From the CAP Cytopathology Committee;
Jordan Paul Reynolds, MD, Derek B. Allison, MD, Editors
Derek B. Allison, MD
Jordan P. Reynolds, MD
Lauren Stark, DO
May 2025—Granulomas are organized clusters of immune cells that form as part of the body’s chronic inflammatory response, typically triggered by persistent antigens, chronic infections, or immune dysregulation. They develop when macrophages are activated, transforming into epithelioid histiocytes and multinucleated giant cells (MGC) in response to ongoing immune stimulation. Granulomas are usually surrounded by T lymphocytes, fibroblasts, and extracellular matrix components. While granulomas are most often associated with infections (Fig. 1) or autoimmune conditions like sarcoidosis, they can arise in a variety of other processes, including malignancy.1
In the context of malignancy, granulomas may develop in response to cancer-related antigens, abnormal keratin, extracellular mucin, or necrotic tumor debris, or as part of the immune response to treatment. Granulomatous inflammation can present a diagnostic challenge, particularly when it obscures an underlying tumor or leads to an immediate misinterpretation as infection, causing the rest of the features to be overlooked. This challenge is relevant in cytopathology, particularly in the context of quick decision-making during rapid onsite assessment (ROSE). A granuloma seen during ROSE does not automatically exclude malignancy; careful evaluation of all features is required, as malignancy may still be present despite the granulomatous reaction.
Mechanisms linking tumors and granulomas. Granulomatous inflammation can be an important immune response to eliminate tumor cells or their byproducts. As previously mentioned, tumor-induced granulomas can form in reaction to necrotic tissue, materials like keratin and mucin, or chronic immune stimulation within the tumor microenvironment. Furthermore, particularly in areas where certain fungal organisms are endemic, patients may have coexisting malignancy and fungal organisms, particularly in the lung or mediastinal lymph nodes.2
In addition, certain therapies, including chemotherapy and immune checkpoint inhibitors (ICI), can induce granulomatous inflammation. For example, ICIs such as anti-PD-1 (nivolumab, pembrolizumab), anti-PD-L1 (atezolizumab), and anti-CTLA-4 (ipilimumab) enhance T-cell activity against tumors but can also induce granulomatous inflammation as a secondary effect.3 Tumor necrosis or microbial components released during immunotherapy may act as antigens, triggering macrophage activation and granuloma formation.
Granulomas resulting from ICI therapy can mask underlying tumor progression or be mistaken for infections if the tumor component is not generously sampled on FNA. In addition, ICIs are known to reactivate latent infections (e.g. tuberculosis, histoplasmosis) or cause sarcoidosis-like reactions, which may lead to new or worsening granulomas that are not secondary to malignant progression.4 Chemotherapeutic agents, such as taxanes, methotrexate, and bleomycin, can also lead to granulomatous inflammation. Taxanes are linked to granulomatous pneumonitis and dermatitis,5,6 methotrexate to pulmonary granulomas and sarcoidosis-like reactions,7 and bleomycin to granulomas in the context of lung disease.8 Therapy-induced granulomas can complicate the evaluation of cytology samples, particularly when clinical features such as bilateral hilar lymphadenopathy and pulmonary nodules raise concern for tumor recurrence, sarcoidosis, or infection.
Tumors commonly associated with granulomas. Granulomas can coexist with malignancy, but key findings can help the cytopathologist avoid misdiagnosis.
Squamous cell carcinomas. Granulomas can be seen surrounding keratin debris and necrotic tumor cells in squamous cell carcinomas (SqCC) from any primary site. However, rather than viewing the granuloma in isolation, it is crucial to continue evaluating the entire slide to recognize any SqCC component that may be present. During FNA of neck lymph nodes, granulomatous inflammation is frequently seen in metastatic SqCC. Distinguishing SqCC from granulomas involves identifying key characteristics such as cellular atypia, keratin pearls, and high mitotic activity, which contrast with the organized structure of granulomas composed of epithelioid macrophages and MGC. Identifying this atypical population is crucial to avoid misinterpretation of granulomatous inflammation as simply infectious, rather than as a secondary reaction to the underlying cause—malignancy.
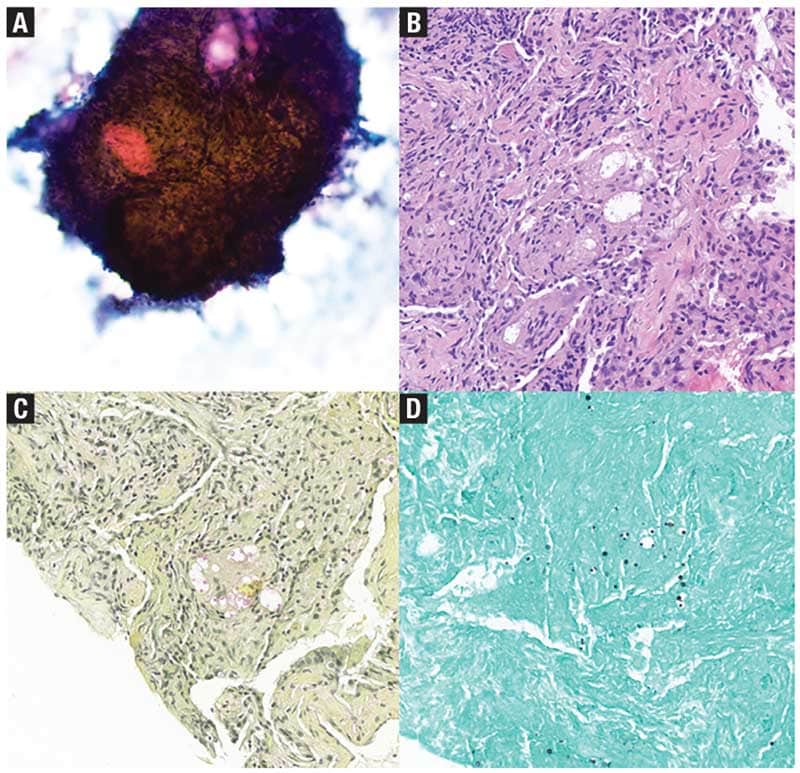
Adenocarcinomas. Mucin from gastrointestinal, pancreatic, or lung adenocarcinomas can act as a foreign substance during tissue invasion and metastasis, leading to a granulomatous reaction (Fig. 2). Granulomas may be observed within mucin where MGC may attempt to phagocytize the material. On Romanowsky-stained slides, mucin typically appears as pale, slightly vacuolated material that may exhibit a faint metachromatic or blue hue. In contrast, on Pap-stained slides, mucin is more distinctly visible as pale blue to greenish, granular, or vacuolated material. Neoplastic cells are often embedded within this material on smears, where granulomas may also be present. When malignant gland-forming structures are not evident, identifying atypical neoplastic cells within mucin aggregates or in a background of necro-inflammatory debris—particularly in cases with colorectal primaries—can aid in making the diagnosis.
Hodgkin lymphoma. Granulomas are occasionally associated with Hodgkin lymphoma, particularly around Reed-Sternberg cells, making for a convincing mimicker of sarcoidosis. The simplest way to identify Hodgkin lymphoma is to see the Reed-Sternberg cells, appearing as large binucleate/multinucleate cells with prominent nucleoli. However, these hallmark cells can be elusive even in excisional biopsy specimens and may not be present in smaller specimens such as an FNA. Abundant eosinophils or prominent fibrous septa may be helpful when Reed-Sternberg cells are rare.
Non-Hodgkin lymphomas. Non-Hodgkin lymphomas such as follicular lymphoma, small lymphocytic lymphoma, diffuse large B-cell lymphoma (DLBCL), and some T-cell neoplasms have also been associated with granulomatous inflammation, though less commonly than in Hodgkin lymphoma. DLBCL cells may occasionally have abundant cytoplasm and can share some morphologic features with epithelioid macrophages. However, this population should be abundant and associated with abnormal chromatin and apoptotic debris. At the time of ROSE, lymphoid neoplasms tend to smear as single cells, whereas granulomas often stay partially intact. When a lymphoid neoplasm is suspected in the background of granulomatous inflammation, every effort should be made to obtain material for flow cytometry and not just microbiology cultures.
Seminomas. Seminomas are the most common type of testicular germ cell tumors and frequently metastasize to retroperitoneal lymph nodes, as well as other sites. Furthermore, mediastinal germ cell tumors may present with mediastinal adenopathy or lung metastasis. On FNA, seminomas usually display discohesive tumor cells with clear cytoplasm and centrally located and enlarged nuclei, often with prominent nucleoli. Tumors are often associated with a robust lymphocytic-rich inflammatory response, and well-formed granulomas may be found in this inflammatory milieu. Epithelioid cells in granulomas are smaller than the seminoma cells and have less prominent, slightly elongated nuclei. The key is recognizing the enlarged, discohesive tumor cells, which often show a tigroid background on smears, which should not be present in infectious or autoimmune-related granulomatous inflammation.
High-grade sarcomas. High-grade sarcomas may have extensive tumor necrosis, leading to the formation of obscuring granulomatous inflammation. The underlying sarcoma can be distinguished by atypical features such as nuclear pleomorphism, hyperchromasia, and distinct (sometimes bizarre) mitotic figures. The identification of spindled cells can be helpful, but many sarcomas can have epithelioid cells that may resemble the epithelioid macrophages of a granuloma. MGC can be seen in both granulomas and in association with sarcomas. Histologically, MGC within granulomatous inflammation should be seen in a central location surrounded by macrophages and lymphocytes whereas the MGC of sarcomas are haphazardly arranged and surrounded by atypical neoplastic cells. However, on FNA smears and core biopsy touch preps, recognizing this regional distribution is not possible. The cytopathologist should pay close attention to all of the features on the slide.
Melanoma. Granulomatous inflammation is an uncommon but recognized reaction in association with metastatic melanoma, which most frequently occurs as a reaction to melanin pigment spillage. The simplest way to identify the tumor is to identify markedly atypical cells with prominent nucleoli that may or may not have pigment. In most cases, the macrophages with pigment will be distinguishable for their lack of cytologic atypia.
Practical ancillary testing. At the time of ROSE, the Romanowsky stain can be helpful in identifying mucin production as a potential etiology for granulomatous inflammation, as well as to highlight any fungal organisms. On alcohol-fixed Pap-stained smears, keratin production is well visualized and may obviate the need for additional testing. If cell block material is available, a mucicarmine stain can be used to confirm mucin production while immunohistochemical stains can be employed to highlight any obscured malignant population. A standard panel of IHC, such as pancytokeratin, p40, PAX8, CDX2, and others, can be chosen based on the morphology of the cancer, the location of the lesion, and the clinical history.
Nonepithelial tumors that are associated with granulomatous inflammation will require their own unique stains. OCT3/4, SALL4, and CD117 will be positive in seminomas. Melanomas may be positive for any combination of MelanA, S100, SOX10, or HMB45, though more poorly differentiated melanomas will express fewer of these markers. Hodgkin lymphoma can be challenging to diagnose on small specimens, even with IHC staining. Reed-Sternberg cells will be negative for CD45, show expression of typical B-cell markers such as CD20 or BOB1, and express CD15 and CD30. Flow cytometry and molecular studies for clonality are helpful in non-Hodgkin lymphomas but do not typically aid in the diagnosis of Hodgkin lymphoma. In situ hybridization for Epstein-Barr virus can assist in identifying lymphoproliferative disorders such as lymphoid granulomatosis and variants of DLBCL or Hodgkin lymphoma.
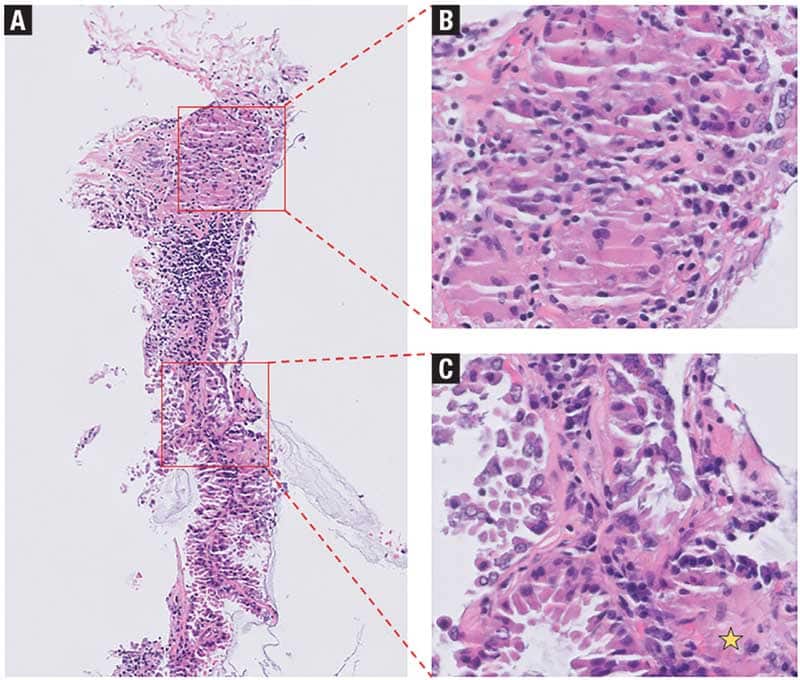
If all avenues have successfully ruled out a malignant process, AFB, PAS, and GMS may be performed to exclude infectious causes of granulomas.
Conclusion. Granulomas can coexist with tumors, potentially masking their presence or even mimicking a malignant process in the absence of a tumor, leading to diagnostic pitfalls that carry significant risks for the patient. As a result, it is crucial to remember that granulomatous inflammation is not a specific finding for infection or autoimmune disease, and every effort must be made to evaluate all the features of the case. Ancillary tests, such as immunohistochemical staining and special stains, provide additional clarity, particularly in challenging cases where routine preparations are inconclusive due to abundant necrosis, poor cellularity, or other obscuring artifacts. Understanding these diagnostic challenges and employing a comprehensive approach ensures that patients receive the most accurate diagnosis and appropriate, timely treatment.
- Pagán AJ, Ramakrishnan L. The formation and function of granulomas. Annu Rev Immunol. 2018;36(1):639–665.
- Devalla L, Ghewade B, Wagh P, Alone VD, Annareddy S. Co-existing fungi: an unforeseen combo creating a dilemma in diagnostic morale. Cureus. 2024;16(8):e67600.
- Cornejo CM, Haun P, English J III, Rosenbach M. Immune checkpoint inhibitors and the development of granulomatous reactions. J Am Acad Dermatol. 2019;81(5):1165–1175.
- Stroh GR, Peikert T, Escalante P. Active and latent tuberculosis infections in patients treated with immune checkpoint inhibitors in a non-endemic tuberculosis area. Cancer Immunol Immunother. 2021;70(11):3105–3111.
- Leger P, Limper AH, Maldonado F. Pulmonary toxicities from conventional chemotherapy. Clin Chest Med. 2017;38(2):209–222.
- Prieto-Torres L, Llamas-Velasco M, Machan S, et al. Taxanes-induced cutaneous eruption: another histopathologic mimicker of malignancy. J Eur Acad Dermatol Venereol. 2016;30(4):638–644.
- Di Francesco AM, Pasciuto G, Verrecchia E, et al. Sarcoidosis and cancer: the role of the granulomatous reaction as a double-edged sword. J Clin Med. 2024;13(17):5232.
- Dong J, Yu X, Porter DW, Battelli LA, Kashon ML, Ma Q. Common and distinct mechanisms of induced pulmonary fibrosis by particulate and soluble chemical fibrogenic agents. Arch Toxicol. 2016;90(2):385–402.
Dr. Allison is associate professor of pathology and urology and vice chair for research at the University of Kentucky College of Medicine, Lexington. Dr. Reynolds is professor of laboratory medicine and pathology and director of cytology, Mayo Clinic Florida. Dr. Stark is a PGY-3 pathology resident at Washington University School of Medicine in St. Louis. All are members of the CAP Cytopathology Committee.

