Summary
A recent article in Archives of Pathology & Laboratory Medicine emphasizes the importance of gender-inclusive care in cytopathology laboratories. The authors, members of the CAP Cytopathology Committee, identify four key areas for improvement: laboratory information systems and terminology, cervicovaginal Pap test screening, HPV testing, and anal Pap test screening. They advocate for gender-inclusive data entry, understanding changes from hormonal therapy, equitable access to HPV testing, and expanding anal Pap test screening infrastructure.
Valerie Neff Newitt
November 2025—Words matter, and patient advocacy does too, even from behind the scenes.
That’s the essence of an article published in August in Archives of Pathology & Laboratory Medicine on gender inclusion in the cytopathology laboratory (Crumley S, et al. Arch Pathol Lab Med. Published online Aug. 20, 2025. doi:10.5858/arpa.2024-0498-CP).
The authors, all of whom are current or past members of the CAP Cytopathology Committee, reviewed the literature on gender-inclusive care and identified the opportunities that would move cytopathology labs, and thus patient care, forward.
Those opportunities fall into four areas: laboratory information systems and terminology, cervicovaginal (or neovaginal) Pap test screening, HPV testing, and anal Pap test screening.

“It’s people from many different institutions across the country who feel this topic is important, who’ve been taking care of patients and understand this is a gap and we can contribute to doing better,” says coauthor Suzanne Crumley, MD, of those who collaborated to write the article as a call to action to address disparities. She is a member of the CAP Cytopathology Committee and associate professor, Department of Pathology and Immunology, Washington University School of Medicine in St. Louis.
“Trying to understand where people are coming from, learning from patients what they need, being open to that information, and using the knowledge we have from our experience in the laboratory to be the voice of advocacy improves our work in cytopathology and the entire spectrum of health care,” Dr. Crumley says.
“Even though we’re often behind the scenes and not as forward-facing as other members of the clinical team, we are still patient advocates,” she adds.
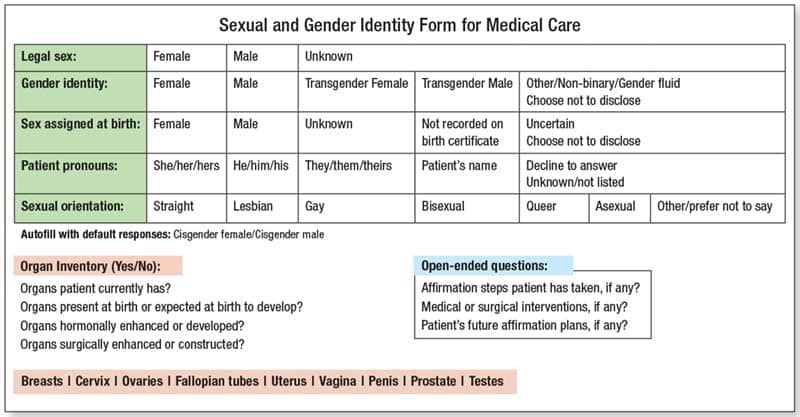
One of the things they advocate is gender-inclusive data entry in the LIS for cytology accessioning, and they provide an example—adapted from a form used in one of the authors’ institutions—of how patient characteristics can be documented uniformly and accurately in the medical record (Fig. 1).
“One of the things I really like is the organ inventory section where the clinician can select the organs the patient currently has and the organs present at birth or expected at birth to develop,” Dr. Crumley says of the form. “There’s a lot more complexity to gender identity than sex at birth, and being able to do that organ inventory, having the space to document the complexity and communicate it effectively, are important aspects of our job.” It’s important in many patient cases, among them the workup of a cancer of unknown primary. “If we don’t have a correct gender or sex or even know what primary organs that patient may have, we could go down the wrong path,” Dr. Crumley says. The template is a good starting point for other institutions.
“We in pathology know more than anyone that words are important,” she says. In the early days of work on their paper, committee members spoke of the ways in which their work is limited by the constraints on input of data when requisitions are generated.
Dr. Crumley and coauthors write in the article that identifying cancers or metastatic disease from the Müllerian and Wolffian ducts may be problematic without an accurate documentation of patient sex or natal organs at birth. “But it’s important to recognize there may be legal or other barriers to providing some of this information,” Dr. Crumley says of patients and providers. Thus, in any given case, the morphologic features may not match assumptions or expectations, she adds, crediting the feedback and other input provided by members of the CAP Diversity, Equity, Inclusion, and Accessibility Committee.
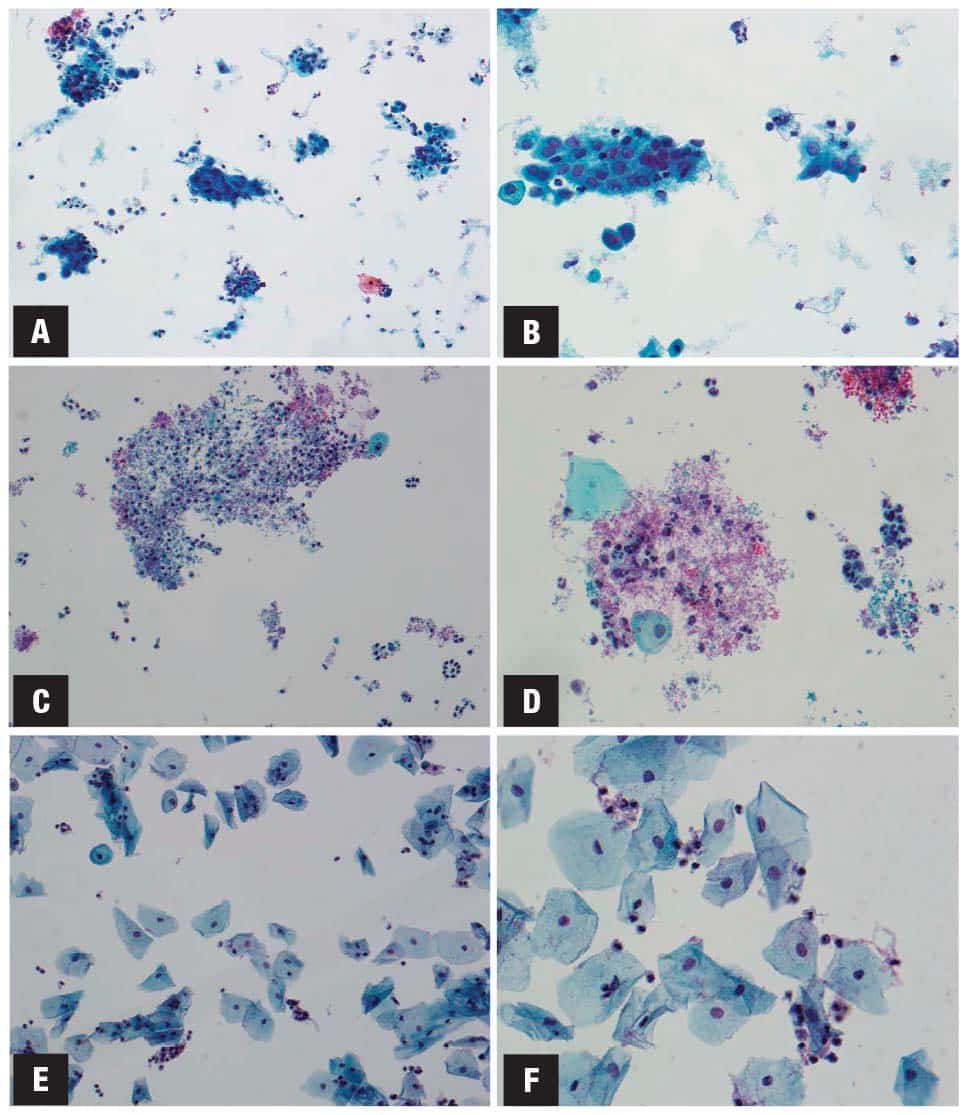
Another of the gaps they identify and advocate is an understanding of the changes that can be seen with hormonal or gender-affirming therapy (Fig. 2).
The changes have been well reported, she says, and can mimic dysplasia. “Furthermore, if there is a neovaginal space formed of a different type of epithelium, cells would look different than expected and there could be fewer cells than would normally be seen in a Pap test,” Dr. Crumley says. Cytopathologists need to be aware of the three cell types seen in neovaginal Pap tests: squamous, intestinal glandular type, and mesothelial/peritoneal.
HPV testing is the third area they address, from ensuring equitable access to screening to examining its validity in anal, neovaginal, and cervicovaginal sites, as well as understanding the reference HPV infection rate in minority patient populations. Self-collection is important, they say, noting the Food and Drug Administration’s recent approval of self-collection by vaginal swab in a health care setting. But understanding the logistics is important too, including the appropriate follow-up, she says. “I think a lot of that is still being worked out, for all populations.” The hope, she says, is that it will be approved someday for use at home or in other private settings and reach people who may not otherwise have access to cervical or other HPV-related cancer screening.
Also unknown now is how primary HPV screening will perform in a transgender population. “It’s a small subset and one that is difficult to study for various reasons,” Dr. Crumley says.
The final area the authors address is anal Pap test screening, which Dr. Crumley says fewer practitioners offer or feel comfortable with compared to cervicovaginal Pap tests, creating uneven access. “And when you dig into the issue of better access to screening, you learn that this [anal Pap test screening] applies to broader populations.”
The International Anal Neoplasia Society’s guidelines for anal cancer screening were published last year (Stier EA, et al. Int J Cancer. 2024;154[10]:1694–1702). The IANS recommendations provide guidance for which populations should be offered screening and for managing abnormal screening results. The guidelines provide a basis for expanding screening infrastructure to all at-risk populations, the guideline authors write.
“It could benefit many more people,” Dr. Crumley says of anal Pap test screening, “and there’s a lot to be learned from it that can be applied in other areas.” Thus there’s a need to broaden education across the board.
Understanding and advocating for all patients is the message she and her coauthors leave their readers with in their call to action as it’s set forth in the Archives article. They write, “Greater awareness and incorporation of these practices in the cytopathology laboratory will help to provide more accurate and inclusive care for all patients.”
Cytopathologists collectively have the experience in screening for cervical cancer with the Pap test, quality assurance in laboratory testing, and laboratory information systems to improve care for all patients, Dr. Crumley says, “but it is especially valuable in ensuring optimal gender-affirming care.”
“We can do better,” she says.
Valerie Neff Newitt is a writer in Audubon, Pa.

