The morphologic and immunophenotypic features favored infantile fibrosarcoma (IFS), but the patient’s age, tumor location, and marker profile prompted consideration also of malignant peripheral nerve sheath tumor, synovial sarcoma, and spindle cell rhabdomyosarcoma. IFS is most commonly driven by ETV6::NTRK3 fusions; by contrast, NTRK1 fusions are relatively rare in the pediatric IFS spectrum and broader pediatric mesenchymal tumors, underscoring the value of directed testing in diagnostically ambiguous cases.1,2
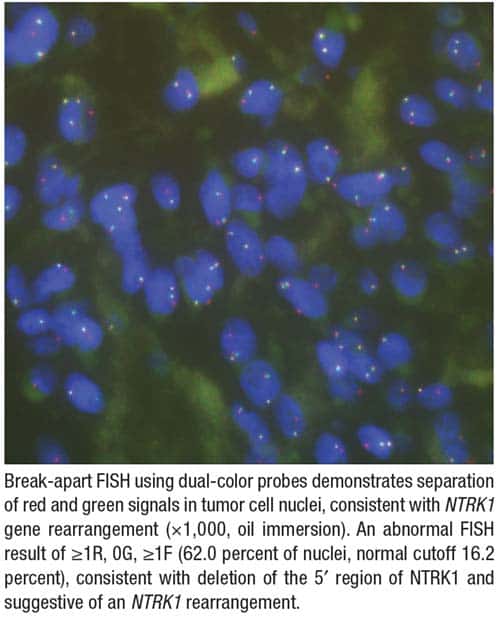
Given the diagnostic uncertainty, fluorescence in situ hybridization for NTRK gene rearrangements demonstrated separation of red and green signals consistent with an NTRK1 rearrangement (Fig. 2). This established the diagnosis of NTRK1-rearranged spindle cell sarcoma, IFS-like—an entity now recognized within the spectrum of kinase fusion-driven spindle cell neoplasms.3,4 Although RNA-based fusion testing was not available, targeted DNA-based next-generation sequencing using the genomic sequencing panel on the Illumina MiSeq identified an in-frame TPR::NTRK1 fusion (TPR exon 10, NM_003292; NTRK1 exon 10, NM_002529), providing molecular confirmation of the FISH finding.
NTRK-rearranged spindle cell neoplasms frequently show S100 expression with variable coexpression of myoid or CD34 markers and often feature hemangiopericytoma-like vasculature and stromal hyalinization, aligning with our histology and IHC pattern.5 While most NTRK fusions in IFS are ETV6::NTRK3, several series highlight the relative scarcity of NTRK1 fusions in pediatric mesenchymal tumors, including IFS-like lesions.1,2
Clinically, identification of an NTRK fusion is therapeutically actionable. TRK inhibitors such as larotrectinib and entrectinib have demonstrated high response rates across tumor types and ages, including children, with durable benefit in pooled analyses and basket trials. This supports consideration of targeted therapy in advanced or unresectable disease settings.6–8

- Kang J, Park JW, Won JK, et al. Clinicopathological findings of pediatric NTRK fusion mesenchymal tumors. Diagn Pathol. 2020;15(1):114.
- Tauziède-Espariat A, Duchesne M, Baud J, et al. NTRK-rearranged spindle cell neoplasms are ubiquitous tumors of myofibroblastic lineage with a distinct methylation class. Histopathol. 2023;82[4]:596–607.
- Sbaraglia M, Bellan E, Dei Tos AP. TRK-rearranged spindle cell neoplasm (emerging). In: WHO Classification of Tumours Editorial Board, eds. Soft Tissue and Bone Tumours. Vol. 3. IARC Press; 2020:287–289. WHO Classification of Tumours; 5th ed.
- Suurmeijer AJH, Dickson BC, Swanson D, et al. A novel group of spindle cell tumors defined by S100 and CD34 co-expression shows recurrent fusions involving RAF1, BRAF, and NTRK1/2 genes. Genes Chromosomes Cancer. 2018;57(12):611–621.
- Brčić I, Godschachner TM, Bergovec M, et al. Broadening the spectrum of NTRK rearranged mesenchymal tumors and usefulness of pan-TRK immunohistochemistry for identification of NTRK fusions. Mod Pathol. 2021;34(2):396–407.
- Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in TRK fusion–positive cancers in adults and children. N Engl J Med. 2018;378(8):731–739.
- Cocco E, Scaltriti M, Drilon A. NTRK fusion-positive cancers and TRK inhibitor therapy. Nat Rev Clin Oncol. 2018;15(12):731–747.
- Rosa K. FDA expands entrectinib indication in pediatric patients with NTRK+ solid tumors. OncLive. Oct. 20, 2023.
Dr. Qiu is a pathology resident and Dr. Takeda is attending pathologist—both in the Department of Pathology, Westchester Medical Center and New York Medical College.

