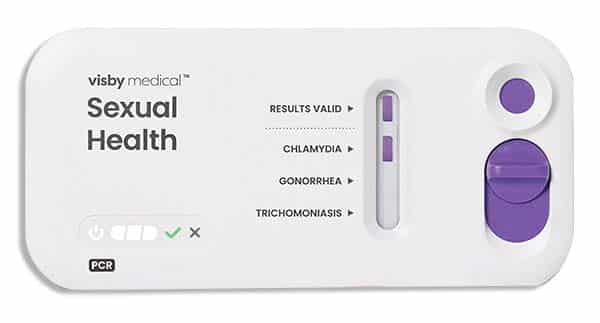
April 2023—Visby Medical has received 510(k) clearance and was granted a CLIA waiver from the FDA for its second-generation Sexual Health test. The point-of-care test uses PCR technology to detect sexually transmitted infections caused by Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis. Visby says the new device features improvements in workflow, manufacturability, and reliability. Test accuracy is about 97 percent and results are available in less than 30 minutes.
Read More »Home >> Tag Archives: Visby Medical
Visby POC COVID-19 PCR test gets CLIA waiver
March 2021—Visby Medical announced FDA authorization of the company’s rapid PCR COVID-19 test for use at the point of care by organizations with a CLIA certificate of waiver.
Read More »EUA for personal PCR device for COVID-19 testing
November 2020—The FDA has issued an emergency use authorization for Visby Medical’s single-use personal PCR device, a rapid test for detecting COVID-19.
Read More »Visby Medical’s personal PCR device for COVID-19 testing gets EUA
Oct. 23, 2020—The FDA has issued an emergency use authorization for Visby Medical’s single-use personal PCR device, a rapid test for detecting COVID-19.
Read More » CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management