Karen Lusky

April 2017—In a presentation at CAP16 on common patterns of liver injury, Robert M. Najarian, MD, called drug-induced liver injury the great mimicker in liver pathology. “When in doubt, rule drugs out,” he said, including herbal agents, supplements, and over-the-counter agents.
The effect of drugs on the liver can imitate many different patterns of liver injury, including hepatitic, vascular, and cholestatic, among others, said Dr. Najarian, a consultant in gastrointestinal, hepatobiliary, and pancreatic pathology at Beth Israel Deaconess Medical Center and Harvard Medical School. He described the image of an amoxicillin-clavulanic acid (Augmentin)-induced liver injury as essentially simulating a cholestatic pattern with bile ductular proliferation, portal tract edema, and neutrophils. “You could easily call this duct obstruction, if you didn’t know better that a patient had started a new drug recently,” he said (Fig. 1).
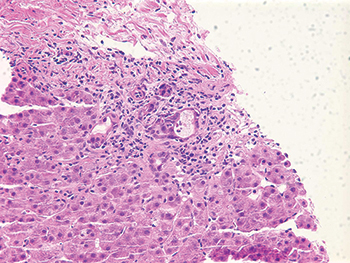
“So when in doubt, I say rule out drugs, including over-the-counter drugs [and herbal supplements] that patients may be taking but not telling their physicians about,” Dr. Najarian advised. “We have certainly seen several cases of acute liver failure from herbal supplements, oftentimes muscle-building supplements, causing a lot of damage and acute liver damage that is persistent.”
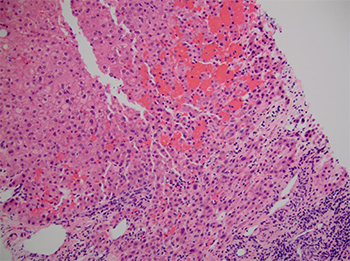
Dr. Najarian displayed a biopsy image of an acute toxic liver injury caused by over-the-counter weight-loss supplements (Fig. 2). “Zone 1 hepatocytes around the portal tract itself are preserved because these hepatocytes are the best perfused and least susceptible to oxidative stress, while centrilobular hepatocytes have become damaged,” he said in an interview with CAP TODAY. An acute toxic injury can mimic the pattern of an acute vascular ischemic injury to the liver.
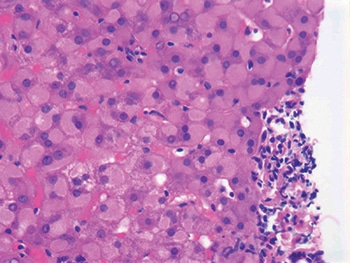
Cytotoxic chemotherapeutic agents, among others, can damage the specialized sinusoidal endothelial cells, which causes a vascular pattern of liver injury with congestion and hemorrhage around the terminal hepatic or central vein, Dr. Najarian says. “With chronic damage, complete occlusion of the central vein can occur, resulting in what’s clinically known as sinusoidal obstruction syndrome or veno-occlusive disease.”
Dr. Najarian shared an image of what he called “a relatively bland-looking liver injury,” except for a centrally located bile plug. The patient was on chemotherapy for multiple myeloma. “The question that we often are asked: Is it the drug or is it the disease that’s causing the liver injury? Sometimes we can tell, but sometimes we can’t.” When patients are started on cytotoxic chemotherapeutic agents, he noted, some respond with liver injury from the drug, while infection and the neoplastic processes can cause the injury in other instances.
People often want to know what sort of pattern of injury he would expect if the patient were taking a certain drug. “Sometimes we have an answer because of what has been reported in the literature,” he said. Allopurinol use presents with fibrin ring granulomas, while amiodarone mirrors the histologic features of alcoholic steatohepatitis.
“But what we have certainly found, and it’s probably your experience too,” Dr. Najarian told CAP16 attendees, “is that many drugs have a multitude of injury patterns.” For additional information, he recommended a review article on drug-induced liver injury in the July 2015 Archives of Pathology & Laboratory Medicine, coauthored by Romil Saxena, MBBS, a professor of pathology and laboratory medicine and a professor of medicine in the Division of Gastroenterology and Hepatology, Indiana University School of Medicine (Fisher K, et al. Arch Pathol Lab Med. 2015;139[7]:876–887).

It can take weeks to many months for a drug-induced liver injury to resolve, Dr. Najarian says. “Often these medications are only taken for a week or so, but they can still cause a prolonged period of injury. Thus, it’s important for pathologists to ask those who send their patients for liver biopsy about a patient’s drug/medication use history for up to a year prior to the procedure,” including dosage adjustments.
Some drugs, such as those that cause autoimmune hepatitis, may create “irreversible, self-perpetuating damage,” Dr. Saxena tells CAP TODAY. “The autoimmunity might be self-perpetuating. It won’t go away in some patients. What probably happens is the drug unmasks a hidden epitope or it combines with molecules within a cell to create a ‘novel epitope’ that the body recognizes as foreign.”
In many but not all cases of drugs mimicking autoimmune hepatitis, or AIH, elevated antinuclear antibody titers and significantly elevated anti-smooth muscle titers will be seen, Dr. Najarian said. “So do not use merely these as confirmation of a primary autoimmune hepatitis. You really need help from your clinician to exclude a drug as the cause of this acute pattern of immune-mediated injury.”
Pathologists can also use LiverTox (livertox.nlm.nih.gov). LiverTox is “a very unbiased source of a compound’s potential to cause drug toxicity,” says Naga Chalasani, MD, an investigator for the Drug-Induced Liver Injury Network, known as DILIN, which the National Institutes of Health established in 2004. “The network’s purpose is to better characterize this disease,” Dr. Chalasani explains. LiverTox “isn’t part of DILIN but has been involved in its development.”
DILIN is conducting a prospective study of nearly 1,900 patients enrolled at several centers in the U.S. who are followed from six months to two years so the condition can be studied, says Dr. Chalasani, who is the David W. Crabb professor and director of the Division of Gastroenterology and Hepatology, Indiana University School of Medicine. (DILIN also has an ongoing retrospective study but the network’s primary objective is the prospective study.)

The DILIN study protocol requires eliminating all competing etiologies of the patient’s liver disease. “In clinical practice, you want to exclude competing etiologies,” Dr. Chalasani says. “Sometimes what seems very much like a drug-induced liver injury is not.” Even some of the cases enrolled in the DILIN study that appeared to be due to anesthetics and other things turned out to be hepatitis C or E. And some cases that appeared to be caused by Augmentin turned out to be pancreatic cancer. “So it goes both ways,” Dr. Chalasani says. Sometimes practitioners don’t suspect drug-induced liver injury, and sometimes they over-diagnose it just because the patient was put on a medication.
“There are instances where drugs may exacerbate underlying autoimmune hepatitis or other types of underlying liver disease,” Dr. Chalasani says. Drugs—tamoxifen, for example—can worsen fatty liver disease. Although it’s not directly a drug-induced injury, immunosuppressants or chemotherapy can activate hepatitis B or C.
“Twenty percent of drug-induced liver injury cases we see are from dietary supplements, which are poorly regulated and poorly labeled,” Dr. Chalasani says. “What’s on the label is not necessarily what you sometimes find when you actually test those supplements chemically.” Weight-loss and body-building supplements make up the two large groups of herbal and dietary products to cause drug-induced liver injury.
Herbal extracts can have thousands of chemical compounds to which some people will react badly, says George K. Michalopoulos, MD, PhD, a professor in and chair of the Department of Pathology at the University of Pittsburgh. He points out that drugs and xenobiotic compounds are largely metabolized by the cytochrome P450 (CYP) family of liver enzymes. “Typically, hydrophobic compounds are converted to water-soluble metabolites to facilitate excretion. In some situations, the CYP-dependent metabolism generates intermediate products that act as free radicals and can cause widespread [liver] damage.”
If someone takes an herbal medicine, he says, each one of the compounds in there may alter the relative proportions of the CYP family members. “And you may have a situation where the person who took [the herbal extract] will have a very different CYP variation and change in the proportion of these enzymes so that some drugs that normally wouldn’t have caused any problem would now become problematic.” There’s either an increase in the CYPs that make more of the dangerous metabolites or a change in the proportion of the CYP family members in the liver, so some individuals may produce more of CYP2E1 as opposed to CYP4A11, for example, depending on which herbal supplements they took and where they came from.
In almost all the studies on drug-induced liver injury performed in North America and Europe, Dr. Saxena says, antibiotics come to the top of the list, with Augmentin first. “I think that’s going to change,” she says, “because we have a lot of new drugs to treat autoimmune diseases and cancers, and I think we are going to see some of those rise to the top of the list. We don’t know enough about the PD-1 and PD-L1 inhibitors yet. We will have to watch those.”
“At the present time, however, antibiotics such as Augmentin continue to be the leading causes of liver injury in the United States apart from acetaminophen,” says James H. Lewis, MD, a professor of medicine and director of hepatology at Georgetown University Hospital in Washington, DC. Augmentin appears to pose a nearly negligible risk of hepatotoxicity in children, Dr. Chalasani says, with the risk increasing after age 50.
What if the patient is taking multiple drugs that could be injuring the liver? Dr. Saxena reports that at Indiana University School of Medicine and other institutions experienced in drug-induced liver injury, they attempt to establish a timeline and then may stop the most likely drug first to see what happens. “Very rarely will we stop all the drugs, although it depends—you can’t stop a drug that’s being taken for something that’s life-threatening, for example. We might change the drug to a different class [or] sometimes a different drug belonging to the same class.”

Before the Drug-Induced Liver Injury Network was established, Dr. Chalasani says, the knowledge gaps about such injuries were plentiful. “So there are better definitions, a better understanding of the risk factors and the natural history, and also trying to better understand the genetic basis for it. We have made important contributions on all of these drugs.” He and others reported in 2015 on the features and outcomes of 899 patients with drug-induced liver injury (Chalasani N, et al. Gastroenterology. 2015;148[7]:1340–1352). “Among the five most frequent classes of prescription drugs (antimicrobials, cardiovascular agents, CNS agents, antineoplastic agents, analgesics),” the authors write, “there were few clinical differences in the liver injury attributed to their use.”
DILIN and other researchers are also searching for new drug-induced liver injury biomarkers. As Georgetown University’s Dr. Lewis and colleague wrote in a 2016 review article: “Given the relatively limited therapeutic armamentarium currently available to treat specific causes of acute DILI, the ability to predict who is at risk of hepatotoxicity is the subject of active research efforts across the globe” (Stine JG, et al. Expert Rev Gastroenterol Hepatol. 2016;10[4]:517–536).
Says Dr. Lewis: “The currently available liver tests that signal hepatic injury have been in use for dec-ades and are not able to predict liver damage before it happens. Indeed, liver enzymes such as the aminotransferases (AST and ALT) have been around since the 1950s, and injury must occur before those enzymes rise. So the horse is already out of the barn.”
Dr. Lewis notes that many researchers, such as the group headed by Daniel Antoine, PhD, in the United Kingdom, have been looking at newer biomarkers like microRNAs. “Based on early results, these and other potential liver injury biomarkers, including high mobility box 1 and keratin 18, in acetaminophen-overdose patients, might be able to tell us who is at risk for liver damage before it happens. So we might be better able to tailor the antidote to those who are predicted to develop hepatotoxicity,” he says.
The value of measuring liver enzymes, Dr. Michalopoulos says, is that it tells you whether damage involving death of hepatocytes has taken place and whether that injury process is continuing or decreasing. “That’s why it’s valuable, because you may have an acute injury where the levels go up and then over the next five or six days they keep continuously going down. If they don’t, this means the process is more extensive and continues.
“Now what would be good is if we knew exactly what drugs caused that,” Dr. Michalopoulos adds, noting there’s only statistical evidence from drug-induced liver injury cases that appear worldwide. When it becomes possible to have a registry of all variants of the specific CYP enzymes so that some kind of molecular assay, which isn’t currently available, can determine all possible variations of CYP family members in a person’s liver, pathology laboratories utilizing such complex evidence may be able to determine which substances specific individuals should avoid, he says.
Regardless of the nature of the offending chemical, the main importance of liver biopsy is that it allows the pathologist to clearly establish the type of injury that occurred in the liver, Dr. Michalopoulos says.
“The cause (the specific chemical causing the injury) is not under the microscope. A diagnostic pathologist may make a statement that the microscopic findings are most compatible with a viral infection or most compatible with a cholestatic drug injury or something like that.” The pathologist could “definitely orient and focus the process where there is a diagnostic dilemma.”
[hr]
A focus on liver regeneration
Pathologists and researchers continue to home in on how the liver heals from drug-induced injury.
Romil Saxena, MBBS, had an interesting case in which a patient took a medication for weight loss that destroyed the bile ducts in the liver; the patient developed jaundice. “But then the bile ducts regenerated themselves and came back over time and the patient improved,” says Dr. Saxena, a professor of pathology and laboratory medicine and a professor of medicine in the Division of Gastroenterology and Hepatology, Indiana University School of Medicine.
The case “wasn’t so much about the drug but it was about the regenerative capacity of the liver that was totally fascinating for us,” Dr. Saxena adds. She and colleagues published their observations as a case report (Vuppalanchi R, et al. Am J Surg Pathol. 2006;30[12]:1619–1623).

The liver’s ability to repair when injured is “well appreciated” and has been portrayed even by ancient Greeks, says James Luyendyk, PhD, an associate professor of pathobiology and diagnostic investigation, Michigan State University.
“Across multiple models of acute and chronic liver damage,” Dr. Luyendyk says, “there’s strong experimental evidence to suggest there is increased intrahepatic coagulation.” In many cases, this can be detected by markers, including “clot deposition” within an animal’s injured liver, as well as plasma measures of coagulation.
He and his research colleagues initially hypothesized that the fibrin clots in the liver were damaging. “That turned out to be very wrong,” Dr. Luyendyk says. Their research revealed that in acute liver injury, “the primary pro-repair function of fibrinogen” is a non-hemostatic one that drives macrophage activation. “Fibrin binds to the leukocyte integrin Mac-1 (CD11b/CD18),” Dr. Luyendyk explains. Collaboration with Matthew Flick, PhD, of Cincinnati Children’s Hospital Medical Center, “who had developed specific tools to interrogate this particular function of fibrinogen,” enabled this discovery, he says.
Dr. Luyendyk acknowledges that multiple studies have indicated that anticoagulants can reduce early acetaminophen (APAP)-induced liver damage in experimental mice. “And our results completely agree with that. The challenge is that these studies looked only at early injury,” which he defines as six hours after the mice receive APAP. “However, as you let the injury evolve to later times—for example, 24 hours after acetaminophen administration—where the repair processes get kicked on, the effect of anticoagulant on injury switches, revealing protective effects of coagulation.”
“We were also able to utilize a small molecular allosteric activator of Mac-1 called leukadherin-1 to examine whether the fibrinogen-Mac-1 interaction could be therapeutically enhanced to reduce liver injury in APAP-challenged mice,” Dr. Luyendyk says. The drug “ramped up” the protective pathway’s activity and diminished liver injury. Anna Kopec, PhD, an assistant professor and researcher in his lab, presented those findings last month in a poster session at the Society of Toxicology meeting in Baltimore. Their previous research is reported in this month’s Journal of Hepatology (Kopec AK, et al. 2017;66[4]:787–797).

George K. Michalopoulos, MD, PhD, a professor in and chair of the Department of Pathology at the University of Pittsburgh, says that in most scenarios, “if the patient survives the acute crisis, the liver injury eventually cures itself by regeneration. Over the course of one to two weeks, the dead hepatocytes are removed and this is where Dr. Luyendyk is suggesting that we have the cells, the macrophages that eat the dead hepatocytes and take them away.”
“Once that sort of housekeeping process has taken place, then regeneration starts and the live hepatocytes are proliferating to make up the cells that were lost and reconstitute the liver histology.”
If there is a way to get the macrophages to do that housekeeping more effectively, it would certainly be helpful, Dr. Michalopoulos says. Patients should be on appropriately modified diets to avoid metabolic overload of the liver as it regenerates. “And the injured liver should not be given other drugs that are known to cause liver injury. Pharmacological management and a systematic follow-up need to take place.”
Dr. Michalopoulos is the author of an article published this month in Hepatology on how “regeneration is great” if someone has an acute liver injury and recovers (Michalopoulos GK. 2017;65[4]:1384–1392). But with chronic hepatocyte loss and damage, regeneration persists “in an obsessive compulsive way,” he says. “That regeneration can occur against an onslaught of inflammation, of reactive oxygen radicals, lipid peroxides, and a whole variety of things that cause genotoxic damage to the proliferating hepatocytes.” When the genotoxic damage builds up, it “can potentially lead to cancer depending on the genes that have been affected.”
[hr]
Karen Lusky is a writer in Brentwood, Tenn. All images are courtesy of Robert M. Najarian, MD.

