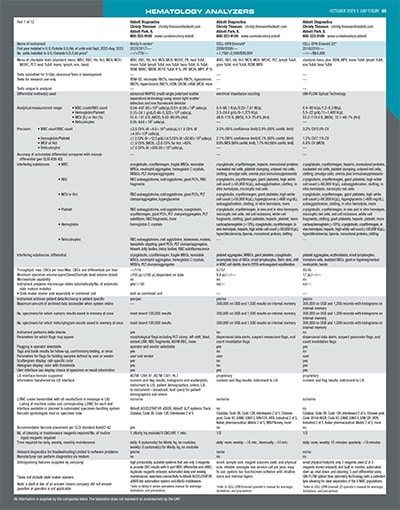
[wpdatatable id=87]
[one_third]
[/one_third]
[two_third_last]
In hematology, making the most of automated solutions
October 2023—Hematology analyzers and the related workflow, expertise, efficiency, and IT matters were the topic of a roundtable when CAP TODAY publisher Bob McGonnagle met online Aug. 29 with two pathologists and representatives from Horiba, Siemens, Sysmex, CellaVision, Sight, and Abbott. Their conversation follows. Click here for CAP TODAY’s guide to hematology analyzers.

Fernando Chaves, what are the advances in artificial intelligence in the field of hematology, particularly automated hematology, since we spoke during our roundtable at this time last year?
Fernando Chaves, MD, global head of hematology, Siemens Healthineers: Technology now enables full-field digital morphology, a full image of the entire slide scan. Now we can do with hematology what has been done for over a decade in surgical pathology. It brings benefits to customers because it not only preclassifies cells and facilitates the technologist’s work but also enables full remote consultation.
Having digital images under a full slide context creates an opportunity for clinical innovations. It’s already happening with applications for reviewing bone marrows and interpreting morphological abnormalities in the blood, and it could also be done in the future through automation and artificial intelligence solutions. Some of the sepsis parameters that are based on hematology are primarily morphologic parameters that are identified through a histogram. Now we have more sensitive technology that can identify abnormalities such as granularity of cells and heterogeneity of volume. All of that is because images are now digital and can be quantitatively analyzed through artificial intelligence image analysis algorithms.
Jonathan Galeotti, for years we’ve hypothesized that there’s more to be learned from studying the cells than we’ve been able to realize. Now we seem to be on the brink of breaking through in several important areas. Fernando mentioned sepsis but there’s also a plethora of hematologic malignancies that we may be able to understand better. How are you dealing with this at the clinical level in your institution at Chapel Hill?
Jonathan Galeotti, MD, clinical assistant professor, Department of Pathology and Laboratory Medicine, Division of Hematopathology, University of North Carolina School of Medicine: As the field moves forward, it takes a little time for it to work its way through academic labs and into clinical practice. We’re on the front end and have not yet incorporated it much into our hematology practice. There is certainly a need—we still have staffing issues. Anything you can do to streamline the process, to make review and autoverification easier, is on our minds all the time. Hope is there, but we haven’t realized it yet.
Continue reading …
[/two_third_last]