[one_third]
[/one_third]
[one_third]
[/one_third]
[one_third_last]
[/one_third_last]
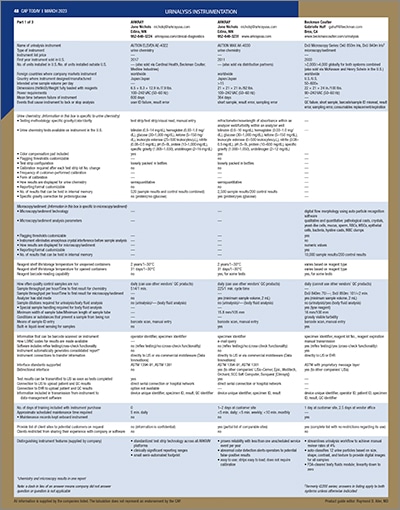
[wpdatatable id=67]
[one_third]
[two_third_last]
In urinalysis, reflex algorithms and other efficiencies
March 2023—Urinalysis was at the heart of a Feb. 7 discussion between CAP TODAY publisher Bob McGonnagle; Ron Jackups Jr., MD, PhD, of Washington University School of Medicine; and Jason Anderson of Sysmex America. “There’s a lot of room to explore what the optimal parameters are to use with the best specificity and sensitivity for a reflex to the sediment analysis or the culture,” Anderson said. Here’s what he and Dr. Jackups said about reflex testing, automation, and middleware.
Since the urinalysis roundtable in December 2021, three things continue to be hot issues regardless of instrumentation, field of analysis, and subspecialty. Number one is automation in workflow, having something that’s robust for high volume, low staffing. Number two is the need for fit of an instrument line. In other words, there’s a core lab urinalysis instrument that’s ideal and yet there are smaller clinics, hospitals, all kinds of sites that can benefit from the same technology but in a differently configured unit. Number three is understanding if we’re extracting the optimum clinical information from the analysis in fields like hematology and urinalysis.

Ron Jackups, talk to us about reflex testing in urinalysis and then more specifically about what you’ve been working on at Barnes-Jewish Hospital.
Ron Jackups Jr., MD, PhD, associate professor of pathology and immunology, Washington University School of Medicine, and associate chief medical information officer for laboratory informatics, BJC HealthCare: Reflex testing in general has two big benefits. The first is it focuses the diagnostic process, which reduces waste. We have seen in urinalysis and other areas that providers, rather than order a test and wait for the result and then order another test based on that result and wait for the next result, et cetera, tend to do what we call shotgun testing—they order all the tests they think might be relevant at once and then react to the results after they get them. This is wasteful in many situations if the tests further down the line were not necessary and can also be dangerous if one of the tests down the line is a false-positive. Part of the goal of reflex testing is to identify situations where there’s a low diagnostic value and high risk of false-positives in future tests that could be prevented by simply not doing them. That’s the first reason to do reflex testing.
Continue reading …
[/two_third_last]