemia in the WHO classification.
On the issue of therapy-relatedness, Dr. Loghavi cited the clonal hematopoiesis risk score. It provides a prognostic framework for clonal hematopoiesis of indeterminate potential and clonal cytopenia of undetermined significance by distinguishing a high-risk minority from the majority of CHIP/CCUS, which has a minimal risk of progression to myeloid neoplasm (Weeks LD, et al. NEJM Evid. Published online April 25, 2023. doi:10.1056/EVIDoa2200310).
“If healthy individuals who have a TP53 mutation are not exposed to chemotherapeutic agents, you don’t disrupt the environment, they can live a long time with a high-VAF TP53. It will just sit there,” Dr. Loghavi said. “For reasons that are biologically unclear to me, when you do the stain, you know there is a mutation, but it seems that the cells are able to discard the mutant p53, so you don’t see an abnormal pattern of staining. But once you expose these patients to chemotherapy, then you select for these TP53-mutated clones because you’re killing the wild-type cells, but these cells don’t undergo apoptosis. So you’re selecting for them and waiting for the second hit to happen. Once the other copy is deleted or mutated, it explodes. This is how you get therapy-related myeloid neoplasms enriched for TP53 mutations compared with de novo TP53-mutated neoplasms.”
Does the allelic status matter in the setting of TP53-mutated myeloid neoplasms? “At least in one study, if you have a therapy-related myeloid neoplasm, it doesn’t matter if you’re multi-hit or single hit,” Dr. Loghavi said. “You’re going to do poorly. The only thing that matters is the burden of the clone” (Hiwase D, et al. Blood. 2023;141[9]:1087–1091).
In the case of a therapy-related myeloid neoplasm with a TP53 mutation, as long as the VAF is less than 10 percent (in practice such cases are rare, she said), patients may not do as poorly, Dr. Loghavi said. “You can postulate that maybe the therapy-related neoplasm, in the presence of a VAF less than 10 percent, is not really the driver of the disease. Maybe the driver of the disease is something else.”
Amy Carpenter is CAP TODAY senior editor.

