[one_third]
[/one_third]
[one_third]
[/one_third]
[one_third_last]
[/one_third_last]
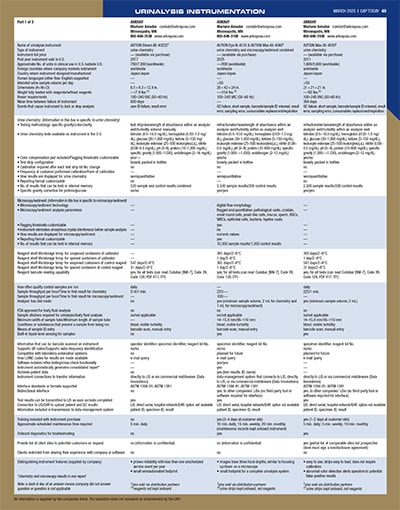
[wpdatatable id=120]
[one_third]
[/one_third]
[two_third_last]
On value, preanalytics, and personnel in urinalysis
March 2025—Urinalysis reflex rules, reimbursement, instruments, research, and specimen handling and transport are some of what came up in the Jan. 15 roundtable with eight participants online and CAP TODAY publisher Bob McGonnagle leading the discussion. What they told CAP TODAY begins here, and our revised guide to urinalysis instrumentation is here.
In our urinalysis roundtable last year there was emphasis on reflex testing as a solution to some of the need for efficiency in the urinalysis workflow. Of course, people in pharmacy and the infectious disease physicians have different views about reflex criteria in urinalysis. We also talked about the need for working rules in the interest of efficiency and workflow amid automation and artificial intelligence. Lastly, we talked about the science of urinalysis, which seems to be underappreciated in the United States. Jason Anderson, can you comment on the current relevancy of those themes?
Jason M. Anderson, MPH, MT(ASCP), senior product manager, urinalysis solutions IVD product marketing, Sysmex America: Reflex testing, when done appropriately, can provide a host of benefits, from improved laboratory efficiency to more timely information for clinicians and ultimately a higher standard of patient care. Determining optimal reflex algorithms can be a challenge. Laboratory practice in urinalysis is not always standardized. Even within an integrated health network, you might see the use of different cutoffs or reflex rules at different facilities. Establishing reflex rules, agreeing on those with the various stakeholders, and making sure they’re optimized and evidence-based to the specific patient populations served are key.
With the progress of AI, machine learning, and expert system disciplines, in conjunction with increasing evidence-based outcome data, I believe we’ll see a strong emphasis on developing even more powerful and advanced reflex testing algorithms that are population specific in many cases. Add to this that analyzer technology is allowing for greater standardization, sensitivity, and specificity in laboratory testing and I see a recipe for making a positive impact on patient care.
Continue reading …
[/two_third_last]