August 2021—Verichem Laboratories offers liquid-stable, protein-based clinical reference materials for total and direct bilirubin assays. The Bilirubin Standard Kit, Tru-Zero Bilirubin Standard, and extended-range Level F Bilirubin Standard are intended for CLIA calibration verification with wet chemistry testing systems. Verichem’s BR2 Bilirubin Calibrator is available for use exclusively with the BR2 Bilirubin Stat Analyzer from Advanced Instruments.
The clinical reference materials incorporate a proprietary stabilization process, the company says, and do not have azides, glycols, or surfactants. Each vial contains 5 mL of material; refrigerated open-vial stability is five days and the shelf life is 18 months when stored at 2°–8°C. Concentration levels are verified using pure crystalline bilirubin and analyzed by the Doumas reference method.
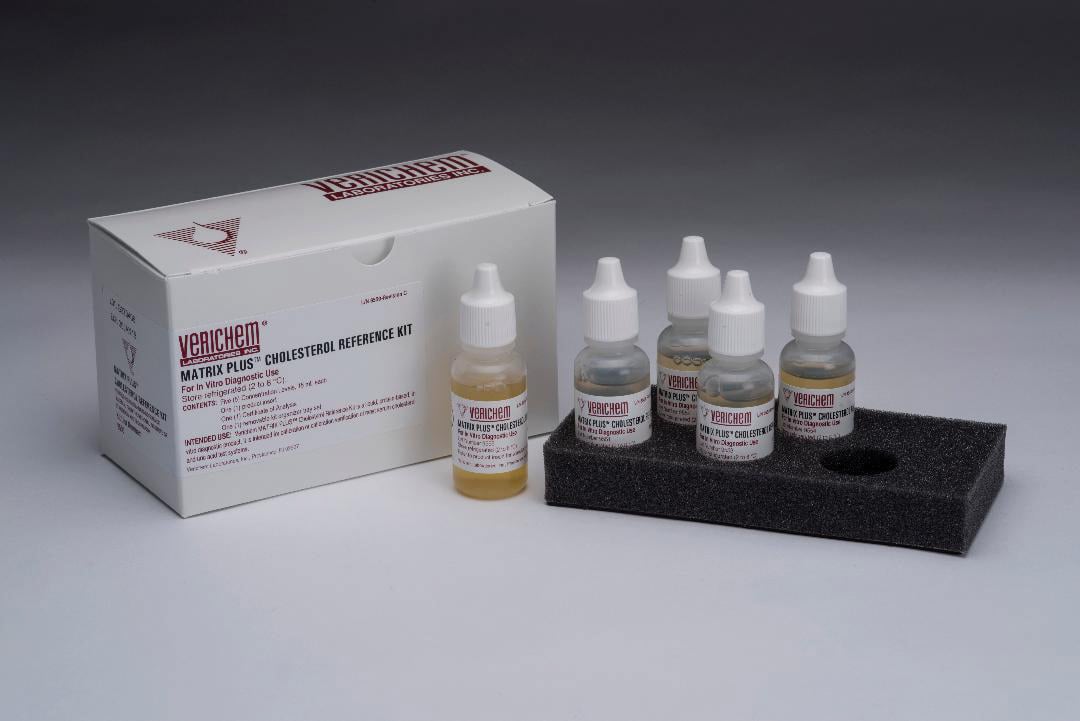
The kit is packaged in a ready-to-use, liquid stable format, incorporating a bovine-sourced, protein-based matrix containing esterified and free cholesterol for true serum-like reactivity. The formulation features minimal lot-to-lot variation and is free of glycols, surfactants, azides, and other interfering substances. Each active component is verified using available standard reference materials from the National Institute of Standards and Technology and documented with a certificate of analysis. The shelf life is 21 months from the manufacturing date.
Verichem Laboratories, 800-552-5859

